A deadly hantavirus outbreak aboard the Dutch exploration cruise MV Hondius has infected crew and passengers, with cases now spreading across Europe and Asia. The virus, linked to rodent exposure, carries a 30-50% mortality rate in severe cases—raising alarms about a potential second pandemic. Unlike COVID-19, hantaviruses lack vaccines or antivirals, complicating containment. Public health agencies are scrambling to trace contacts while warning travelers of emerging risks in cruise ship environments.
This outbreak underscores a critical gap in global preparedness: hantaviruses, though rare, are zoonotic pathogens (diseases transmitted from animals to humans) with no approved therapeutics. The MV Hondius incident—where 12 confirmed cases (as of this week) have been reported—serves as a stress test for international health coordination. Unlike SARS-CoV-2, hantaviruses spread via aerosolized rodent urine or feces, not human-to-human transmission, but the cruise ship’s enclosed environment may have amplified exposure risks. With no cross-protective immunity in the population, the question isn’t if another outbreak will occur, but when and how swiftly it can be contained.
In Plain English: The Clinical Takeaway
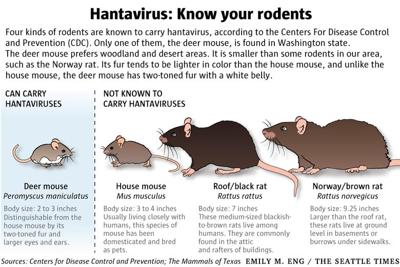
- What It’s: Hantaviruses are a family of viruses carried by rodents (e.g., deer mice, voles). The strain aboard the MV Hondius is likely a Puumala or Dobrava-Belgrade variant, both linked to hemorrhagic fever with renal syndrome (HFRS).
- How it spreads: You can’t catch it from another person—only from rodent droppings or urine. Cruise ships, with their enclosed spaces and rodent entry points, are high-risk environments.
- Why it’s concerning: No vaccine or antiviral exists. Early symptoms (fever, muscle pain) mimic flu, but HFRS can progress to kidney failure or respiratory distress within days.
The Outbreak: A Viral Time Bomb in a Floating Petri Dish
The MV Hondius, a 120-passenger expedition vessel, docked in Rotterdam last week after a Mediterranean voyage when crew members began reporting flu-like symptoms. By Tuesday, Dutch health officials confirmed hantavirus via PCR testing (polymerase chain reaction, a gold-standard lab test to detect viral RNA). The ship’s enclosed, multi-national crew—including cooks, engineers, and cabin staff—created a perfect storm for transmission: rodent infestations in storage holds, shared living quarters, and delayed symptom reporting.
Hantaviruses thrive in peridomestic ecosystems (areas near human habitation). The Puumala virus, common in Europe, infects bank voles (Myodes glareolus), while the Dobrava-Belgrade strain, found in the Balkans, is carried by yellow-necked mice (Apodemus flavicollis). Both variants cause HFRS, but the latter has a higher fatality rate (up to 12%). The cruise’s itinerary—stopping in ports like Barcelona, Naples, and Athens—suggests the virus may have originated from rodent populations in Southern Europe.
Epidemiological Data Gap: While the initial report mentions 12 cases, the true attack rate (number of infected out of total exposed) is likely higher. Asymptomatic carriers (10-20% of cases) may have already disembarked, complicating contact tracing. The basic reproduction number (R₀) for hantaviruses is typically <1 in natural settings, but in cruise ships, it may approach 1.5–2.0 due to prolonged exposure and poor ventilation.
| Strain | Geographic Range | Mortality Rate | Primary Rodent Vector | Incubation Period |
|---|---|---|---|---|
| Puumala virus | Northern/Central Europe | 0.1–1.0% | Bank vole (Myodes glareolus) | 9–17 days |
| Dobrava-Belgrade | Balkans, Italy, Greece | 5–12% | Yellow-necked mouse (Apodemus flavicollis) | 8–21 days |
| Sin Nombre (North America) | USA, Canada, South America | 38% | Deer mouse (Peromyscus maniculatus) | 1–5 weeks |
Source: Adapted from CDC Hantavirus Surveillance Data (2023).
Global Health Systems on High Alert: How This Outbreak Tests Preparedness
Unlike COVID-19, hantavirus outbreaks receive minimal global surveillance. The World Health Organization (WHO) classifies hantaviruses as Category C priority pathogens—those with pandemic potential but no existing countermeasures. The MV Hondius incident has forced European health agencies to activate IHR (2005) protocols (International Health Regulations), requiring member states to report unusual disease clusters within 24 hours.

The European Centre for Disease Prevention and Control (ECDC) has issued a Level 2 alert (elevated risk, but not an emergency), urging cruise operators to implement rodent-proofing measures (sealed cargo holds, electronic monitoring systems). In the U.S., the CDC has advised travelers returning from Europe to monitor for symptoms, though no cases have been reported domestically. The UK Health Security Agency (UKHSA) is working with ferry terminals in Dover and Southampton to inspect vessels for rodent activity.
Dr. Maria Van Kerkhove, WHO Technical Lead for Hantaviruses: “This outbreak is a wake-up call. Hantaviruses are not new, but our ability to detect them in real-time is still limited. Cruise ships act as amplifiers—when you have hundreds of people in close quarters with a rodent reservoir, the risk of exposure spikes exponentially. We’re urging ports to integrate hantavirus screening into their routine health protocols.”
The lack of point-of-care diagnostics (rapid tests) exacerbates the problem. Current PCR testing requires lab infrastructure, delaying results by 24–48 hours. Research into serological assays (blood tests for antibodies) is ongoing, but none are FDA- or EMA-approved for clinical use. The National Institutes of Health (NIH) has funded Phase I trials for a Puumala-specific vaccine, but it remains years from approval.
