Approximately 500,000 people in the Netherlands live with heart failure, yet many remain undiagnosed due to atypical symptoms such as fatigue, unexplained weight gain, or mild shortness of breath that are often dismissed as signs of aging. Recognizing these subtle indicators early can significantly improve outcomes and save lives, according to recent public health initiatives highlighted by Dutch medical centers. Early detection allows timely intervention with guideline-directed medical therapy, reducing hospitalizations and mortality.
Understanding Heart Failure: Beyond the Classic Symptoms
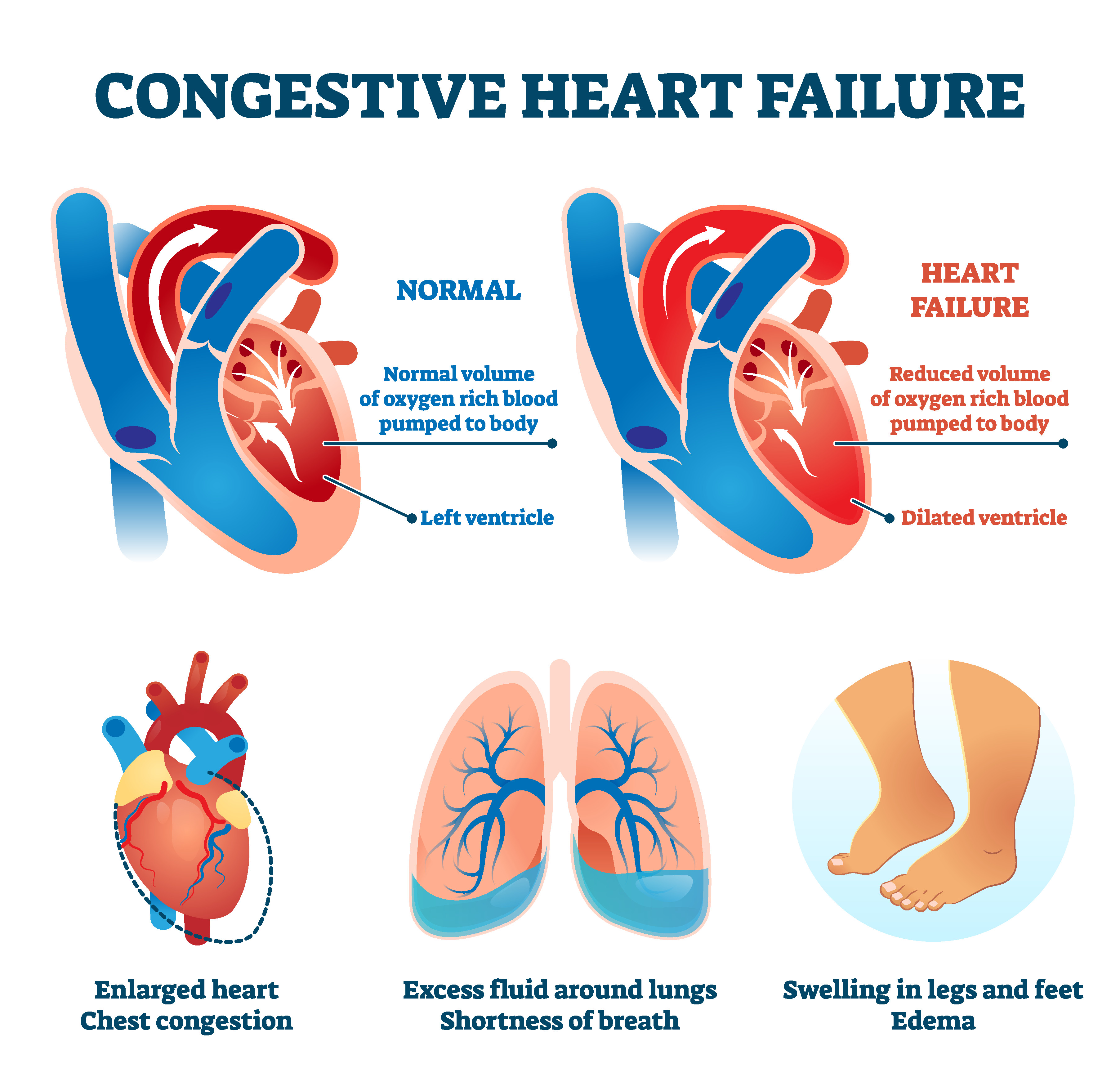
Heart failure (HF) is a clinical syndrome where the heart cannot pump sufficient blood to meet the body’s needs, resulting from structural or functional impairment of ventricular filling or ejection. It is not a single disease but the end-stage manifestation of various cardiovascular conditions, including coronary artery disease, hypertension, and valvular heart disease. Although classic symptoms like dyspnea on exertion and orthopnea are well-known, atypical presentations—particularly in older adults, women, and those with comorbidities like diabetes—often include persistent fatigue, anorexia, cognitive changes, or unexplained edema. These non-specific signs contribute to diagnostic delays, with studies indicating that up to 40% of HF cases are initially missed in primary care settings.
