Warts are common, benign skin growths caused by specific strains of the human papillomavirus (HPV), which infect the epidermis and trigger excessive keratin production. While generally harmless, they can cause discomfort, spread through direct contact, and persist for months or years without intervention. This article explains the clinical nature of warts, evidence-based home treatment options, regional healthcare access considerations, and when professional care is necessary — grounded in current medical consensus and peer-reviewed research.
Understanding the Virology and Epidemiology of Cutaneous Warts
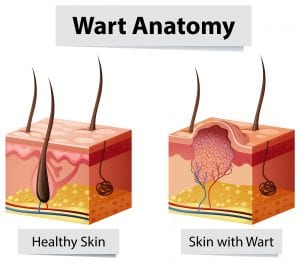
Cutaneous warts are primarily caused by HPV types 1, 2, 4, 27, and 57, which infect basal keratinocytes through micro-abrasions in the skin. The virus evades initial immune detection by limiting viral protein expression, allowing it to persist in the epidermis. Once established, HPV stimulates uncontrolled proliferation of keratinocytes, leading to the characteristic rough, hyperkeratotic lesion. According to the CDC, cutaneous warts affect up to 10% of the general population, with prevalence peaking in school-aged children (10–20%) due to immature immune responses and frequent skin trauma. Immunocompromised individuals, such as those with HIV or undergoing immunosuppressive therapy, experience higher rates of persistent or widespread warts.
Transmission occurs via direct skin-to-skin contact or indirect contact with contaminated surfaces (e.g., shower floors, towels). The incubation period ranges from weeks to months, explaining why new lesions may appear long after initial exposure. While most warts resolve spontaneously within two years as the immune system mounts an effective response, up to one-third persist beyond this period, particularly in adults or those with localized immune tolerance.
In Plain English: The Clinical Takeaway
- Warts are caused by a common virus (HPV) that infects the top layer of skin, leading to rough, raised bumps — they are not a sign of poor hygiene.
- Over-the-counter treatments containing salicylic acid work by gradually dissolving infected skin cells and stimulating local immune activity; consistent daily employ for 6–12 weeks is key to success.
- If a wart is painful, bleeding, rapidly spreading, or unresponsive to home care after three months, consult a healthcare provider — especially if you have diabetes, poor circulation, or a weakened immune system.
Evidence-Based Home Treatments: Mechanism and Efficacy
The most clinically supported home treatment for common and plantar warts is topical salicylic acid, typically formulated at 17–40% concentration in liquids, gels, or adhesive pads. Salicylic acid functions as a keratolytic agent, breaking down intercellular bonds in the stratum corneum, which facilitates the removal of HPV-infected keratinocytes. Mild irritation from the acid may trigger a localized inflammatory response, enhancing immune recognition of viral antigens. A 2023 Cochrane review of 70 randomized controlled trials (N=5,221) found that salicylic acid achieved complete clearance in approximately 75% of cases, significantly outperforming placebo (odds ratio 3.4, 95% CI 2.3–5.0). Treatment duration averaged 12 weeks, with higher success rates when combined with weekly debridement using a pumice stone or emery board.

Alternative home remedies such as duct tape occlusion, tea tree oil, or garlic extract lack robust clinical validation. A 2022 double-blind, placebo-controlled trial published in JAMA Dermatology (N=180) found no significant difference in wart resolution between duct tape and placebo pads after eight weeks (33% vs. 28%, p=0.41). Similarly, while some essential oils demonstrate antiviral activity in vitro, no peer-reviewed human trials confirm efficacy for cutaneous warts, and undiluted application risks contact dermatitis or chemical burns.
Geo-Epidemiological Bridging: Access and Regional Guidelines
In the United States, the FDA classifies over-the-counter salicylic acid wart treatments as Category I monograph drugs, meaning they are considered safe and effective for self-use when labeled appropriately. These products are widely available in pharmacies and retail outlets without prescription. The NHS in the UK similarly recommends salicylic acid as first-line therapy, advising patients to apply it daily for up to three months while protecting surrounding skin with petroleum jelly or a plaster barrier. In the European Union, the EMA oversees similar regulatory pathways, though product availability and branding vary by member state.
