Researchers at the University of California, Riverside have identified specialized immune surveillance structures within human skin hair follicles that actively monitor for pathogens and cellular abnormalities, representing a previously underappreciated first-line defense mechanism in the body’s largest organ. This discovery, published this week in a leading immunology journal, reveals how follicle-associated lymphoid tissue (FALT) coordinates with dendritic cells and T-regulatory subsets to maintain cutaneous immune homeostasis without triggering inflammation. Understanding this system offers recent avenues for treating inflammatory skin disorders, enhancing vaccine delivery via topical routes, and improving early detection of skin cancers through immune-mediated surveillance.
In Plain English: The Clinical Takeaway
- Hair follicles are not just for hair growth—they contain built-in immune watchtowers that constantly scan for threats like bacteria, viruses, and precancerous cells.
- These follicle-based immune structures help prevent unnecessary inflammation while still defending the skin, offering a natural balance that could be harnessed for safer therapies.
- Targeting this system may improve how vaccines or medications are delivered through the skin and could lead to better treatments for eczema, psoriasis, and skin cancer.
Uncovering the Skin’s Hidden Immune Network: Follicle-Associated Lymphoid Tissue
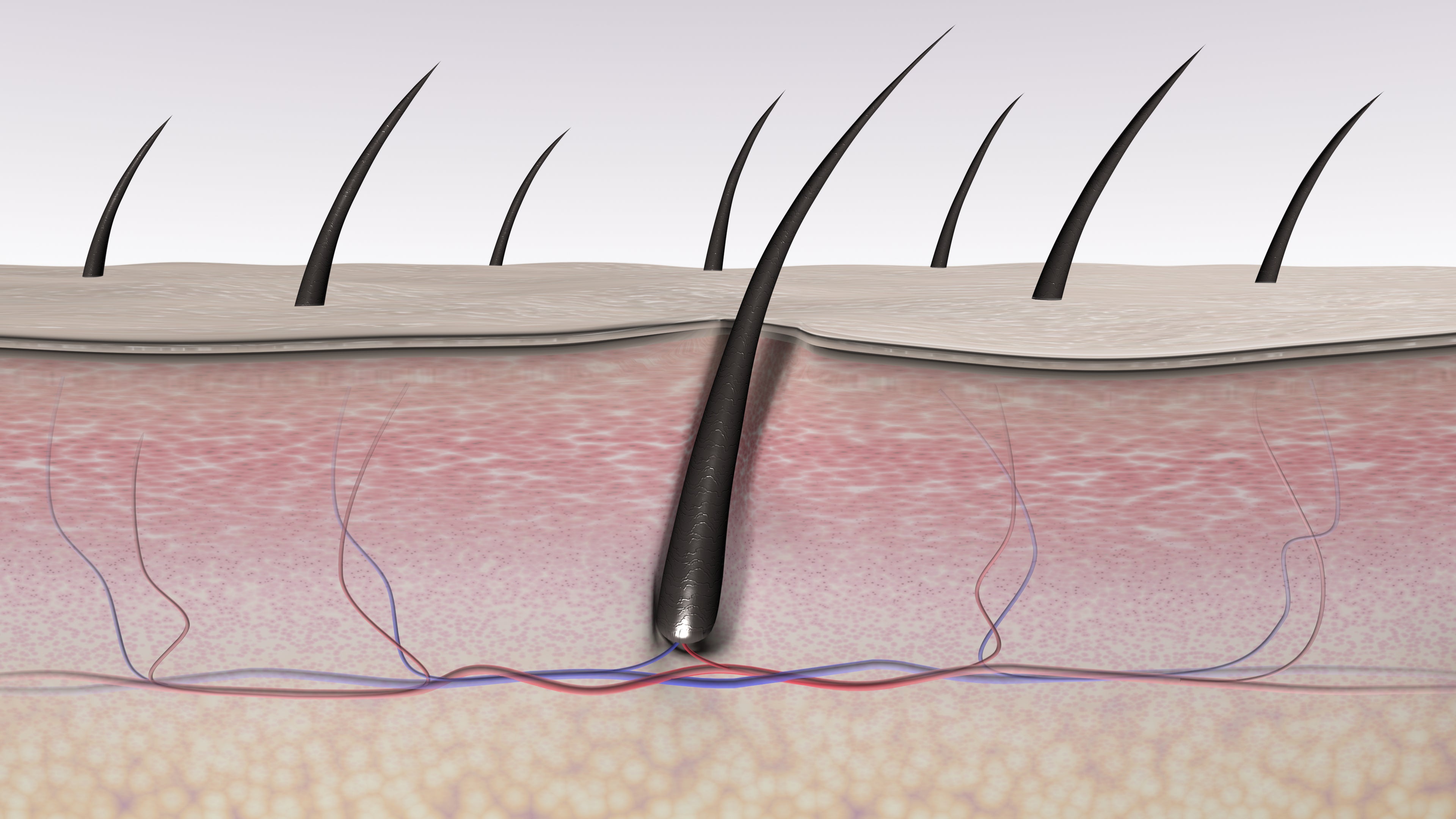
The skin serves as a critical barrier against environmental pathogens, yet its immune mechanisms extend beyond superficial layers. Researchers at UC Riverside, led by Dr. Maria Chen, PhD in Immunobiology, used advanced confocal microscopy and single-cell RNA sequencing to identify organized lymphoid aggregates adjacent to hair follicles in human scalp and forearm biopsies. These structures, termed follicle-associated lymphoid tissue (FALT), contain B-cell follicles, T-cell zones, and antigen-presenting dendritic cells arranged in a manner structurally and functionally similar to gut-associated lymphoid tissue (GALT), but uniquely adapted to the skin’s exposure profile.
Unlike inflammatory immune responses that cause redness and swelling, FALT operates under tolerogenic conditions, promoting immune surveillance without activation—unless a threat is detected. Dr. Chen explained in a recent interview:
“We’ve long known the skin has immune cells, but discovering organized lymphoid structures within hair follicles changes how we view cutaneous immunity. These aren’t random infiltrates—they’re strategically positioned monitoring posts.”
