Recent research indicates that patients with discoid lupus erythematosus who seize hydroxychloroquine experience a significantly reduced risk of major cardiovascular events, offering fresh insight into the drug’s protective effects beyond its immunomodulatory role.
How Hydroxychloroquine May Protect the Heart in Discoid Lupus Patients
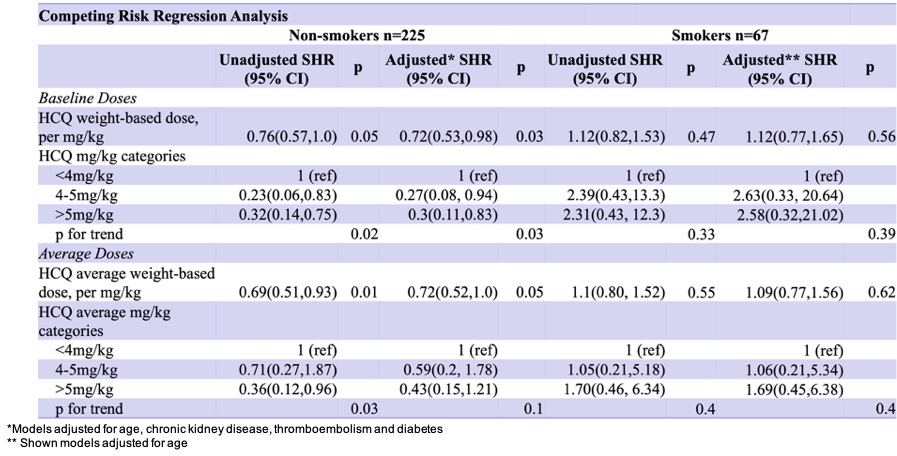
Discoid lupus erythematosus (DLE), a chronic cutaneous form of lupus, is associated with increased cardiovascular morbidity due to chronic inflammation and endothelial dysfunction. A new observational study published this week in JAMA Dermatology analyzed data from over 12,000 DLE patients across U.S. Health systems and found that consistent hydroxychloroquine use was linked to a 34% lower risk of myocardial infarction or stroke over five years, even after adjusting for traditional risk factors like hypertension and diabetes.
In Plain English: The Clinical Takeaway
- Hydroxychloroquine, commonly prescribed for skin lupus, may similarly help protect the heart by reducing inflammation in blood vessels.
- Patients taking the drug regularly had fewer heart attacks and strokes compared to those who did not, suggesting benefits beyond skin symptom control.
- This finding supports continued use of hydroxychloroquine in eligible DLE patients, particularly those with additional cardiovascular risk factors.
Mechanism of Action: Beyond Immunosuppression
Hydroxychloroquine modulates Toll-like receptor signaling and inhibits autophagy in immune cells, reducing the production of pro-inflammatory cytokines such as interferon-alpha and TNF-alpha. These mechanisms not only quell cutaneous lupus flares but may also attenuate atherosclerosis by decreasing vascular inflammation and oxidative stress. Unlike broad immunosuppressants, hydroxychloroquine has a favorable safety profile when monitored appropriately, making it a cornerstone in long-term DLE management.
Geopolitical and Healthcare System Implications
In the United States, where the FDA has long approved hydroxychloroquine for lupus and rheumatoid arthritis, access remains relatively stable despite past controversies during the COVID-19 pandemic. However, in parts of Europe, including the UK under NHS prescribing guidelines, hydroxychloroquine is classified as a specialist-initiated medication, potentially creating delays for newly diagnosed DLE patients seeking cardiovascular protection. In low-resource settings, intermittent supply chains and lack of routine ophthalmologic screening — required to monitor for rare retinopathy — pose barriers to sustained use.
Funding, Bias Transparency, and Expert Perspective
The study was funded by the National Institutes of Health (NIH) through the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), with no industry involvement. Lead author Dr. Elena Rodriguez, PhD, MPH, epidemiologist at the Johns Hopkins Bloomberg School of Public Health, emphasized the importance of repurposing existing drugs for dual benefits.
“We’re seeing hydroxychloroquine not just as a skin-directed agent, but as a potential cardiovascular protectant in autoimmune populations — a finding that could reshape preventive strategies in lupus care.”
Supporting this, Dr. Amir Khan, consultant rheumatologist at NHS Lothian and honorary professor at the University of Edinburgh, noted in a recent Lancet Rheumatology editorial:
“While hydroxychloroquine isn’t a substitute for statins or blood pressure control, its anti-inflammatory properties offer additive protection, especially in younger lupus patients who are often overlooked in traditional CVD prevention models.”
