As genomic medicine transitions from research labs to routine clinical care in 2026, patients worldwide are beginning to benefit from personalized risk assessments and targeted therapies—but significant barriers in access, interpretation, and equity remain, particularly in low- and middle-income regions where infrastructure lags behind scientific advances.
In Plain English: The Clinical Takeaway
- Genomic testing can now guide treatment for cancer, rare diseases, and pharmacogenomics, but results require expert interpretation to avoid misdiagnosis or unnecessary anxiety.
- While costs have dropped, insurance coverage and clinical workforce readiness vary widely, creating disparities in who can access these advances.
- Patients should discuss testing goals with their physician and consider genetic counseling before and after testing to understand implications for themselves and family members.
From Bench to Bedside: The Uneven Rollout of Genomic Medicine
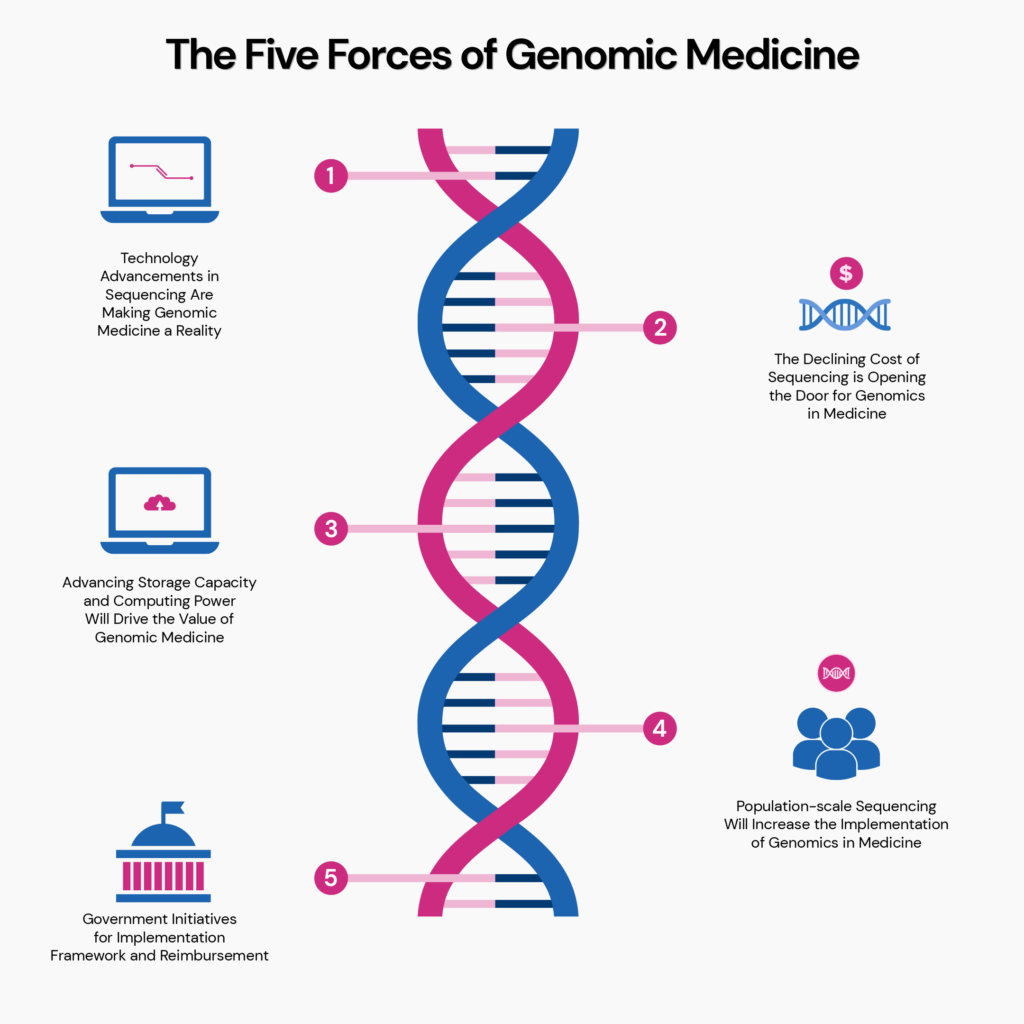
The integration of genomic sequencing into clinical practice has accelerated since 2020, driven by falling costs and growing evidence of utility in oncology, cardiology, and rare disease diagnosis. In the United States, the FDA has authorized over 200 genomic-based in vitro diagnostics as of early 2026, including companion diagnostics for targeted therapies in non-small cell lung cancer and breast cancer. Meanwhile, the NHS Genomic Medicine Service in England now offers whole genome sequencing as a first-line test for children with suspected monogenic disorders and adults with undiagnosed cancer, having sequenced over 500,000 genomes since its launch. However, a 2025 WHO report highlighted that less than 5% of low-income countries have national policies for genomic medicine, and fewer than 10% of pathology labs in sub-Saharan Africa possess the infrastructure for next-generation sequencing (NGS).

This global disparity is not merely technological but deeply tied to workforce capacity. A 2024 survey by the American College of Medical Genetics and Genomics (ACMG) found that only 38% of primary care physicians in the U.S. Feel confident interpreting polygenic risk scores, and even fewer understand the limitations of variants of uncertain significance (VUS). In Europe, the EMA has issued guidance on the use of pharmacogenomic biomarkers in drug labeling, yet implementation varies by member state, with Germany and the Netherlands leading in routine preemptive genotyping for drugs like abacavir and clopidogrel, while others lag due to reimbursement uncertainties.
