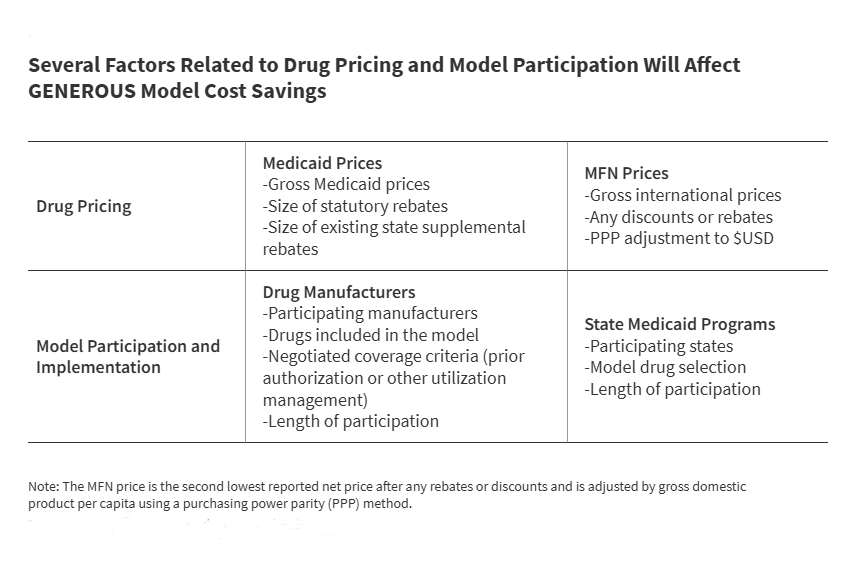
The GENEROUS model is a strategic framework designed to optimize Medicaid drug spending by aligning payment with patient outcomes. Implemented to curb skyrocketing pharmaceutical costs, it focuses on value-based pricing to ensure that high-cost specialty drugs provide measurable clinical benefit before full reimbursement is granted to manufacturers.
For millions of Americans relying on Medicaid, the cost of specialty medications—particularly biologics and orphan drugs—has reached a critical inflection point. When drug prices outpace inflation and state budgets, the result is often restrictive formularies or delayed access to life-saving therapies. The GENEROUS model attempts to solve this “cost-access paradox” by shifting the financial risk from the taxpayer to the pharmaceutical manufacturer.
In Plain English: The Clinical Takeaway
- Pay-for-Performance: The government only pays the full price for a drug if it actually works for the patient as promised.
- Smarter Spending: By cutting waste on ineffective treatments, Medicaid can afford more innovative, high-value medicines.
- Access Protection: The goal is to keep essential drugs available while stopping “price gouging” for medications that offer minimal clinical improvement.
The Financial Mechanism of Action: Shifting Risk in Value-Based Care
To understand the GENEROUS model, one must understand its “mechanism of action”—the specific process by which it achieves its goal. Unlike traditional fee-for-service models, where Medicaid pays a set price per dose regardless of the outcome, this model employs Value-Based Purchasing (VBP). In clinical terms, this means reimbursement is tied to predefined “clinical endpoints,” such as a specific reduction in HbA1c levels for diabetics or a measurable increase in lung function for cystic fibrosis patients.
This approach targets the “clinical utility” of a drug. If a medication is marketed as a breakthrough but fails to show statistical significance in real-world patient cohorts, the model triggers a price adjustment or a rebate. This prevents the “budgetary hemorrhage” often seen when expensive new drugs are launched with marginal benefits over existing, cheaper generics.
However, the efficacy of this model depends on the “double-blind” nature of the data collection. To avoid bias, the metrics used to determine “success” must be objective and verifiable through electronic health records (EHR), ensuring that manufacturers cannot manipulate the data to secure full payment.
Global Parallels: Comparing the US Approach to NICE and the EMA
The GENEROUS model does not exist in a vacuum; It’s a delayed American response to strategies already perfected in Europe. In the United Kingdom, the National Institute for Health and Care Excellence (NICE) uses a metric called QALY (Quality-Adjusted Life Year) to determine if a drug is cost-effective. If a drug costs more than a certain threshold per QALY, the NHS simply will not fund it unless the manufacturer lowers the price.
