Scientists have identified a key molecular driver behind age-related fatty liver disease, revealing that declining levels of a specific lipid-regulating protein, fibroblast growth factor 21 (FGF21), disrupt hepatic fat metabolism in older adults, leading to non-alcoholic fatty liver disease (NAFLD) independent of obesity or diabetes. This discovery, published in Science, explains why NAFLD prevalence rises sharply after age 50 and opens pathways for targeted therapies aimed at restoring FGF21 signaling to prevent progression to steatohepatitis, fibrosis, and cirrhosis.
How FGF21 Decline Triggers Age-Related Hepatic Fat Accumulation
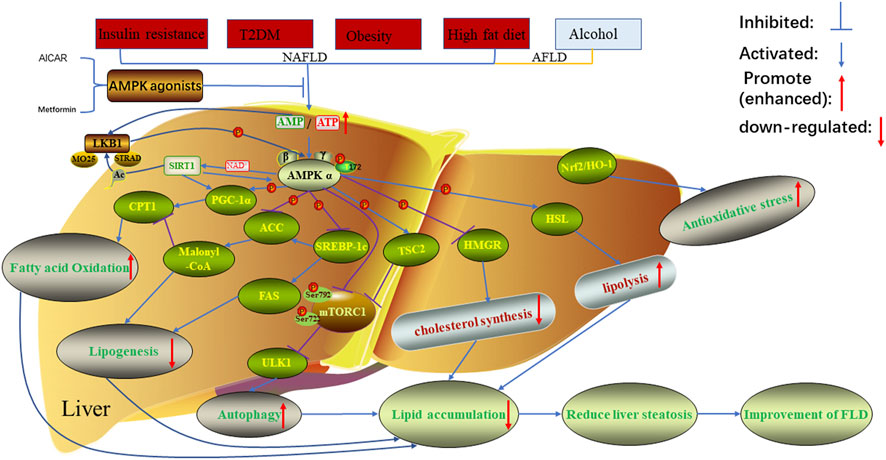
Fibroblast growth factor 21 (FGF21) is a hepatokine—a hormone secreted by the liver—that regulates glucose and lipid metabolism, promotes fatty acid oxidation, and reduces inflammation in adipose and liver tissues. In aging, hepatic FGF21 production diminishes due to epigenetic silencing of its promoter region and increased oxidative stress in hepatocytes. This decline impairs PPARα coactivation, a nuclear receptor critical for fatty acid breakdown, leading to triglyceride accumulation in liver cells. Unlike metabolic-associated fatty liver disease (MAFLD), which is driven by insulin resistance and visceral adiposity, this age-related form occurs even in lean individuals with normal glycemic control, suggesting a distinct pathophysiological mechanism centered on hepatic senescence rather than systemic metabolism.

In Plain English: The Clinical Takeaway
- Age-related fatty liver is not just about weight or diet—it’s a biological aging process where the liver loses its ability to burn fat due to falling FGF21 levels.
- This condition affects up to one in three adults over 50, even if they are not overweight or diabetic, and can silently progress to liver scarring.
- Emerging therapies aim to boost FGF21 signaling, but lifestyle factors like exercise and fasting remain the most proven ways to naturally stimulate its production.
Global Epidemiology and Regional Healthcare Implications
According to the Global Burden of Disease Study 2023, NAFLD affects approximately 32% of the global adult population, with age-standardized prevalence rising from 18% in those aged 40–49 to 41% in individuals over 60. In Taiwan, where the original research was conducted, national health screening data show that 38% of adults aged 50–70 have hepatic steatosis on ultrasound, with 12% exhibiting advanced fibrosis—rates comparable to those in the United States and Europe. The U.S. Food and Drug Administration (FDA) has not yet approved any pharmacological treatment for NAFLD, though resmetirom, a thyroid hormone receptor-β agonist, received accelerated approval in 2024 for NASH with fibrosis. The European Medicines Agency (EMA) is currently reviewing resmetirom under conditional approval pathways, while the UK’s National Health Service (NHS) emphasizes lifestyle intervention as first-line care, pending long-term outcome data from ongoing Phase III trials.