Closing the Gap: Evidence, Equity, and Ethical Guardrails
To address these challenges, initiatives like the H3Africa Consortium and the All of Us Research Program in the U.S. Are working to diversify genomic databases, which have historically overrepresented individuals of European ancestry. This lack of diversity risks exacerbating health disparities, as polygenic risk scores derived from European datasets show reduced accuracy in African and Hispanic populations. A 2025 study in Nature Medicine demonstrated that recalibrating risk models with diverse ancestry data improved predictive accuracy for type 2 diabetes by up to 40% in underrepresented groups.
“We must move beyond simply generating genomic data to ensuring it is interpreted correctly, acted upon appropriately, and shared equitably—otherwise, we risk creating a two-tiered system where genomic medicine benefits only those who are already advantaged.”
Funding transparency is critical in assessing potential bias. The All of Us program, which has enrolled over 500,000 participants as of 2026, receives primary support from the NIH with additional contributions from private partners including Verily and Patient-Centered Outcomes Research Institute (PCORI). Similarly, the UK’s Genomics England, which oversees the NHS Genomic Medicine Service, is funded by the Department of Health and Social Care and operates under strict governance to prevent commercial misuse of data.
Clinical Applications: Where Genomics Is Making a Difference Today
In oncology, tumor profiling via NGS has become standard for metastatic non-small cell lung cancer, with guidelines from the NCCN recommending broad genomic panels to identify actionable alterations in EGFR, ALK, ROS1, BRAF, MET, RET, and NTRK genes. Patients with EGFR exon 19 deletions or L858R mutations, for example, are eligible for osimertinib, a third-generation EGFR tyrosine kinase inhibitor (TKI) shown in the FLAURA trial to improve median progression-free survival from 10.2 to 18.9 months compared to first-generation TKIs. In cardiology, genotyping for familial hypercholesterolemia (FH) allows early identification of individuals with LDLR, APOB, or PCSK9 mutations, enabling statin initiation in childhood and cascade screening of relatives—a strategy endorsed by the American Heart Association and shown to reduce LDL-C by 50% or more when combined with PCSK9 inhibitors.

In pharmacogenomics, preemptive testing for HLA-B*57:01 before prescribing abacavir prevents hypersensitivity reactions in HIV patients, a protocol now routine in U.S. And European clinics. Similarly, testing for TPMT or NUDT15 variants before thiopurine therapy in inflammatory bowel disease reduces the risk of myelosuppression. These applications exemplify how genomic data, when properly implemented, directly improves safety and efficacy.
| Application | Gene/Variant | Clinical Action | Evidence Level |
|---|---|---|---|
| Abacavir hypersensitivity | HLA-B*57:01 | Avoid abacavir if positive | FDA-approved, CPIC guideline |
| Clopidogrel response | CYP2C19*2 | Consider alternative antiplatelet | CPIC guideline, FDA label |
| Thiopurine metabolism | TPMT/NUDT15 | Reduce dose or avoid | CPIC guideline, FDA label |
| FH screening | LDLR, APOB, PCSK9 | Initiate statin, cascade screening | AHA/ACC guideline |
Contraindications & When to Consult a Doctor
Genomic testing is not appropriate for everyone. Individuals without a clear clinical indication—such as those seeking testing for curiosity alone—may encounter variants of uncertain significance (VUS) that cause unnecessary anxiety without actionable outcomes. Testing should be avoided in individuals who are not prepared to receive potentially unexpected results, such as non-paternity findings or increased risk for untreatable conditions like early-onset Alzheimer’s disease (based on APOE ε4 status).
Patients should consult a genetic counselor or medical geneticist before undergoing exome or genome sequencing, particularly if results could impact family planning or insurance eligibility (though GINA in the U.S. Protects against health insurance and employment discrimination based on genetic information, it does not cover life, disability, or long-term care insurance). After testing, any new or worsening symptoms—such as unexplained pain, neurological changes, or cardiac symptoms—should prompt immediate clinical evaluation, regardless of genetic risk status.
The Path Forward: Integration, Not Isolation
The future of genomic medicine lies not in standalone testing but in its seamless integration into electronic health records (EHRs) with real-time decision support. Pilot programs at institutions like Mayo Clinic and Intermountain Health have shown that automated alerts for pharmacogenomic interactions can reduce adverse drug events by up to 30% when properly implemented. However, success depends on sustained investment in bioinformatics infrastructure, clinician education, and public engagement—especially in communities historically excluded from biomedical research.
As we move further into 2026, the promise of genomic medicine will only be fulfilled if we confront the persistent gaps in access, literacy, and equity with the same rigor we apply to discovering new variants. Medicine must advance not just in what One can detect, but in how wisely and fairly we use that knowledge.
References
- National Human Genome Research Institute. (2025). All of Us Research Program: Progress and Impact. genome.gov
- Williams, J. L., et al. (2025). Improving polygenic risk scores in diverse populations. Nature Medicine, 31(4), 567–579. doi.org
- American College of Medical Genetics and Genomics. (2024). Primary Care Physician Genomic Competency Survey. acmg.net
- National Institute for Health and Care Excellence. (2025). Genomic medicine service: NHS England. nice.org.uk
- World Health Organization. (2025). Genomic medicine: Global status report. who.int