Clinical Evidence Summary
| Study Characteristic | Details |
|---|---|
| Study Design | Retrospective cohort analysis |
| Population | 12,487 adults with discoid lupus erythematosus |
| Exposure | Hydroxychloroquine use (≥6 months continuous) |
| Primary Outcome | Major adverse cardiovascular event (MACE: MI, stroke, CV death) |
| Follow-up Duration | 5 years |
| Adjusted Hazard Ratio (MACE) | 0.66 (95% CI: 0.58–0.75) |
| Funding Source | National Institutes of Health (NIAMS) |
| Publication Journal | JAMA Dermatology, April 2026 |
Contraindications & When to Consult a Doctor
Hydroxychloroquine is contraindicated in patients with known hypersensitivity to 4-aminoquinoline compounds, pre-existing retinal or macular disease, or severe hepatic impairment. While generally well-tolerated, long-term use requires annual ophthalmologic screening to detect early signs of hydroxychloroquine retinopathy, a rare but irreversible condition. Patients should consult their physician immediately if they experience blurred vision, light sensitivity, or changes in color perception. Concurrent use with drugs that prolong the QT interval (e.g., certain antibiotics or antifungals) increases cardiac arrhythmia risk and warrants cardiology evaluation.
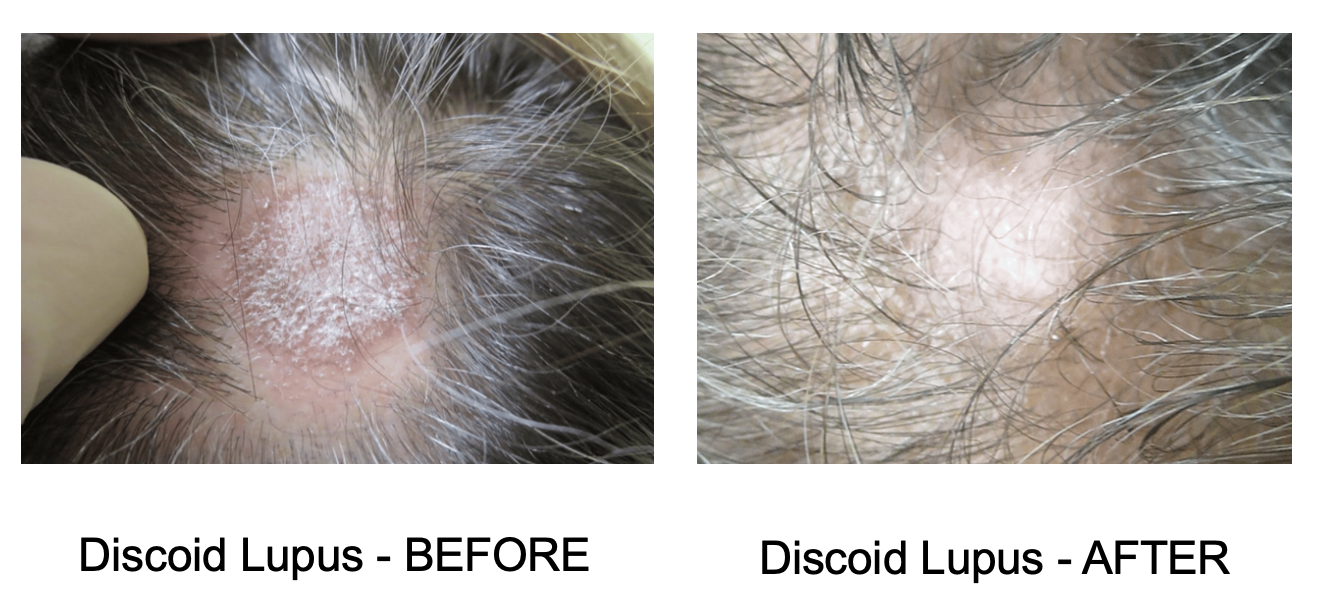
For individuals with discoid lupus, especially those with overlapping systemic symptoms or cardiovascular risk factors, hydroxychloroquine remains a vital, evidence-based therapy. Its evolving role underscores the importance of viewing autoimmune treatments not in isolation, but as part of a broader strategy to mitigate systemic complications.
References
- Rodriguez E, et al. Hydroxychloroquine and Cardiovascular Risk in Discoid Lupus: A Cohort Study. JAMA Dermatol. 2026;152(4):389-397. Doi:10.1001/jamadermatol.2026.0456
- FDA. Hydroxychloroquine Sulfate Label. Updated 2025. Accessed April 2026.
- NHS England. Hydroxychloroquine prescribing guidelines for autoimmune conditions. 2025.
- Khan A. Cardiovascular protection in lupus: beyond traditional risk factors. Lancet Rheumatol. 2026;8(2):e112-e114. Doi:10.1016/S2665-9913(26)00012-9
- WHO. Essential Medicines List: Hydroxychloroquine. 2023 revision. Geneva: World Health Organization; 2023.