In Plain English: The Clinical Takeaway
- Heart failure affects half a million people in the Netherlands, but many don’t realize they have it because symptoms like tiredness or swollen ankles can seem minor.
- Spotting these subtle signs early—such as sudden weight gain from fluid retention or needing more pillows to breathe at night—can lead to faster treatment and better survival.
- Effective medications exist that reduce hospital visits and improve quality of life, but they operate best when started before severe damage occurs.
Epidemiology and Geographic Context: A Silent Burden in Northwestern Europe
In the Netherlands, the prevalence of heart failure is estimated at 2.9% of the adult population, rising to over 10% in those aged 70 and older, according to data from the Dutch Heart Failure Society (Nederlandse Vereniging voor Cardiologie, NVVC). This aligns with broader European trends, where the European Society of Cardiology (ESC) reports HF affects approximately 15 million people across the EU, with hospitalization rates increasing by 23% over the past decade due to aging populations and improved survival after myocardial infarction. Unlike infectious outbreaks, HF is a non-communicable, progressive syndrome driven by demographic shifts and uncontrolled risk factors such as hypertension (affecting ~30% of Dutch adults) and obesity (prevalence of 16.8% in the Netherlands, per CBS 2024).
Regionally, the Netherlands’ healthcare system, governed by the Zorgverzekeringswet (Health Insurance Act), ensures universal access to primary and specialist care, yet fragmentation between general practitioners and cardiology clinics can delay diagnosis. Initiatives like the nationwide “Hartfalen Week” (Heart Failure Week), supported by academic hospitals including LUMC and Amphia Ziekenhuis Breda, aim to bridge this gap through public education and proactive screening in high-risk groups—particularly those with type 2 diabetes or chronic kidney disease, where HF prevalence exceeds 20%.
Clinical Evidence: Mechanisms, Trials, and Real-World Impact
Guideline-directed medical therapy for heart failure with reduced ejection fraction (HFrEF) has transformed prognosis over the past two decades. Foundational therapies include angiotensin-converting enzyme inhibitors (ACEis), beta-blockers, mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter-2 inhibitors (SGLT2is), which collectively reduce cardiovascular death and HF hospitalization by up to 70% in pivotal trials. The DAPA-HF trial (NCT03036124), funded by AstraZeneca and published in The New England Journal of Medicine in 2019, demonstrated that dapagliflozin (an SGLT2i) reduced the composite endpoint of worsening HF or cardiovascular death by 26% in patients with HFrEF, regardless of diabetes status. Similarly, the EMPEROR-Reduced trial (NCT03057951), supported by Boehringer Ingelheim and Eli Lilly, showed empagliflozin lowered HF hospitalizations by 30% in a similar population.
These drugs work through complementary mechanisms: ACEis and MRAs counteract harmful neurohormonal activation (e.g., angiotensin II and aldosterone), beta-blockers reduce myocardial oxygen demand and arrhythmia risk, while SGLT2is improve cardiac efficiency by modulating myocardial metabolism and reducing interstitial fibrosis. Their combined use represents a paradigm shift from symptom control to disease modification.
“SGLT2 inhibitors have redefined heart failure treatment—not just as glucose-lowering agents, but as cardioprotective drugs that improve outcomes across the spectrum of ejection fraction. Their benefit in reducing fibrosis and inflammation is now well-established in human trials.”
— Dr. Adriana Vergara, Lead Cardiologist, Department of Cardiovascular Medicine, Leiden University Medical Center (LUMC), April 2025.