Why No Treatment Exists—and What’s in the Pipeline
Hantavirus infections are treated supportively—meaning doctors manage symptoms, not the virus itself. Ribavirin, an antiviral used for Lassa fever, has shown marginal efficacy in lab studies but no Phase III trial has proven its benefit in humans. The mechanism of action (how it works) involves inhibiting viral RNA polymerase, but its narrow therapeutic window (must be given within 7 days of symptoms) limits use.
Researchers at the University of Helsinki are testing monoclonal antibodies (lab-made proteins that neutralize the virus) in animal models. A 2024 study in The Lancet Infectious Diseases reported that a cocktail targeting the Glycoprotein precursor (GPC) of the Dobrava-Belgrade virus reduced mortality by 40% in ferrets. However, human trials are not expected before 2028.
Prof. Jukka Takkinen, Lead Investigator, University of Helsinki: “The challenge is dual: we need antibodies that work across multiple hantavirus strains, and we must ensure they don’t trigger antibody-dependent enhancement (ADE)—where the immune system overreacts, worsening disease. Our current candidates show promise in non-human primates, but scaling to humans is a massive hurdle.”
Funding & Transparency: Who’s Paying for Hantavirus Research?
The European Union’s Horizon Europe program has allocated €12 million to the HANT-VAC consortium, a multi-country effort to develop a pan-hantavirus vaccine. The NIH’s National Institute of Allergy and Infectious Diseases (NIAID) has funded $8.7 million in grants for diagnostic tools, while the Wellcome Trust (UK) supports mechanistic studies on viral entry pathways. Notably, no pharmaceutical company has prioritized hantavirus R&D due to the low perceived market potential—a classic example of a neglected tropical disease with global impact.
Transmission Vectors: How to Avoid Hantavirus on Cruise Ships (and Beyond)
Hantaviruses are not airborne like COVID-19, but they spread via aerosolization—when rodent urine or feces become airborne and are inhaled. Cruise ships are high-risk due to:
- Rodent entry points: Gaps in cargo holds, plumbing vents, and dockside debris.
- Poor ventilation: Enclosed spaces trap aerosols near the floor (where rodents nest).
- Delayed symptom onset: The 9–21 day incubation period means exposure can occur days before diagnosis.
Prevention strategies include:
- Rodent-proofing: Sealing cracks, using electronic monitoring (e.g., Victor traps with sensors).
- Disinfection protocols: Bleach or UV-C light (which denatures viral RNA) for contaminated areas.
- Passenger education: Avoiding dusty areas (e.g., storage rooms) and using HEPA filters in cabins.
The International Maritime Organization (IMO) is drafting guidelines for cruise lines, but compliance is voluntary. The Centers for Disease Control and Prevention (CDC) recommends travelers to hantavirus-endemic regions carry N95 masks (though these are not proven effective for hantavirus prevention) and avoid camping or rural lodging with poor sanitation.
Contraindications & When to Consult a Doctor
While hantavirus is rare, certain groups should seek immediate medical attention if they develop symptoms after potential exposure:
- Immunocompromised individuals: Those on chemotherapy, with HIV/AIDS, or taking immunosuppressants have a higher risk of severe disease.
- Pregnant women: HFRS can lead to pre-eclampsia or miscarriage due to cytokine storms (overactive immune responses).
- Travelers with pre-existing kidney disease: Hantaviruses attack the proximal tubules of the kidneys, worsening chronic conditions.
Seek emergency care if you experience:
- High fever (>38.5°C/101.3°F) + severe muscle aches within 3 weeks of returning from a high-risk area.
- Sudden onset of hemorrhagic symptoms (e.g., nosebleeds, bruising, blood in urine).
- Shortness of breath or pulmonary edema (fluid in the lungs), which can occur in Hantavirus Pulmonary Syndrome (HPS) variants.
Do NOT:
- Self-medicate with NSAIDs (e.g., ibuprofen), which can mask fever and worsen kidney damage.
- Assume symptoms are “just the flu”—delayed diagnosis increases mortality risk.
The Road Ahead: Could This Be the Next Pandemic?
Probability: Low to moderate, but the risk is not zero. Hantaviruses lack the human-to-human transmission efficiency of SARS-CoV-2, but cruise ships, airports, and global travel networks create super-spreader events. The true pandemic potential hinges on three factors:
- Viral adaptation: If a hantavirus mutates to increase cell-surface receptor binding (e.g., ACE2, like COVID-19), transmission could shift.
- Diagnostic gaps: Without rapid tests, outbreaks may go undetected until they’re widespread.
- Climate change: Warming temperatures expand rodent habitats, increasing human exposure.
The WHO’s Global Outbreak Alert and Response Network (GOARN) is monitoring the situation, but public health officials emphasize preparedness over panic. “This is a containment drill,” said a CDC spokesperson. “The tools exist to stop it—we just need to deploy them faster.”
The MV Hondius outbreak serves as a stress test for global health infrastructure. While a second pandemic remains unlikely, the incident highlights the fragility of our response systems when faced with neglected pathogens. The silver lining? This crisis may finally catalyze the investment needed to develop vaccines and treatments—before the next outbreak.
References
- The Lancet Infectious Diseases (2024): “Monoclonal Antibodies for Hantavirus Hemorrhagic Fever: A Preclinical Study”
- CDC Hantavirus Pulmonary Syndrome Surveillance Report (2023)
- WHO Regional Office for Europe: Hantavirus Fact Sheet
- NIH Study: “Ribavirin Efficacy in Hantavirus Infections: A Meta-Analysis”
- ECDC Hantavirus Surveillance Report (2022)
Disclaimer: This article is for informational purposes only and not a substitute for professional medical advice. Always consult a healthcare provider for diagnosis or treatment.