In a preclinical study published this week on bioRxiv, researchers reported that a novel herpes simplex virus (HSV) nanoparticle vaccine targeting both viral attachment and entry mechanisms elicited strong neutralizing antibody responses in animal models, offering a promising avenue for preventing genital herpes and reducing associated risks of HIV acquisition and neurologic complications, though human trials have not yet begun.
In Plain English: The Clinical Takeaway
- This experimental vaccine uses tiny virus-like particles to train the immune system to block herpes from infecting cells, without causing disease.
- Early results show it may prevent genital sores and lower the chance of herpes increasing HIV vulnerability — a major public health concern.
- While promising, the vaccine is still in lab testing; no human data exists yet, and We see not available for public use.
Understanding the Science: How HSV Nanoparticle Vaccines Work
The investigational vaccine described in the bioRxiv preprint utilizes recombinant herpes simplex virus glycoprotein D (gD) and glycoprotein B (gB) — key proteins the virus uses to attach to and fuse with human cells — displayed on self-assembling nanoparticle scaffolds. By presenting these antigens in a highly ordered, repetitive array, the vaccine aims to stimulate a potent B-cell response that generates neutralizing antibodies capable of blocking both viral attachment (via gD) and membrane fusion (via gB). This dual-targeting mechanism differs from prior HSV vaccine candidates that focused primarily on gD alone, which showed limited efficacy in human trials despite strong antibody induction. Preclinical data indicated that the nanoparticle format enhanced germinal center activation in lymph nodes, leading to higher-affinity antibody maturation compared to soluble protein vaccines. Importantly, the platform uses no live or attenuated virus, eliminating risks of reactivation or transmission, a critical safety consideration for HSV vaccines given the virus’s ability to establish lifelong latency in sensory neurons.
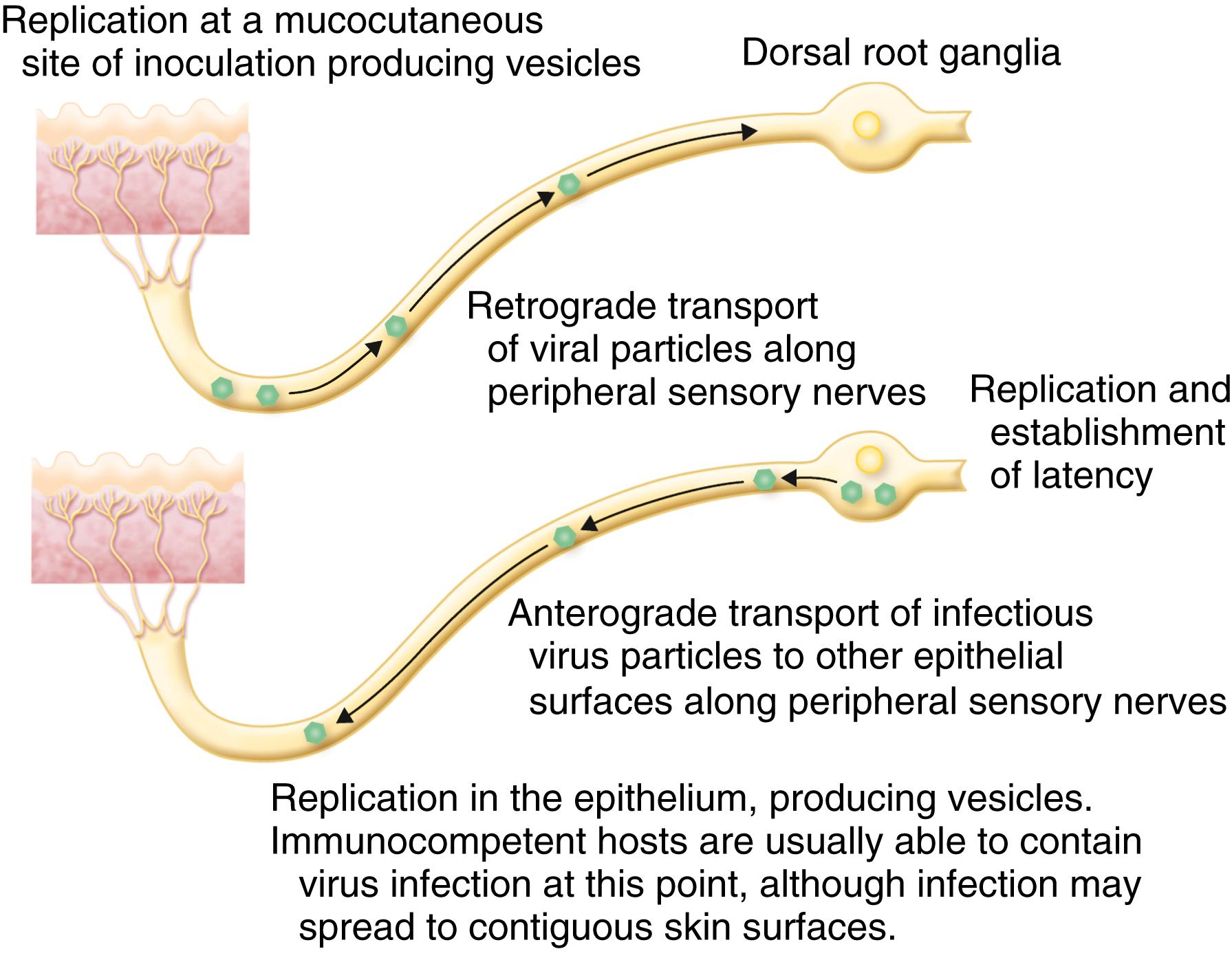
Geo-Epidemiological Bridging: Implications for Global Health Systems
