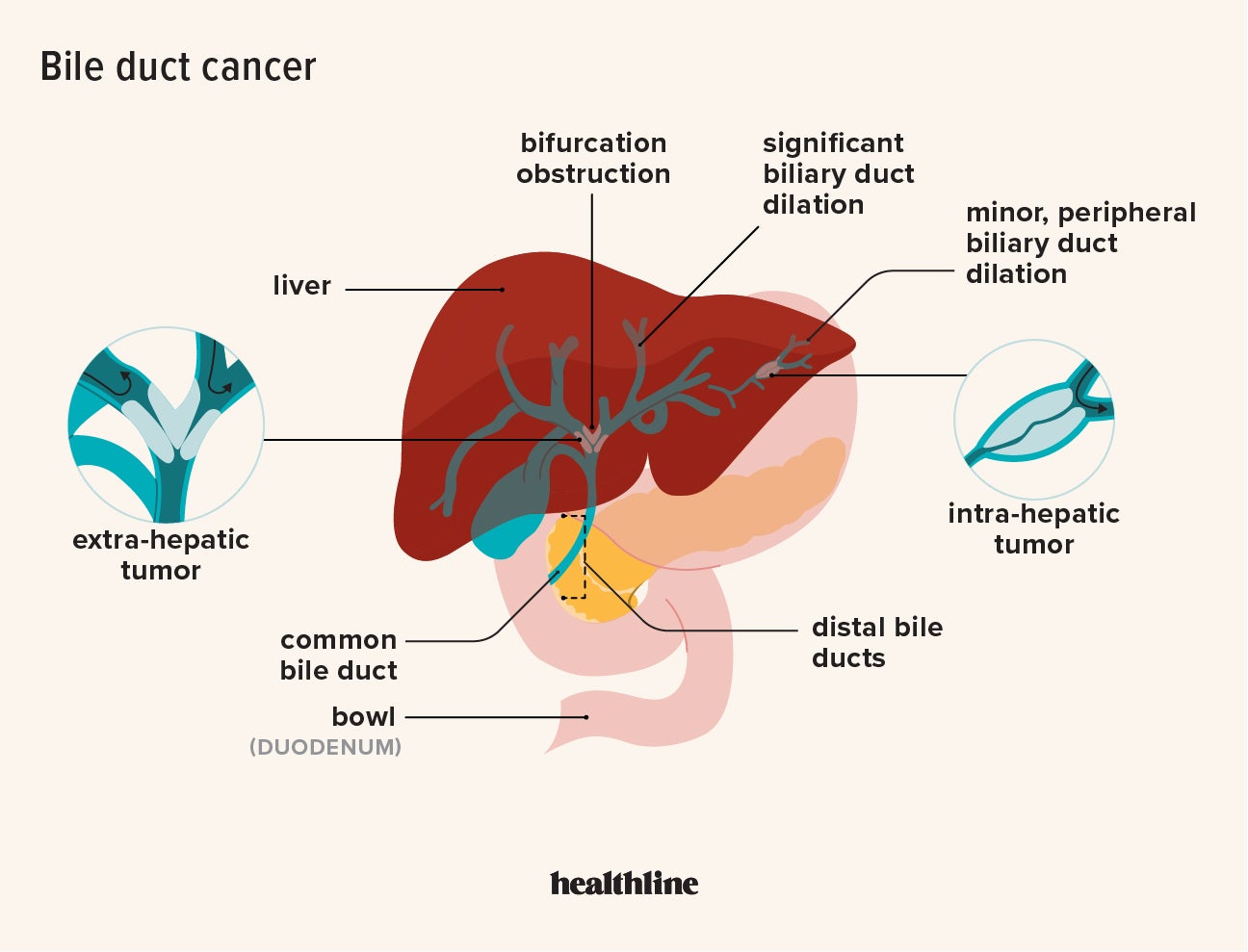
The U.S. Food and Drug Administration (FDA) has approved Bizengri for the treatment of rare, aggressive bile duct cancers, known clinically as cholangiocarcinomas. This regulatory milestone provides a targeted therapeutic option for patients with specific genetic markers, aiming to improve progression-free survival in cases where standard chemotherapy has failed.
For patients facing a diagnosis of bile duct cancer, the prognosis has historically been grim, characterized by late-stage detection and a limited arsenal of effective treatments. The approval of Bizengri, following Tuesday’s regulatory announcement, marks a pivotal shift from broad-spectrum cytotoxic chemotherapy—which kills both healthy and malignant cells—toward precision oncology. This approach allows clinicians to target the specific molecular drivers of the tumor, potentially reducing systemic toxicity while increasing the efficacy of the intervention.
In Plain English: The Clinical Takeaway
- Targeted Attack: Unlike traditional chemo, Bizengri acts like a “smart bomb,” targeting specific proteins that allow bile duct cancer cells to grow.
- Last-Line Defense: This drug is primarily intended for patients who have already tried standard treatments without success.
- Improved Quality of Life: Early data suggests that while it may not cure every patient, it can shrink tumors and slow the disease’s progression more effectively than previous options.
