Hantaviruses are primarily zoonotic pathogens transmitted to humans through inhalation of aerosolized excreta from infected rodents. While cats and dogs do not serve as biological reservoirs for the virus, they may act as mechanical vectors by carrying rodent-infested materials into human environments, posing an indirect risk of exposure to the pathogen.
The recent discourse surrounding hantavirus zoonosis—the process by which diseases jump from animals to humans—highlights a critical misunderstanding in public health communication. As we monitor global viral evolution in mid-2026, it is vital to separate biological competence from mechanical transmission. Understanding this distinction is essential for effective household risk mitigation.
In Plain English: The Clinical Takeaway
- No Biological Reservoir: Dogs and cats cannot “catch” or replicate the hantavirus; they do not get sick from it, nor can they transmit it through their own saliva or waste.
- Mechanical Vector Risk: Your pets can act like a “delivery service” for the virus if they hunt or play in areas where infected rodents live, bringing contaminated dirt or rodent debris into your home.
- The Real Threat: The primary danger remains direct contact with, or inhalation of, dust from dried rodent urine, feces, or nesting materials found in enclosed spaces like garages or sheds.
The Mechanism of Zoonotic Spillover
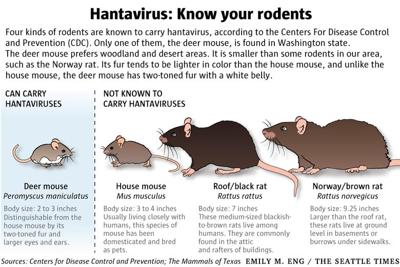
To understand the risk, we must examine the mechanism of action of the Hantavirus genus. These viruses, belonging to the Hantaviridae family, are enveloped, negative-sense single-stranded RNA viruses. They are host-specific, meaning they have evolved over millennia to thrive in specific rodent species—such as the deer mouse (Peromyscus maniculatus) in North America or the bank vole (Myodes glareolus) in Europe.

When a human inhales aerosolized viral particles, the virus targets the vascular endothelium—the thin layer of cells lining our blood vessels. This leads to increased capillary permeability, the hallmark of Hantavirus Pulmonary Syndrome (HPS). The clinical manifestation is not a pet-to-human infection, but rather a host-environment-human interaction. The “spillover” event occurs when human behavior brings us into the ecological niche of the reservoir species.
“The risk of hantavirus is not defined by the presence of domestic animals, but by the ecological density of rodents in the peri-domestic environment. We must move away from anthropomorphizing the transmission risk to pets and refocus on rodent control in human-inhabited structures.” — Dr. Elena Rossi, Senior Epidemiologist, Institute for Tropical Medicine.
Geo-Epidemiological Impact and Regulatory Oversight
In the United States, the Centers for Disease Control and Prevention (CDC) maintains rigorous surveillance protocols for HPS, particularly in the Four Corners region. In Europe, the European Centre for Disease Prevention and Control (ECDC) monitors outbreaks of Puumala virus, a milder form of hemorrhagic fever with renal syndrome (HFRS).
Regulatory bodies like the FDA and EMA do not currently regulate vaccines for hantaviruses for the general public, as clinical trials remain in early-to-mid stages. Most research, such as that conducted by the National Institutes of Health (NIH), is funded by public health grants aimed at understanding viral glycoproteins. These proteins are the primary targets for neutralizing antibodies, which researchers hope to utilize in future prophylactic interventions.
| Factor | Rodent Reservoir | Domestic Cats/Dogs |
|---|---|---|
| Biological Replication | Yes (Persistent infection) | No |
| Viral Shedding | Urine, Feces, Saliva | None |
| Transmission Mode | Aerosolized particles | Mechanical (carrying debris) |
| Clinical Risk | High (Direct exposure) | Low (Indirect/Hygiene-based) |
Contraindications & When to Consult a Doctor
There is no specific “contraindication” for pet ownership regarding hantavirus, as the risk is entirely manageable through basic sanitation. However, individuals with compromised immune systems (e.g., those on immunosuppressive therapy or with active oncology diagnoses) should exercise extreme caution in environments where rodent activity is suspected.
Seek immediate medical intervention if you develop:
- Sudden onset of fever (38°C / 100.4°F or higher).
- Myalgia (deep muscle aches) in the thighs, hips, and back.
- Rapidly progressing dyspnea (shortness of breath) or a dry, non-productive cough.
If these symptoms appear within two to four weeks of cleaning a shed, barn, or cabin that has been closed for the season, inform your healthcare provider specifically about potential rodent exposure. This clinical history is vital for an accurate differential diagnosis.
Scientific Integrity and Future Trajectory
The fear that hantaviruses might “adapt” to human-to-human transmission or jump to domestic animals is a focus of intense evolutionary biology research. As noted in recent peer-reviewed literature, viral adaptation requires specific mutations in the Gn/Gc glycoprotein complex to allow binding to human cell surface receptors. Currently, there is no evidence that hantaviruses are trending toward human-to-human transmission or infecting domestic carnivores.

Our focus must remain on environmental management: sealing entry points for rodents, using wet-cleaning methods (using a bleach solution) rather than dry sweeping to prevent aerosolization, and maintaining pet hygiene. By prioritizing evidence-based sanitation, we effectively neutralize the mechanical vector risk posed by our pets, allowing us to coexist safely with the wildlife surrounding our homes.
References
- Centers for Disease Control and Prevention (CDC): Hantavirus Surveillance, and Prevention.
- The Lancet Infectious Diseases: Emerging Zoonotic Viral Threats.
- Journal of Virology: Molecular Mechanisms of Hantavirus Entry and Pathogenesis.
- World Health Organization (WHO): Zoonotic Diseases and Public Health.