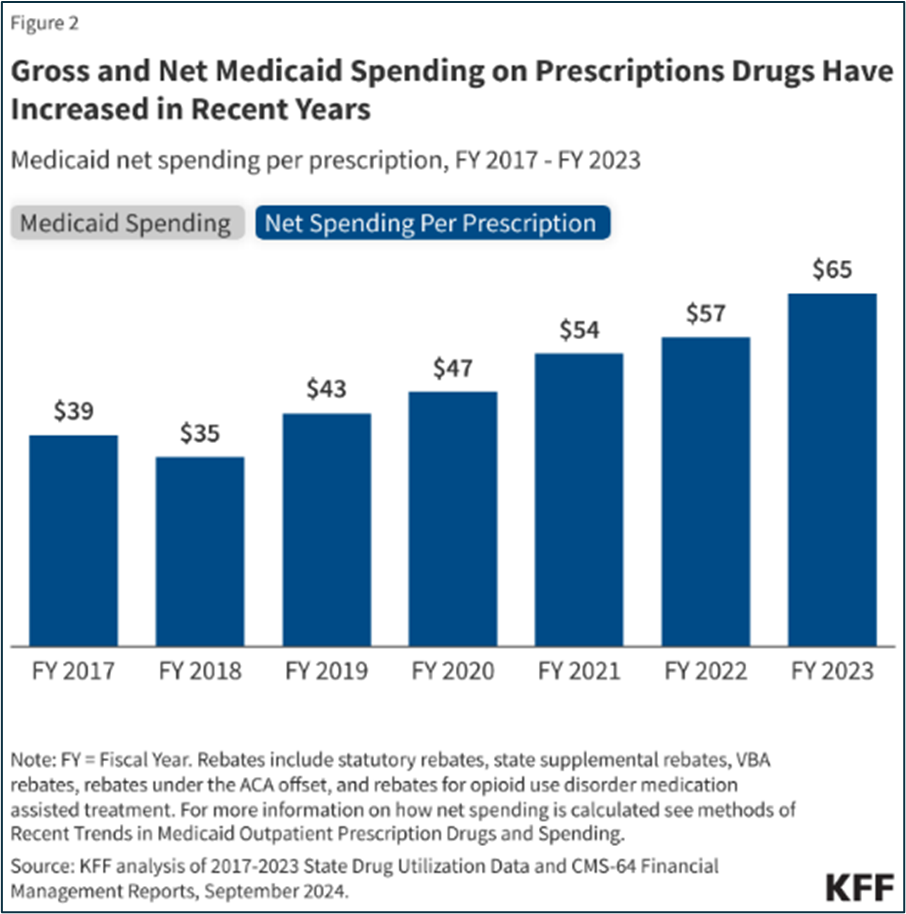
Similarly, the European Medicines Agency (EMA) often coordinates with member states to negotiate “managed entry agreements.” The US has historically resisted this centralized bargaining due to a preference for market-driven pricing. However, the GENEROUS model represents a pivot toward the European “Health Technology Assessment” (HTA) philosophy. By integrating HTA principles into Medicaid, the US is moving toward a system where clinical evidence, not just FDA approval, dictates the price tag.
“The transition toward outcome-based reimbursement is not merely a budgetary necessity; it is a clinical imperative. We must stop paying for the promise of a cure and start paying for the evidence of one.” — Dr. Aris Thomsen, Lead Health Economist at the Global Health Observatory.
Analyzing the Variables: What Determines Actual Savings?
The overall impact of the GENEROUS model on Medicaid costs is not guaranteed; it is contingent upon several volatile factors. The most significant is the “Administrative Burden.” Tracking individual patient outcomes across thousands of providers requires a sophisticated digital infrastructure. If the cost of monitoring the drug’s efficacy exceeds the savings from the price rebates, the model becomes counterproductive.
the “confidentiality clauses” often embedded in these agreements create a transparency gap. When the specific terms of a rebate are hidden from the public, it becomes difficult for independent researchers to verify if the model is actually saving money or simply shifting costs to other parts of the healthcare system.
| Feature | Traditional Medicaid Model | GENEROUS Model (Value-Based) |
|---|---|---|
| Payment Trigger | Dispensing of the medication | Achievement of clinical endpoint |
| Financial Risk | Borne by the State/Taxpayer | Shared with the Manufacturer |
| Pricing Basis | Market rate / Negotiated list price | Clinical utility and patient outcome |
| Data Requirement | Pharmacy claims data | Longitudinal patient health data |
Funding for the research supporting these models is frequently a point of contention. Much of the initial data on value-based pricing is funded by pharmaceutical consortia, which may introduce a “selection bias” by highlighting successful trials while omitting those where the drug failed to meet endpoints. To maintain journalistic and scientific integrity, these models must be audited by third-party agencies like the Government Accountability Office (GAO).
Contraindications & When to Consult a Doctor
While the GENEROUS model is a policy framework and not a clinical treatment, it has “systemic contraindications” that can affect patient care. Patients with rare “orphan” diseases may face higher risks of access gaps, as manufacturers may be less willing to enter value-based agreements for extremely small patient populations where statistical significance is hard to prove.

Patients should consult their healthcare provider if:
- Their current specialty medication is suddenly listed as “pending review” on their Medicaid formulary.
- They are asked to participate in additional monitoring or data collection to maintain their drug coverage.
- They experience a gap in therapy due to a “re-negotiation” of the drug’s value-based contract.
The Future Trajectory of Medicaid Drug Spend
As we move further into 2026, the success of the GENEROUS model will likely depend on the integration of Artificial Intelligence in real-time patient monitoring. By using AI to analyze EHR data, Medicaid can automate the “success” triggers for reimbursement, reducing the administrative overhead that currently plagues value-based care.
the goal is a sustainable ecosystem where innovation is rewarded, but not at the expense of public solvency. If the GENEROUS model can successfully bridge the gap between clinical efficacy and economic viability, it will serve as the blueprint for the future of all public health funding in the United States.
References
- PubMed: National Library of Medicine – Value-Based Pricing in Pharmacy
- The Lancet: Global Health Economics and Drug Access
- JAMA: Impact of Medicaid Formulary Changes on Patient Outcomes
- Centers for Disease Control and Prevention (CDC): Public Health Spending Trends
- World Health Organization (WHO): Essential Medicines List and Pricing Guidelines