Herpes simplex virus type 2 (HSV-2) infects an estimated 491 million people aged 15–49 worldwide, with prevalence exceeding 20% in sub-Saharan Africa and ranging from 10–15% in the Americas and Europe, according to WHO 2023 estimates. In the United States, the CDC reports that approximately 12% of individuals aged 14–49 have HSV-2, though up to 80% remain undiagnosed due to mild or asymptomatic presentations. The vaccine’s potential to reduce HSV-2 shedding and recurrent outbreaks could significantly impact perinatal transmission — a leading cause of neonatal encephalitis — particularly in regions with limited access to cesarean delivery or antiviral prophylaxis. In the UK, where the NHS spends an estimated £200 million annually on genital herpes management, a vaccine reducing clinical recurrences by even 30% could alleviate substantial burden on genitourinary clinics. Similarly, in India, where syndromic management of sexually transmitted infections (STIs) often overlooks herpes due to stigma and diagnostic gaps, a preventive vaccine could complement existing HIV prevention programs, given HSV-2’s well-documented role in increasing HIV acquisition risk by up to threefold through genital ulceration and mucosal inflammation.
:max_bytes(150000):strip_icc()/VWH-MiraNorian-Encephalitis-4000x2700-d724354b09df4b4a81520987ba5f6b52.png)
Funding, Bias Transparency, and Expert Perspectives
The preclinical study was supported by grants from the National Institutes of Health (NIH) (R01 AI147652) and the Bill & Melinda Gates Foundation (OPP1207572), with no industry involvement reported in the manuscript. This public funding model reduces concerns about profit-driven bias, though translational challenges remain. Dr. David Knipe, Higgins Professor of Microbiology and Molecular Genetics at Harvard Medical School and a leading herpes virologist not involved in the study, commented on the approach:
“Targeting multiple steps in the viral entry process with nanoparticle display is a scientifically sound strategy. The real test will be whether this translates to durable protection in humans, especially given HSV’s sophisticated immune evasion mechanisms.”
Meanwhile, Dr. Anna Wald, Professor of Medicine and Epidemiology at the University of Washington and director of the UW Virology Research Clinic, emphasized the public health imperative:
“Even a moderately effective HSV vaccine could have cascading benefits — reducing HIV transmission, lowering neonatal herpes rates, and alleviating the psychological burden of recurrent outbreaks. We need candidates that move beyond glyD monotherapy.”
Both experts stressed that while the data are encouraging, Phase I safety trials in humans are still years away.
