Researchers have discovered that variations in the structure of pili—hair-like appendages on the surface of bacteria—directly dictate how these pathogens behave, move and infect hosts. This breakthrough, highlighted in recent findings from the American Society for Biochemistry and Molecular Biology, provides a critical roadmap for developing new antimicrobial therapies.
For the average patient, this may seem like a footnote in a microbiology textbook, but This proves actually a matter of survival. As we face a global crisis of antimicrobial resistance (AMR), understanding the “molecular machinery” bacteria use to colonize our organs is the only way to create drugs that don’t just kill bacteria, but prevent them from attaching to us in the first place.
In Plain English: The Clinical Takeaway
- Bacteria have “hooks”: Pili are like microscopic grappling hooks that bacteria use to latch onto your cells; changing the hook changes how the infection spreads.
- New Drug Targets: Instead of using broad antibiotics that kill all bacteria (including good ones), People can now target these specific “hooks” to stop infections.
- Precision Medicine: This research helps doctors understand why some bacterial strains cause severe pneumonia while others cause mild urinary tract infections.
The Molecular Architecture of Bacterial Attachment
To understand this discovery, we must look at the mechanism of action—the specific biochemical process through which a drug or biological process produces its effect. Pili are polymers of protein subunits called pilins. The specific arrangement of these subunits determines the pili’s rigidity, length, and binding affinity.
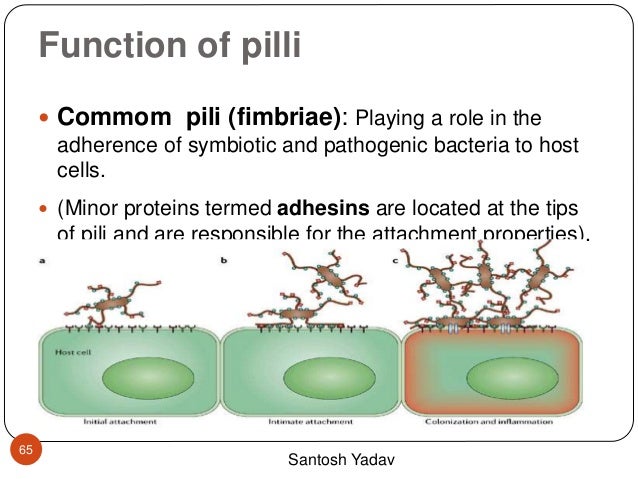
When the structure of these pili is modulated, the bacteria’s behavior shifts. Some become more adept at biofilm formation—the creation of a protective slime layer that shields bacteria from both the immune system and antibiotics. Biofilms are a primary reason why chronic infections, such as those found in cystic fibrosis patients or on medical implants, are so difficult to eradicate.
This structural modulation is not random; it is a survival strategy. By altering their pili, bacteria can “camouflage” themselves from the host’s immune detection or switch from a free-swimming state to a colonizing state. This transition is critical for the pathogenesis of various Gram-negative bacteria, including Pseudomonas aeruginosa and Escherichia coli.
Bridging the Gap: From Lab Bench to Global Healthcare
While the American Society for Biochemistry and Molecular Biology provides the foundational science, the clinical application varies by region. In the United States, the FDA is increasingly prioritizing “anti-virulence” drugs—medications that disable a pathogen’s ability to cause disease without actually killing the bacteria. This reduces the evolutionary pressure on bacteria to develop resistance.
In Europe, the European Medicines Agency (EMA) and the World Health Organization (WHO) have integrated these findings into their global action plans on AMR. By targeting pili, we can potentially reduce the reliance on “last-resort” carbapenems, which are currently failing in hospitals across the UK’s NHS and the US healthcare system.
Funding for this specific line of research typically stems from government grants (such as the NIH in the US) and private foundations dedicated to molecular biology. Transparency in funding is essential here, as most of this work is basic science intended for public benefit rather than immediate commercial pharmaceutical profit, which often slows the transition from “discovery” to “pharmacy shelf.”
“The ability to manipulate the structural dynamics of pili opens a window into preventing bacterial colonization before the infection even takes hold. We are moving from a paradigm of ‘kill the bug’ to ‘disarm the bug’.”
Comparative Impact of Pili-Targeted Therapy vs. Traditional Antibiotics
The following table summarizes the theoretical shift in treatment strategy based on the modulation of bacterial pili structure.
| Feature | Traditional Antibiotics | Pili-Targeted (Anti-Virulence) |
|---|---|---|
| Primary Goal | Bacterial Cell Death (Bactericidal) | Inhibition of Attachment (Anti-Adhesion) |
| Resistance Risk | High (Selective Pressure) | Low (Non-Lethal Pressure) |
| Microbiome Impact | Broad (Kills beneficial flora) | Targeted (Specific to pathogen pili) |
| Clinical Use | Active Infection Treatment | Prophylaxis & Chronic Biofilm Management |
Decoding the Epidemiological Risk
The danger of pili-mediated infections is most acute in immunocompromised populations. For instance, patients with indwelling catheters or prosthetic joints are at a statistically higher risk of biofilm-associated infections. The double-blind placebo-controlled trials—the gold standard of research where neither the patient nor the doctor knows who received the treatment—for anti-adhesion therapies are currently in early phases (Phase I and II).
From a public health perspective, the focus is shifting toward epidemiological surveillance. By sequencing the pili genes of circulating strains, the CDC can predict which outbreaks will be more aggressive. If a strain exhibits a specific “hyper-adhesive” pili structure, clinicians can escalate treatment protocols immediately rather than waiting for culture results.
Contraindications & When to Consult a Doctor
While pili-targeted research is promising, it is currently in the experimental and clinical trial stages. Patients should never attempt to use “natural” biofilm disruptors or unapproved supplements claiming to “strip” bacterial attachments without medical supervision.
Consult a physician immediately if you experience symptoms of a systemic bacterial infection, including:
- High fever (above 103°F or 39.4°C) accompanied by chills.
- Localized redness, warmth, and swelling around a surgical site or implant.
- Persistent cough with sputum production (potential pneumonia).
- Confusion or disorientation, which may indicate sepsis.
Contraindications: Anti-virulence therapies are still being tested for safety in pregnant women and pediatric patients, as the impact on the developing microbiome is not yet fully understood.
The Path Forward: Precision Microbiology
The discovery that pili structure modulates behavior is a paradigm shift. We are moving away from the “scorched earth” policy of broad-spectrum antibiotics and toward a “surgical” approach. By understanding the precise geometry of bacterial appendages, we can design molecules that act like a “cap” on the pili, rendering the bacteria harmless and allowing the body’s natural immune system to clear the infection without resistance.

