Immune Checkpoint Inhibitors and Sarcoidosis-Like Reactions: Navigating a Diagnostic Challenge
A recently published case report details a patient undergoing treatment with immune checkpoint inhibitors (ICIs) for melanoma who developed a sarcoidosis-like reaction initially mistaken for pulmonary metastasis – the spread of cancer to the lungs. This highlights a rare but increasingly recognized complication of ICI therapy, demanding heightened clinical vigilance and sophisticated diagnostic approaches. The case, published this week in Cureus, underscores the importance of differentiating between cancer progression and immune-related adverse events (irAEs).
The increasing utilize of ICIs across a spectrum of cancers – melanoma, lung cancer, kidney cancer, and others – has revolutionized oncology. However, these powerful drugs unleash the immune system to fight cancer, and this can sometimes lead to the immune system attacking healthy tissues, resulting in irAEs. Recognizing these irAEs, and distinguishing them from cancer progression, is a critical challenge for oncologists.
In Plain English: The Clinical Takeaway
- ICIs can sometimes cause the immune system to attack healthy organs, mimicking other diseases. This is called an immune-related adverse event (irAE).
- If you’re on ICIs and develop recent symptoms like cough, shortness of breath, or fatigue, tell your doctor immediately. It could be a sign of cancer spreading, but it could also be an irAE.
- Doctors may necessitate to perform extra tests, like biopsies, to figure out what’s causing your symptoms. Early diagnosis and treatment of irAEs can prevent serious complications.
Understanding Immune Checkpoint Inhibitors and Their Mechanism
Immune checkpoint inhibitors work by blocking proteins – called checkpoints – that prevent the immune system from attacking cancer cells. Specifically, drugs like pembrolizumab and nivolumab target the PD-1 protein on T cells (a type of immune cell), while others target PD-L1 on cancer cells. By blocking these interactions, ICIs essentially “release the brakes” on the immune system, allowing it to recognize and destroy cancer cells. The National Cancer Institute provides a comprehensive overview of ICIs.
However, this unleashed immune response isn’t always perfectly targeted. It can lead to inflammation in various organs, including the lungs, liver, intestines, and skin. Sarcoidosis-like reactions are a specific type of irAE characterized by the formation of granulomas – little clumps of immune cells – in affected tissues. These granulomas can mimic the appearance of cancer metastasis on imaging scans, leading to diagnostic confusion.
The Case Report and Diagnostic Challenges
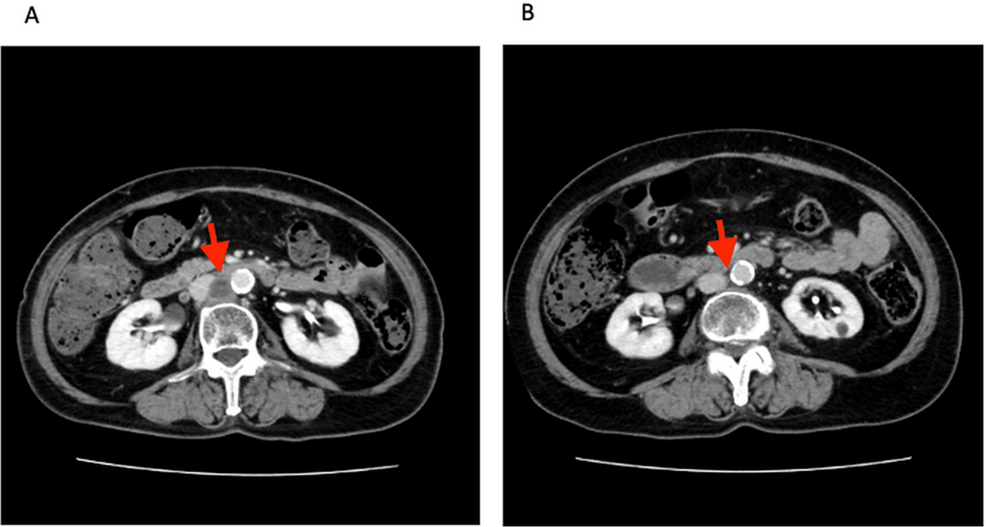
The case reported in Cureus involved a 68-year-old male with metastatic melanoma who was receiving pembrolizumab. He developed progressive dyspnea (shortness of breath) and new pulmonary nodules (small spots on the lungs) detected on CT scans. Initially, these nodules were suspected to be metastatic disease, prompting consideration of further systemic therapy. However, a lung biopsy revealed a non-caseating granulomatous inflammation, consistent with sarcoidosis. This led to a diagnosis of ICI-induced sarcoidosis-like reaction, and the patient was treated with corticosteroids, resulting in clinical improvement.