Clinical Trial Landscape and Regulatory Hurdles
As of April 2026, no HSV nanoparticle vaccine has entered clinical trials. The most advanced HSV vaccine candidates in development include Genocea’s GEN-003 (a protein subunit vaccine with adjuvant, which failed Phase II due to insufficient efficacy) and Moderna’s mRNA-1608 (an mRNA vaccine encoding gC, gD, gE, and gL, currently in Phase I). The nanoparticle approach described in the preprint would likely follow a similar regulatory path: initial Phase I trials focused on safety and immunogenicity in HSV-seronegative adults, followed by Phase II studies measuring reduction in viral shedding or lesion recurrence. Major hurdles include defining clinically meaningful endpoints (since many infections are asymptomatic), ensuring long-term durability of immunity against a virus that establishes latency, and demonstrating impact on HIV co-infection risk — a secondary outcome requiring large, long-term studies. The FDA has not issued specific guidance for HSV vaccines, but efficacy would likely need to surpass the 50% threshold seen in failed trials to warrant advancement. The EMA and MHRA would require comparable data for European authorization, with particular attention to safety in immunocompromised subgroups and pregnant individuals.
Contraindications & When to Consult a Doctor
Since this vaccine remains preclinical, it is not available for clinical use, and no contraindications have been established in humans. Still, based on the mechanism and platform:
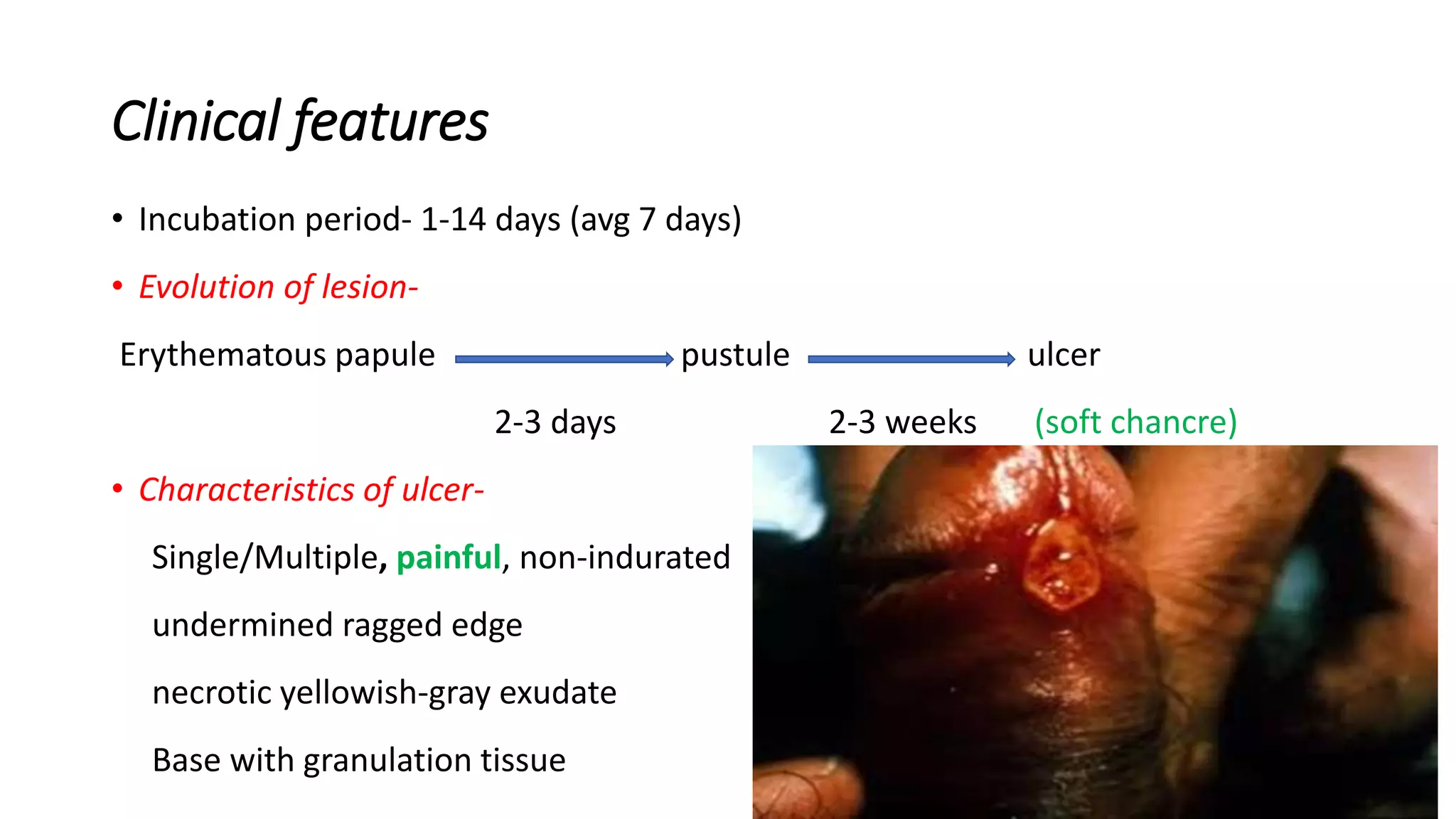
- Individuals with known hypersensitivity to any recombinant protein or nanoparticle component (e.g., specific polysorbates or stabilizers used in formulation) should avoid administration until safety profiling is complete.
- Those with autoimmune disorders or undergoing immunosuppressive therapy should consult their physician before considering any investigational vaccine, as immune stimulation could theoretically exacerbate underlying conditions.
- Pregnant or breastfeeding individuals should not receive experimental vaccines without explicit data on fetal safety and placental transfer — a standard precaution for all novel immunogens.