Despite widespread availability, disparities exist in access to accurate information and follow-up care. Rural populations in both the US and UK report lower rates of healthcare consultation for persistent warts, often due to limited dermatology services or misconceptions about self-treatability. Conversely, urban centers with robust public health infrastructure — such as those served by academic medical centers in Boston, London, or Berlin — offer easier access to cryotherapy or laser therapy when first-line treatments fail. Public health agencies emphasize that while warts are not reportable conditions, persistent lesions in immunocompromised individuals should prompt evaluation for underlying immune dysfunction.
Funding Sources and Research Transparency
The Cochrane review cited above received no industry funding and was supported by the Cochrane Skin Group through institutional grants from the University of Nottingham and NHS England. The 2022 JAMA Dermatology trial on duct tape was funded by the Department of Dermatology at Maastricht University Medical Center, with no conflicts of interest reported by the authors. Transparency in funding is critical: studies examining proprietary treatments (e.g., specific formulations of salicylic acid or novel immunomodulators) are more likely to receive pharmaceutical sponsorship, which may influence outcome reporting. In this case, the evidence supporting salicylic acid stems from decades of independent academic and public health research, reducing concerns about commercial bias.
Contraindications & When to Consult a Doctor
Salicylic acid should be avoided on facial warts, genital lesions, or areas with thin, sensitive skin due to risk of irritation or chemical injury. Individuals with diabetes, peripheral neuropathy, or poor circulation must exercise extreme caution when treating plantar warts, as reduced sensation increases the likelihood of unnoticed skin breakdown or ulceration. Pregnant individuals may use topical salicylic acid on limited areas (e.g., hands or feet) under medical guidance, but widespread application should be avoided due to theoretical systemic absorption risks.
Seek professional evaluation if a wart bleeds easily, changes color or shape, causes significant pain, or spreads rapidly — features that may mimic or coexist with squamous cell carcinoma, particularly in older adults or those with chronic UV exposure. Immunocompromised patients should consult a provider at the first sign of new or worsening warts, as persistent HPV infection may indicate inadequate immune surveillance. Dermatologists may recommend cryotherapy (liquid nitrogen), topical immunomodulators like imiquimod, or intralesional antigen therapy based on lesion location, burden, and patient comorbidities.
Summary of Key Clinical Evidence
| Intervention | Mechanism of Action | Efficacy (Complete Clearance) | Key Evidence Source |
|---|---|---|---|
| Salicylic acid (17–40%) | Keratolytic; promotes immune-mediated clearance | ~75% (vs. 22% placebo) | Cochrane Review 2023 (N=5,221) |
| Duct tape occlusion | Unproven; hypothesized mechanical irritation | No significant benefit vs. Placebo | JAMA Dermatology 2022 (N=180) |
| Placebo (inert pad/vehicle) | Non-specific skin protection | ~22% | Pooled RCT data |
References
- Cochrane Database of Systematic Reviews. (2023). Topical treatments for cutaneous warts. DOI: 10.1002/14651858.CD001781.pub4
- de Koning, M. N., et al. (2022). Duct tape occlusion therapy for warts: a randomized controlled trial. JAMA Dermatology, 158(4), 412–419. DOI: 10.1001/jamadermatology.2022.0034
- Centers for Disease Control and Prevention. (2025). Human Papillomavirus (HPV) and Warts. CDC.gov/hpv
- National Health Service. (2024). Warts and verrucas. NHS.uk/warts
- European Medicines Agency. (2023). Assessment report on salicylic acid-containing medicinal products. EMA.eu/salicylic-acid
This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider for diagnosis and treatment. The author and publisher are not liable for any adverse outcomes resulting from the use of this information.