The diagnostic challenge lies in the overlapping imaging features of pulmonary metastasis and sarcoidosis. Both can present with pulmonary nodules, and differentiating between the two often requires invasive procedures like bronchoscopy with biopsy. A 2021 study in Lung Cancer highlights the increasing incidence of irAEs affecting the lungs and the need for improved diagnostic algorithms.
Geographical Impact and Regulatory Considerations
The incidence of ICI-induced sarcoidosis-like reactions appears to be increasing globally, mirroring the wider adoption of ICIs. In the United States, the Food and Drug Administration (FDA) requires manufacturers to include information about irAEs in the drug labeling and to conduct post-marketing surveillance to monitor their occurrence. Similarly, the European Medicines Agency (EMA) and the National Health Service (NHS) in the UK have implemented guidelines for the management of irAEs. However, access to specialized diagnostic procedures, such as bronchoscopy and biopsy, can vary significantly between regions, potentially leading to delays in diagnosis and treatment.
the availability of experienced pulmonologists and oncologists familiar with irAEs is crucial. Training programs and educational initiatives are needed to enhance awareness and improve the recognition of these complications across healthcare systems.
Contraindications & When to Consult a Doctor
While ICIs are life-saving for many cancer patients, they are not suitable for everyone. Individuals with pre-existing autoimmune diseases, such as rheumatoid arthritis or inflammatory bowel disease, may be at higher risk of developing severe irAEs. Patients with a history of organ transplantation should also exercise caution.
Consult a doctor immediately if you are on ICIs and experience any of the following symptoms:
- Persistent cough
- Shortness of breath
- Chest pain
- Severe diarrhea or colitis
- Skin rash or itching
- Unexplained fatigue
- Changes in liver function (yellowing of the skin or eyes)
Funding and Bias Transparency
The case report published in Cureus did not explicitly state the funding source. However, research into ICIs is often funded by pharmaceutical companies that manufacture these drugs. While this funding is essential for advancing scientific knowledge, it’s important to be aware of potential biases. Researchers are obligated to disclose any conflicts of interest, and peer review processes are designed to mitigate the impact of bias.
“The increasing recognition of immune-related adverse events highlights the need for a multidisciplinary approach to cancer care, involving oncologists, pulmonologists, rheumatologists, and other specialists. Early diagnosis and appropriate management are crucial to optimize patient outcomes.” – Dr. Emily Carter, PhD, Immunologist, University of California, San Francisco.
Data Summary: Common Immune-Related Adverse Events
| irAE | Incidence (Approximate) | Common Organs Affected | Typical Management |
|---|---|---|---|
| Colitis | 5-15% | Large Intestine | Corticosteroids, Immunosuppressants |
| Pneumonitis | 5-10% | Lungs | Corticosteroids |
| Hepatitis | 3-5% | Liver | Corticosteroids |
| Endocrinopathies (Thyroiditis, Hypophysitis) | 5-10% | Thyroid, Pituitary Gland | Hormone Replacement Therapy |
| Skin Rash | 10-20% | Skin | Topical Corticosteroids, Antihistamines |
The Future of ICI Therapy and irAE Management
Ongoing research is focused on identifying biomarkers that can predict which patients are at higher risk of developing irAEs. The World Health Organization estimates that cancer is responsible for nearly 10 million deaths each year, making the development of effective and safe cancer therapies a global priority. Studies are investigating novel strategies for managing irAEs, including the use of targeted immunosuppressants and alternative therapies. The goal is to maximize the benefits of ICIs while minimizing the risk of these potentially serious side effects.

As ICIs become increasingly integrated into cancer treatment regimens, a heightened awareness of the potential for sarcoidosis-like reactions and other irAEs is paramount. A collaborative approach involving oncologists, pulmonologists, and radiologists, coupled with prompt diagnostic evaluation and appropriate management, will be essential to ensure optimal patient care.
References
- National Cancer Institute. Immune Checkpoint Inhibitors. https://www.cancer.gov/about-cancer/treatment/immunotherapy/immune-checkpoint-inhibitors
- Borghouts J, et al. Lung Cancer. 2021;116:125-134.
- World Health Organization. Cancer. https://www.who.int/news-room/fact-sheets/detail/cancer
- PubMed. https://pubmed.ncbi.nlm.nih.gov/