The U.S. Food and Drug Administration (FDA) has approved a new once-daily medication for the treatment of HIV-1, aiming to simplify antiretroviral therapy (ART). Alongside this medical advancement, health experts are emphasizing the impact of specific dietary triggers—namely high-sodium foods and stimulants—on systemic hypertension.
For millions living with HIV, the burden of “pill fatigue”—the psychological and physical exhaustion resulting from complex medication regimens—often leads to decreased adherence, which in turn increases the risk of viral rebound and drug resistance. This regulatory approval represents a shift toward “long-acting” philosophy, reducing the daily cognitive load for patients whereas maintaining viral suppression.
In Plain English: The Clinical Takeaway
- Simplified Dosing: A new FDA-approved option allows patients to manage HIV with a single daily dose, reducing the likelihood of missed medications.
- Blood Pressure Triggers: Common dietary habits, specifically excessive salt and certain caffeinated drinks, can acutely spike blood pressure, complicating cardiovascular health.
- Consistency is Key: Whether managing a chronic virus or hypertension, the primary goal is maintaining a steady state of medication and lifestyle control to prevent organ damage.
The Pharmacology of Viral Suppression: Mechanism of Action
The newly approved therapy focuses on achieving an undetectable viral load, the gold standard of HIV care. Most modern once-daily regimens utilize a combination of drug classes, such as Integrase Strand Transfer Inhibitors (INSTIs) and Nucleoside Reverse Transcriptase Inhibitors (NRTIs).
The mechanism of action—the specific biochemical process by which a drug produces its effect—involves blocking the virus’s ability to integrate its genetic material into the host cell’s DNA. By halting this replication cycle, the medication prevents the virus from spreading to new CD4 T-cells, the white blood cells critical for immune function.
This approach is typically validated through double-blind placebo-controlled trials
, a rigorous scientific standard where neither the participants nor the researchers know who is receiving the active drug versus a placebo. This eliminates bias and ensures that the observed efficacy is due to the drug itself and not a psychological effect.
“The transition toward simplified, high-efficacy regimens is not merely about convenience; it is a critical public health strategy to eliminate the reservoirs of the virus and move closer to the goal of ending the epidemic.” Dr. Anthony Fauci, former Director of the NIAID
Global Access and Geo-Epidemiological Impact
While the FDA provides the initial regulatory green light in the United States, the global impact depends on the European Medicines Agency (EMA) and the World Health Organization (WHO) guidelines. In regions like Sub-Saharan Africa, where the HIV burden is highest, the transition to once-daily or long-acting injectables is a game-changer for healthcare infrastructure.

The integration of these drugs into national health systems, such as the NHS in the UK or public clinics in Egypt and Morocco, requires a balance between cost, and accessibility. Funding for these innovations is predominantly driven by pharmaceutical research and development (R&D) grants, often in partnership with government entities like the National Institutes of Health (NIH).
| Metric | Traditional Multi-Drug Regimens | New Once-Daily/Long-Acting Options |
|---|---|---|