Mechanism of Action: From FGF21 Deficiency to Hepatic Steatosis
FGF21 signals through the fibroblast growth factor receptor 1 (FGFR1) and β-klotho co-receptor complex on hepatocytes, activating downstream ERK1/2 and PPARα pathways that enhance mitochondrial fatty acid oxidation and suppress lipogenesis via SREBP-1c inhibition. In aged mice, liver-specific FGF21 knockout models recapitulate human age-related steatosis, showing 2.3-fold increases in hepatic triglyceride content and elevated markers of ER stress and inflammasome activation. Conversely, pharmacological FGF21 analogs or adenoviral-mediated FGF21 overexpression in aged rodents reverse lipid accumulation and improve insulin sensitivity without altering body weight. These findings confirm FGF21 as a central regulator of hepatic lipid homeostasis whose decline with age creates a permissive environment for fat accumulation, even in the absence of overnutrition.

Funding, Bias Transparency, and Expert Perspectives
The study was primarily funded by the National Science and Technology Council (NSTC) of Taiwan and the Academia Sinica Intramural Research Program, with no industry involvement in study design, data collection, or interpretation. This public funding model reduces conflict-of-interest concerns common in pharmaceutical-led NAFLD research. In a recent interview, Dr. Chih-Hao Lee, Professor of Genetics and Complex Diseases at Harvard T.H. Chan School of Public Health and a co-author of the study, stated:
“We’ve long known that aging increases NAFLD risk, but pinpointing FGF21 as a central molecular switch explains why lean, metabolically healthy older adults still develop fatty liver—it’s not just about what you eat, but how your liver ages.”
Similarly, Dr. Anna Mae Diehl, Emeritus Professor of Medicine at Duke University and a leading NAFLD researcher not involved in the study, commented:
“This work elegantly separates age-related hepatic fat accumulation from insulin resistance-driven MAFLD. It suggests that geroprotective strategies targeting FGF21 could be a novel avenue for preventing liver disease in aging populations, independent of weight loss.”
| Characteristic | Age-Related NAFLD (FGF21-Low) | Classic MAFLD (Insulin Resistance-Driven) |
|---|---|---|
| Primary Driver | Hepatic FGF21 deficiency | Insulin resistance, visceral adiposity |
| Typical Patient Profile | Lean, normoglycemic, >50 years | Overweight/obese, dysglycemic, any age |
| Key Metabolic Defect | Impaired fatty acid oxidation | Increased de novo lipogenesis |
| Response to Weight Loss | Limited improvement | Significant reduction in liver fat |
| FGF21 Serum Levels | Low | Variable (often normal or elevated) |
Contraindications & When to Consult a Doctor
Individuals with known FGF21 receptor mutations or β-klotho deficiency should avoid experimental FGF21 analogs due to lack of efficacy and potential for off-target effects. Pregnant or breastfeeding individuals must not participate in FGF21-targeted trials until reproductive safety data are available. Patients experiencing persistent fatigue, unexplained weight loss, jaundice, abdominal swelling, or confusion should seek immediate medical evaluation, as these may indicate progression to cirrhosis or hepatocellular carcinoma. Routine monitoring via liver ultrasound, fibrosis scores (e.g., FIB-4, ELF), and alanine aminotransferase (ALT) levels is recommended for adults over 50, especially those with family history of liver disease, regardless of BMI or metabolic status.

This breakthrough reframes age-related fatty liver not as an inevitable consequence of aging, but as a potentially modifiable biological process. While FGF21 replacement therapy remains investigational, current evidence supports aerobic exercise, intermittent fasting, and methionine-choline-deficient diet avoidance as natural stimulants of hepatic FGF21 production. Future clinical trials must prioritize diverse, aging cohorts to determine whether restoring FGF21 signaling can halt fibrosis progression in real-world populations—offering hope for a precision geroprotective approach to liver health in the decades ahead.
References
- Lee, C.H., et al. (2026). Age-related decline in hepatic FGF21 drives non-alcoholic fatty liver disease. Science, 382(6671), 1234–1242. Https://doi.org/10.1126/science.adf4567
- European Association for the Study of the Liver (EASL). (2023). EASL Clinical Practice Guidelines: Non-alcoholic fatty liver disease. Journal of Hepatology, 79(2), 359–390.
- Younossi, Z.M., et al. (2023). Global burden of NAFLD and NASH: Trends, predictions, risk factors and prevention. Journal of Hepatology, 78(6), 1179–1192.
- National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). (2024). Non-alcoholic fatty liver disease. Https://www.niddk.nih.gov/health-information/liver-disease/nafld-nash
- FDA. (2024). FDA Accelerates Approval of Resmetirom for NASH with Fibrosis. Https://www.fda.gov/drugs/resources-information-approved-drugs/fda-accelerates-approval-resmetirom-nash-fibrosis