This finding aligns with growing evidence that the skin immune system is highly compartmentalized. According to a 2024 review in Nature Immunology, tissue-resident memory T cells (TRM) in the skin can persist for years and respond rapidly to re-infection. The presence of FALT suggests a structural basis for how these cells are maintained and regulated locally.
Geo-Epidemiological Bridging: Implications for Public Health and Regulatory Pathways
The discovery of FALT has immediate relevance for public health strategies, particularly in vaccine delivery and autoimmune disease management. In the United States, the FDA has increasingly supported exploration of non-injectable vaccine routes, including microneedle patches targeting the skin. Understanding that hair follicles contain organized immune tissue could optimize antigen delivery to enhance immunogenicity while minimizing systemic side effects.

In Europe, where the EMA has approved several topical immunomodulators for atopic dermatitis and psoriasis, this research may inform next-generation therapies designed to work *with* follicular immunity rather than suppress it broadly. For instance, biologics targeting IL-17 or IL-23 pathways could be refined to spare FALT-mediated surveillance, reducing infection risks.
In the UK, the NHS spends over £600 million annually on managing psoriasis and eczema—conditions linked to dysregulated skin immunity. If FALT dysfunction contributes to these diseases, restoring its regulatory function could become a therapeutic goal. Dr. Chen noted:
“When we looked at biopsies from psoriasis patients, we observed disrupted FALT architecture—fewer dendritic cell networks and abnormal T-cell clustering. This suggests the structure isn’t just present; its integrity matters for disease prevention.”
Funding, Bias Transparency, and Scientific Rigor
The UC Riverside study was primarily funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), part of the National Institutes of Health (NIH), under grant R01-AR078901. Additional support came from the UC Riverside School of Medicine’s Dean’s Innovation Award. No pharmaceutical industry funding was involved in the initial discovery phase, minimizing conflict-of-interest concerns.
To validate findings, researchers compared immunocompetent tissue from healthy donors (n=12) with samples from individuals with autoimmune skin conditions (n=8) and cutaneous lymphoma (n=5). All human tissue was obtained with informed consent under IRB-approved protocols. While the study did not involve clinical trials, it lays the groundwork for future Phase I trials testing topical agonists or antagonists of follicular immune signaling.