| Dosing Frequency | Multiple times per day | Once daily or monthly/quarterly |
| Adherence Rate | Moderate (Risk of pill fatigue) | High (Simplified schedule) |
| Primary Goal | Viral Load Suppression | Viral Load Suppression + Quality of Life |
| Common Side Effects | Nausea, Insomnia, Fatigue | Mild Injection Site Reaction or GI distress |
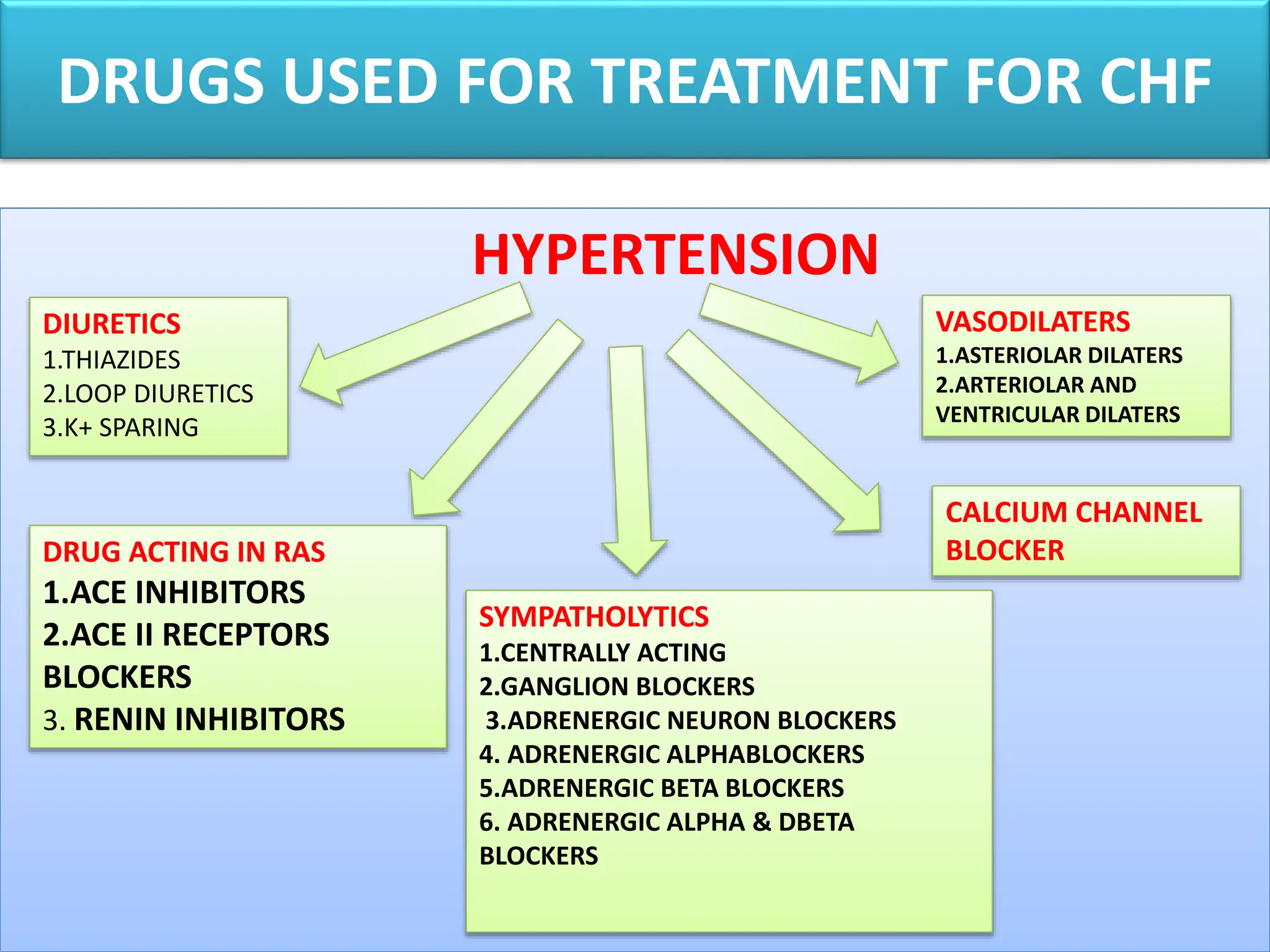
The Hypertension Link: Dietary Triggers and Vascular Stress
Parallel to the news of HIV treatment is the urgent reminder regarding hypertension (high blood pressure). Hypertension is often a “silent killer” because it lacks obvious symptoms until significant damage has occurred in the heart or kidneys.
Certain foods and drinks act as potent triggers for blood pressure spikes. High-sodium processed foods lead to fluid retention, increasing the volume of blood the heart must pump, which elevates pressure against the arterial walls. Excessive intake of stimulants—such as high-caffeine energy drinks or nicotine—triggers the release of adrenaline, causing vasoconstriction (the narrowing of blood vessels), which further increases pressure.
According to the World Health Organization (WHO), reducing salt intake to less than 5 grams per day can significantly lower the risk of cardiovascular events. This lifestyle intervention is complementary to pharmacological treatments like ACE inhibitors or Beta-blockers.
Contraindications & When to Consult a Doctor
Medical treatments are not universal; contraindications—specific situations in which a drug should not be used because it may be harmful—must be screened by a professional.
- HIV Medication: Patients with severe renal (kidney) impairment or hepatic (liver) failure may require dosage adjustments. Those with a history of hypersensitivity to the drug’s components should avoid use.
- Hypertension: Individuals with chronic kidney disease must be extremely cautious with potassium-rich salt substitutes, as they can lead to hyperkalemia (dangerously high potassium levels).
Seek immediate medical attention if you experience any of the following “red flag” symptoms:
- Sudden, severe headache accompanied by blurred vision (potential hypertensive crisis).
- Chest pain or shortness of breath.
- Unexplained fever or rash following the start of a new antiretroviral therapy.
The Path Forward: Integration of Care
The convergence of simplified HIV therapy and a heightened focus on cardiovascular health reflects a broader trend in medicine: the shift toward holistic, patient-centric care. As HIV becomes a manageable chronic condition rather than a terminal illness, the focus is shifting toward managing the long-term comorbidities, including hypertension and metabolic syndrome.
The goal for the coming years is to ensure that these innovations reach the most vulnerable populations, bridging the gap between regulatory approval in wealthy nations and actual bedside delivery in developing regions. Through the efforts of agencies like the CDC and The Lancet‘s published research, the global medical community continues to refine the balance between efficacy and accessibility.
References
- U.S. Food and Drug Administration (FDA) – Center for Drug Evaluation and Research
- World Health Organization (WHO) – Global Health Estimates on Hypertension and HIV
- The Lancet – HIV/AIDS Research and Clinical Trials Archive
- Centers for Disease Control and Prevention (CDC) – HIV Treatment Guidelines
- PubMed – National Library of Medicine (Peer-Reviewed Pharmacological Studies)

