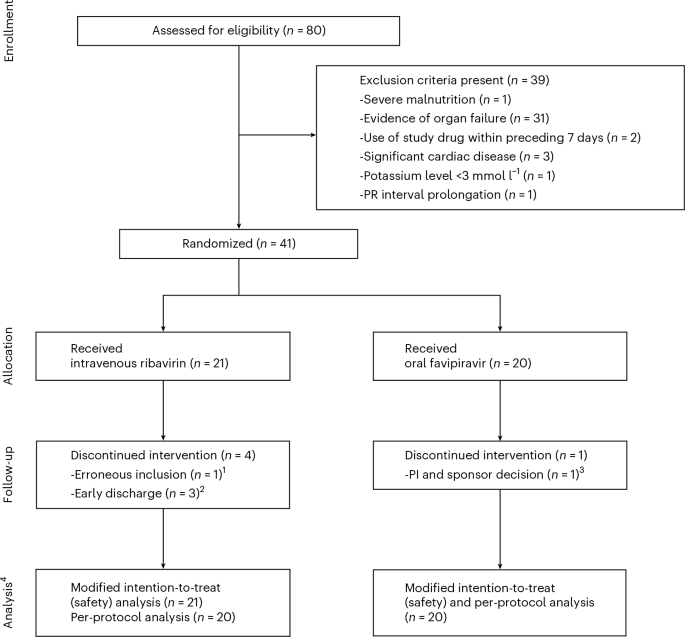
A new phase 2, open-label, randomized controlled trial published in Nature Medicine indicates that favipiravir is a safe, well-tolerated treatment alternative for mild-to-moderate Lassa fever. Conducted in Nigeria, the study compares the drug against the current standard of care, ribavirin, marking a critical step in addressing this viral hemorrhagic fever.
In Plain English: The Clinical Takeaway
- The Drug: Favipiravir is an antiviral medication that prevents the virus from replicating, potentially offering a more manageable side-effect profile than current treatments.
- The Findings: This study suggests the drug is safe for patients with mild to moderate cases, providing a necessary alternative for those who may not tolerate standard therapy.
- Next Steps: While promising, What we have is a mid-stage trial; larger phase 3 studies are required to confirm its effectiveness before it becomes a global standard of care.
The Mechanism of Action: How Favipiravir Targets Lassa
Lassa fever is caused by the Lassa virus (LASV), an arenavirus endemic to parts of West Africa. The virus primarily spreads through contact with the urine or feces of infected Mastomys natalensis rodents. Clinically, the disease presents with a range of symptoms, from mild malaise to severe hemorrhagic fever, which carries a high case-fatality rate if left untreated.
Favipiravir functions as a selective inhibitor of viral RNA-dependent RNA polymerase (RdRp). In simpler terms, it acts as a molecular “saboteur.” By mimicking the building blocks of the virus’s genetic material, the drug forces the virus to incorporate it into its own RNA chain during replication. This results in “lethal mutagenesis”—essentially, the virus creates non-functional copies of itself that cannot spread or survive. Unlike ribavirin, which has been the historical standard but is often associated with significant adverse effects like hemolytic anemia (the destruction of red blood cells), favipiravir’s mechanism is highly targeted, potentially sparing the patient from systemic toxicity.
Bridging the Epidemiological Gap: Global Regulatory Implications
The transition from a phase 2 trial to clinical practice requires rigorous regulatory scrutiny. While this study provides the evidence needed to move forward, the path to approval by bodies like the FDA or the EMA involves navigating complex safety data. For patients in West Africa, the primary challenge remains the “last mile” of healthcare delivery—ensuring that rapid diagnostic testing is available early enough in the disease course to administer antivirals effectively.

“The development of favipiravir for Lassa fever represents a significant shift toward targeted antiviral therapy. However, we must remain cautious; an open-label design, while necessary in outbreak settings, requires validation through larger, double-blind, placebo-controlled trials to definitively establish mortality benefits,” notes Dr. Oyewale Tomori, a virologist and former President of the Nigerian Academy of Science.
The trial was supported by the Coalition for Epidemic Preparedness Innovations (CEPI), a global partnership that funds research to develop vaccines and therapies for emerging infectious diseases. Transparency in funding is essential for maintaining trust, as it ensures that the research remains focused on public health utility rather than commercial interests.
| Feature | Ribavirin (Standard) | Favipiravir (Investigational) |
|---|---|---|
| Mechanism | Inhibits viral RNA synthesis | Selective RdRp inhibitor |
| Known Side Effects | Hemolytic anemia, nausea | Generally well-tolerated |
| Current Trial Status | Standard of Care | Phase 2 Complete |
| Administration | Intravenous/Oral | Oral |
Clinical Efficacy and the Path to Phase 3
In this phase 2 trial, researchers monitored patient outcomes to ensure the drug was both pharmacokinetically stable and clinically effective. A key metric was the time to viral clearance and the reduction in severity scores. Because the trial was open-label—meaning both the researchers and the patients knew which drug was being administered—there is a potential for observer bias. This is why the scientific community emphasizes the necessity of upcoming phase 3 trials, which utilize double-blind methodologies to eliminate such biases.
The global health community is looking at these results to determine if favipiravir can be repurposed for other hemorrhagic fevers, such as Ebola or Marburg, where RdRp inhibition is also a viable therapeutic target. By creating a standardized, portable, and oral treatment option, we move closer to a future where Lassa fever is no longer a death sentence in rural, resource-limited settings.
Contraindications & When to Consult a Doctor
Favipiravir is not a prophylactic; It’s a therapeutic agent for confirmed cases. It is strictly contraindicated in individuals with known hypersensitivity to the drug and is generally not recommended for pregnant women due to potential teratogenicity (the ability to cause birth defects) observed in animal studies. Patients presenting with fever, pharyngitis, or unexplained bleeding in endemic regions must seek immediate medical evaluation at a designated isolation center. Do not attempt to source or use experimental antivirals without direct supervision from infectious disease specialists, as inappropriate use can lead to viral resistance and ineffective treatment.
Conclusion
The Nature Medicine findings offer a beacon of hope for managing Lassa fever, a disease that has historically lacked sufficient therapeutic options. By refining our approach to RdRp inhibition, we are not only improving outcomes for Lassa patients but also building a framework for how we respond to future viral threats. We await the results of subsequent phase 3 trials with the expectation that they will confirm these safety findings and provide the definitive mortality data required for global regulatory endorsement.
References
- Favipiravir for Lassa fever: an open-label, randomized controlled phase 2 trial (Nature Medicine, 2026)
- World Health Organization: Lassa Fever Fact Sheet
- The Lancet Infectious Diseases: Advances in Viral Hemorrhagic Fever Therapeutics
- Mechanism of Action of RNA-Dependent RNA Polymerase Inhibitors (PubMed)