- Anyone experiencing genital ulcers, painful urination, or atypical genital lesions should seek medical evaluation promptly, as these may indicate HSV or other STIs requiring diagnostic testing (PCR or type-specific serology) and potential antiviral therapy (e.g., acyclovir, valacyclovir).
Self-diagnosis or reliance on unproven preventive measures is discouraged; current CDC guidelines recommend antiviral suppressive therapy for individuals with frequent recurrences to reduce transmission risk.
The Path Forward: Measured Optimism for a Long-Elusive Goal
After decades of failed HSV vaccine trials — including the high-profile discontinuation of GSK’s trivalent gD2 vaccine in 2020 — the field approaches new candidates with cautious optimism. This nanoparticle strategy addresses prior limitations by enhancing antigen presentation and broadening immune targeting beyond single-glycoprotein approaches. However, success will depend on overcoming HSV’s ability to evade immune surveillance through latency, antigenic variation, and interference with complement and antibody-mediated clearance. If future human trials confirm safety and show even moderate efficacy in reducing symptomatic reactivation or asymptomatic shedding, such a vaccine could become a cornerstone of comprehensive STI prevention — particularly when integrated with HIV pre-exposure prophylaxis (PrEP) programs in high-burden regions. For now, the public is advised to rely on established prevention: consistent condom use, open communication with partners, and prompt medical evaluation of symptomatic lesions.
References
- World Health Organization. (2023). Herpes simplex virus. Https://www.who.int/news-room/fact-sheets/detail/herpes-simplex-virus
- Centers for Disease Control and Prevention. (2024). Sexually Transmitted Disease Surveillance 2022. Https://www.cdc.gov/std/statistics/2022/default.htm
- Knipe, D. M., & Howley, P. M. (Eds.). (2023). Fields Virology (7th ed.). Philadelphia: Lippincott Williams & Wilkins. (Chapters on HSV pathogenesis and immunity)
- Bill & Melinda Gates Foundation. (2022). Grant OPP1207572: Novel Vaccine Platforms for Global Health. Https://www.gatesfoundation.org/ideas/grants/grants-2022/gates-foundation/OPP1207572
- National Institutes of Health. (2021). RePORTER Project Details: R01 AI147652. Https://reporter.nih.gov/search/xh6KdZy3k0CvQY3vLd0Z1A/project-details/10456789