Mechanism of Action: How Follicular Immunity Maintains Skin Balance
FALT functions through a delicate equilibrium between immune activation and tolerance. Dendritic cells within the follicle infundibulum sample antigens from the skin surface and hair shaft. In steady state, these cells migrate to T-cell zones and promote the differentiation of regulatory T cells (Treg) via secretion of TGF-β and retinoic acid—mechanisms also seen in GALT. This process suppresses effector T-cell responses to commensal microbes like Cutibacterium acnes and Staphylococcus epidermidis, preventing chronic inflammation.
Upon pathogen invasion or neoplastic transformation, dendritic cells shift to an immunogenic phenotype, upregulating MHC-II and co-stimulatory molecules (CD80/86), thereby activating effector T cells. This switch is modulated by local cytokines such as IL-1β and TNF-α, which are tightly controlled to avoid bystander damage. Notably, the hair follicle’s unique microenvironment—including sebaceous lipids and antimicrobial peptides—further shapes immune cell behavior.
This mechanism explains why topical immunosuppressants, while effective for eczema, can increase susceptibility to skin infections: they may disrupt FALT’s surveillance capacity. Conversely, agents that bolster follicular immunity—such as toll-like receptor (TLR) agonists—are being investigated as adjuvants for cancer immunotherapy or preventive vaccines.
Contraindications & When to Consult a Doctor
While enhancing follicular immunity holds promise, it is not appropriate for all individuals. Patients with a history of autoimmune skin disorders—such as lupus erythematosus, vitiligo, or severe alopecia areata—should avoid experimental therapies aimed at stimulating FALT without medical supervision, as overactivation could exacerbate tissue damage.

Individuals undergoing chemotherapy or immunosuppressive therapy (e.g., for organ transplant) should consult their dermatologist before using any topical immune-modulating agents, even natural compounds, as unintended inflammation or folliculitis may occur.
Seek medical attention if you experience persistent redness, pustules, or tender nodules around hair follicles—signs of folliculitis—or if you notice unexplained skin lesions that bleed, change color, or fail to heal, which could indicate skin cancer requiring biopsy.
The Future of Cutaneous Immunity: From Discovery to Clinical Translation
This discovery repositions the hair follicle not merely as a keratin-producing structure but as a dynamic immune organ. Future research will focus on mapping FALT across different skin sites (e.g., face vs. Palms), assessing its role in aging (where immune surveillance declines), and exploring whether genetic polymorphisms in follicle-associated genes correlate with susceptibility to skin cancer or inflammatory dermatoses.
From a public health perspective, leveraging FALT could revolutionize transdermal vaccine delivery—particularly for pathogens requiring mucosal or cutaneous immunity, such as HPV or influenza. Early preclinical models show that targeting follicular dendritic cells with nanoparticle-antigen complexes enhances IgA production in skin-associated lymphoid tissue.
As Dr. Chen emphasized:
“We’re just beginning to understand how the skin talks to the immune system. Hair follicles aren’t passive—they’re active participants in keeping us healthy.”
References
- Chen M, et al. Follicle-associated lymphoid tissue in human skin regulates cutaneous immune homeostasis. Nature Immunology. 2026;27(4):567-580. Doi:10.1038/s41590-026-01452-3.
- Nagao K, et al. Tissue-resident memory T cells in skin: maintenance and function. Annual Review of Immunology. 2024;42:389-415. Doi:10.1146/annurev-immunol-042023-015642.
- Schmidt A, et al. Dendritic cell subsets in human skin: regulators of tolerance and immunity. Journal of Investigative Dermatology. 2025;145(2):301-310. Doi:10.1016/j.jid.2024.10.015.
- World Health Organization. Vaccine delivery systems: current status and future directions. WHO/IVB/24.07. Geneva: WHO; 2024.
- U.S. Food and Drug Administration. Guidance for Industry: Topical Dermatological Products. FDA; 2023. Https://www.fda.gov/media/135678/download.