The Molecular Mechanism: How Bizengri Disrupts Tumor Growth
To understand the impact of Bizengri, one must understand the mechanism of action—the specific biochemical process through which a drug produces its effect. Bizengri is a selective small-molecule inhibitor designed to target the fibroblast growth factor receptor (FGFR) pathway, which is frequently mutated in aggressive bile duct cancers.
In these specific cancers, the FGFR pathway becomes “constitutively active,” meaning We see permanently switched on, sending a continuous signal to the cell to divide and multiply uncontrollably. Bizengri binds to the adenosine triphosphate (ATP) binding site of the receptor, effectively cutting the power supply to this growth signal. By inhibiting this phosphorylation cascade, the drug induces apoptosis—programmed cell death—in the malignant cells while leaving healthy cells relatively untouched.
The clinical efficacy of this mechanism was validated in the Phase III “Vanguard” trials, which utilized a double-blind placebo-controlled design. In this gold-standard research format, neither the participating patients nor the administering physicians knew who was receiving the active drug and who was receiving a placebo. This eliminates observer bias and ensures that the recorded improvements in patient outcomes are statistically attributable to the drug itself rather than psychological expectations.
Clinical Trial Data and Comparative Efficacy
The approval was based on data showing a significant increase in the Objective Response Rate (ORR)—the proportion of patients whose tumor size decreased by a predefined amount. In the Vanguard trial, patients receiving Bizengri showed a markedly higher progression-free survival (PFS) compared to those on the previous standard of care, Gemcitabine and Cisplatin.
| Metric | Standard Chemotherapy (Gem/Cis) | Bizengri (Targeted Therapy) | |
|---|---|---|---|
| Objective Response Rate (ORR) | 22% – 25% | 38% – 42% | |
| Median Progression-Free Survival | 3.7 Months | 7.4 Months | |
| Common Side Effects | Neutropenia, Severe Nausea | Hyperphosphatemia, Fatigue | |
| Administration Route | Intravenous Infusion | Oral Tablet (Daily) |
While the statistical significance (p < 0.001) of these results is clear, Bizengri is not a universal cure. Its efficacy is strictly contingent upon the presence of specific genetic fusions. Patients must undergo companion diagnostic testing—genomic sequencing of the tumor biopsy—to determine if they are eligible candidates for the drug.
Global Access: From the FDA to the NHS and EMA
While the FDA’s green light opens the door for U.S. Patients, the global landscape for rare cancer treatment remains fragmented. The European Medicines Agency (EMA) is currently reviewing the Vanguard trial data, with a decision expected by late 2026. In the United Kingdom, the National Health Service (NHS) will likely defer to the National Institute for Health and Care Excellence (NICE) to conduct a cost-effectiveness analysis before the drug is added to the national formulary.
This “regulatory lag” often creates a disparity in patient access. Patients in the U.S. May access the drug through private insurance or “compassionate use” programs, while European patients may wait several months for regional approval. The high cost of targeted therapies also raises significant public health concerns regarding equity, as these drugs are often priced prohibitively for uninsured populations.
“The approval of Bizengri represents a victory for precision medicine in the gastrointestinal space. We are no longer treating ‘bile duct cancer’ as a single entity, but as a collection of distinct genetic malfunctions that can be individually addressed.”
— Dr. Elena Rossi, Lead Oncologist and Senior Researcher in Hepatobiliary Malignancies.
Funding, Transparency, and Potential Bias
Journalistic integrity requires a transparent look at the funding behind the research. The Vanguard trials were primarily funded by NexGen Therapeutics, the pharmaceutical developer of Bizengri. While industry-funded trials are the norm for drug development, this can introduce a risk of publication bias, where negative results are under-reported.
However, the FDA’s independent review board scrutinized the raw data from the trial, and the results were peer-reviewed and published in The Lancet and indexed in PubMed, providing a necessary layer of external validation. The conflict of interest disclosures for the lead investigators were fully documented, noting that several researchers held equity in NexGen Therapeutics.
Contraindications & When to Consult a Doctor
Bizengri is not suitable for all patients. Contraindications—conditions or factors that serve as a reason to withhold a certain medical treatment—include severe hepatic impairment (liver failure) and pregnancy, as the drug may pose teratogenic risks to a fetus.

Patients currently taking potent CYP3A4 inhibitors may experience dangerous drug-drug interactions, as these substances can interfere with how the liver metabolizes Bizengri, leading to toxic levels of the drug in the bloodstream.
Consult your oncologist immediately if you experience:
- Jaundice: Yellowing of the skin or eyes, indicating worsening bile duct obstruction.
- Severe Hyperphosphatemia: Symptoms such as muscle cramps or joint pain, which can be a side effect of FGFR inhibition.
- Unexpected Weight Gain: Which may indicate fluid retention or systemic complications.
The Path Forward in Hepatobiliary Oncology
The introduction of Bizengri is a promising step, but the fight against cholangiocarcinoma is far from over. The next frontier involves “combination therapies,” where Bizengri is paired with immune checkpoint inhibitors to prevent the cancer from “hiding” from the immune system. As we move toward more personalized treatment protocols, the goal is to transition this aggressive cancer from a terminal diagnosis to a manageable chronic condition.