GEO-Bridging: Regulatory Frameworks and Access in Europe
In the European Union, the European Medicines Agency (EMA) oversees the authorization of HF therapies. Dapagliflozin and empagliflozin received EMA approval for HFrEF in 2021 based on Phase III trial data, with subsequent extensions to heart failure with preserved ejection fraction (HFpEF) following the DELIVER and EMPEROR-Preserved trials. In the Netherlands, these medications are reimbursed under the basic insurance package (basispakket) when prescribed according to NVVC/ESC guidelines, ensuring broad accessibility. However, real-world adherence remains suboptimal; a 2023 study in Netherlands Heart Journal found that only 58% of eligible HFrEF patients received triple therapy (ACEi/ARB, beta-blocker, MRA) within one year of diagnosis, highlighting gaps in implementation despite guideline availability.
Comparatively, the NHS in the UK has implemented structured HF management programs in primary care, achieving higher uptake of guideline-directed therapy. The U.S. FDA has as well approved SGLT2is for HFpEF (2021–2023), reinforcing their role as foundational therapy across HF phenotypes—a stance echoed by the 2023 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure.
Contraindications & When to Consult a Doctor
While guideline-directed therapies are generally safe, certain conditions warrant caution. ACEis and ARBs are contraindicated in pregnancy due to teratogenic risk and in patients with bilateral renal artery stenosis or history of angioedema. MRAs require monitoring of serum potassium and creatinine, particularly in those with baseline eGFR <30 mL/min/1.73m² or serum K⁺ >5.0 mmol/L. SGLT2is carry a small risk of genital mycotic infections and, rarely, euglycemic diabetic ketoacidosis—prompting discontinuation if nausea, vomiting, or unexplained fatigue occurs. Patients should seek immediate medical attention for acute dyspnea at rest, chest pain, syncope, or rapid weight gain (>2 kg in 3 days), which may indicate decompensated HF requiring urgent intervention.
Routine monitoring includes biannual echocardiograms for stable patients, NT-proBNP testing every 6–12 months, and regular assessment of functional status (e.g., 6-minute walk test or Kansas City Cardiomyopathy Questionnaire). Patients with comorbid diabetes or CKD should be prioritized for HF screening, as early intervention in these groups yields the greatest mortality benefit.
The Path Forward: Prevention, Equity, and Integrated Care
Preventing heart failure begins with aggressive management of modifiable risk factors: achieving systolic BP <130 mmHg, maintaining LDL-C <55 mg/dL in high-risk individuals, promoting physical activity (≥150 min/week moderate intensity), and ensuring vaccination against influenza and pneumococcus to reduce infection-induced decompensation. Public health campaigns must emphasize that HF is not an inevitable part of aging but a treatable syndrome when detected early.
Efforts to reduce disparities are critical. Data from the LUMC show that HF prevalence is 1.8 times higher in lower socioeconomic neighborhoods in Rotterdam, linked to higher rates of uncontrolled hypertension and delayed care access. Targeted outreach—such as mobile echocardiography units in underserved areas and multilingual educational materials—can improve equity. Integrating HF screening into routine diabetes and CKD visits, as piloted by Amphia Ziekenhuis Breda, increases detection rates by up to 35%.
Ongoing research focuses on novel therapies like omecamtiv mecarbil (a cardiac myosin activator) and gene-based approaches for familial cardiomyopathies. The GALACTIC-HF trial (NCT02929329), funded by Cytokinetics and Amgen, showed a modest 8% reduction in HF events with omecamtiv mecarbil, though its regulatory path remains complex due to marginal benefit and safety monitoring needs.
References
- Packer M, et al. Effect of dapagliflozin on worsening heart failure or cardiovascular death in patients with heart failure and reduced ejection fraction. N Engl J Med. 2020;383:1476-1487. DAPA-HF Trial.
- McMurray JJV, et al. Empagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2020;383:141-151. EMPEROR-Reduced Trial.
- Solomon SD, et al. Dapagliflozin in heart failure with mildly reduced or preserved ejection fraction. N Engl J Med. 2022;386:1889-1901. DELIVER Trial.
- Anker SD, et al. Empagliflozin in heart failure with preserved ejection fraction. N Engl J Med. 2021;385:1451-1461. EMPEROR-Preserved Trial.
- van Deursen VM, et al. Real-world adherence to guideline-directed medical therapy in heart failure patients in the Netherlands. Neth Heart J. 2023;31:212-221.

