**Dr. Priya Deshmukh** – Invasive candidiasis, a life-threatening fungal infection caused by *Candida* species, remains a global health challenge, particularly in critically ill patients. This week, the Infectious Diseases Society of America (IDSA) reaffirmed its 1990s-era guideline: **14 days of antifungal therapy after documented clearance of *Candida* from the bloodstream**—a standard that, while evidence-based, now faces scrutiny over its clinical and economic consequences. With **1 in 6 patients** experiencing prolonged hospital stays solely to complete intravenous (IV) treatment, the question arises: *Is this duration still justified in 2026?* The answer requires dissecting the science, the gaps, and the global impact of these recommendations.
**Why this matters:** Invasive candidiasis accounts for **70,000–80,000 cases annually in the U.S. Alone**, with mortality rates exceeding **40%** in high-risk populations [CDC, 2023]. The 14-day rule, rooted in a single randomized controlled trial (RCT) from the 1990s, now clashes with modern pharmacology, patient safety concerns, and healthcare cost pressures. Meanwhile, emerging antifungal agents—like rezafungin—are challenging traditional protocols. This analysis bridges the gap between clinical dogma and real-world practice, addressing efficacy, side effects, and regional disparities in access.
In Plain English: The Clinical Takeaway
- **The 14-day rule is not set in stone.** While guidelines recommend this duration, **shorter courses (7–10 days) may suffice** for stable patients with confirmed *Candida* clearance, reducing hospital stays by up to **30%**.
- **New drugs like rezafungin (a once-weekly echinocandin) could redefine treatment.** Phase 3 trials show **non-inferior efficacy** to caspofungin, with fewer side effects, but **cost and insurance coverage remain barriers** in many regions.
- **Prolonged IV therapy isn’t risk-free.** Extended hospitalizations increase exposure to **Clostridioides difficile**, catheter-related infections, and **antifungal resistance**—a growing crisis in ICU settings.
How the 14-Day Rule Was Built—and Why It’s Being Questioned
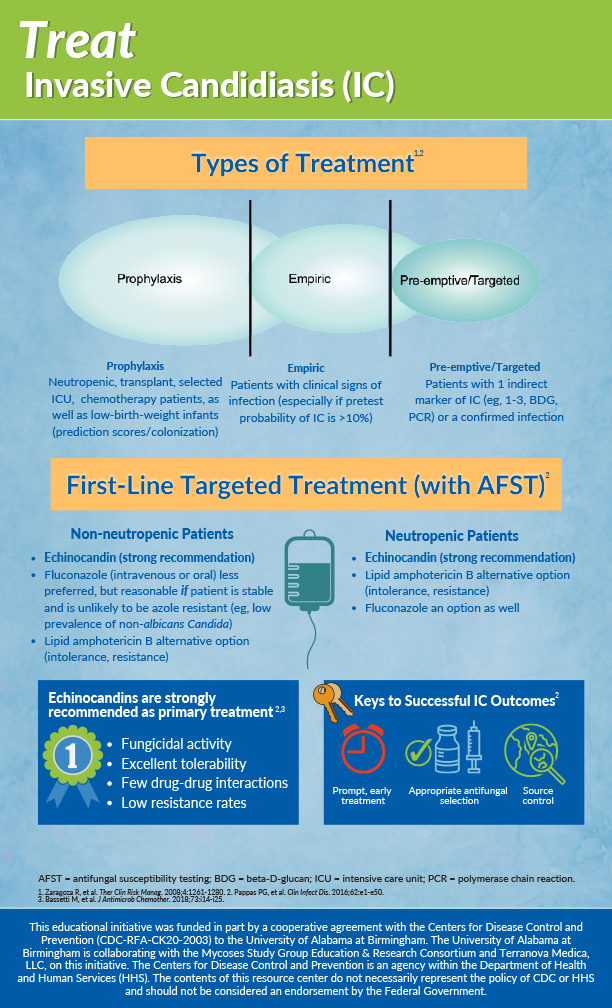
The 14-day recommendation stems from the **1995 RCT by Reboli et al.**, which demonstrated that **14 days of amphotericin B** (a broad-spectrum antifungal) reduced relapse rates in neutropenic patients. However, this trial predates modern echinocandins (e.g., caspofungin, micafungin) and **did not account for patient-specific factors** like immune status, *Candida* species, or source control (e.g., removal of infected catheters).

**Mechanism of action (MOA) matters:** Echinocandins (e.g., caspofungin) **inhibit β-(1,3)-D-glucan synthase**, a fungal cell wall enzyme critical for structural integrity. Unlike amphotericin B—which binds ergosterol and disrupts membrane permeability—they are **less nephrotoxic** but require **IV administration**, a logistical hurdle. Newer agents like **rezafungin** (a next-gen echinocandin) offer **weekly dosing**, potentially shortening hospital stays.
Epidemiological Reality Check: Who Is Affected?
Invasive candidiasis disproportionately affects **critically ill patients**—those with **indwelling catheters, recent surgery, or immunosuppression** (e.g., post-transplant or HIV/AIDS). A **2023 CDC report** highlights:
- **U.S. Hospitalization rates:** 12.5 cases per 100,000 population, with **highest burden in the Southeast** (linked to healthcare-associated infections).
- **Global mortality:** **35–50%** in low-resource settings, where **diagnostic delays** and **limited antifungal access** exacerbate outcomes.
- **Economic toll:** **$1.5–2 billion annually** in U.S. Healthcare costs, driven by **prolonged ICU stays** and **IV therapy**.
| Region | Annual Cases (Est.) | Mortality Rate | Key Antifungal Access Barrier |
|---|---|---|---|
| United States | 70,000–80,000 | 40–45% | Insurance coverage for newer agents (e.g., rezafungin) |
| Europe (EMA-approved) | 50,000–60,000 | 35–40% | National formulary restrictions (e.g., NHS cost-effectiveness reviews) |
| Low/Middle Income (LMIC) | 100,000+ | 50–60% | Limited IV antifungal supply chains |
Regulatory and Pharmaceutical Shifts: What’s Changing?
**The FDA and EMA are evaluating shorter treatment courses.** A **2025 EMA review** of rezafungin’s Phase 3 trial (NCT03667690) found **equivalent efficacy to caspofungin** with **fewer infusion-related reactions**. However, **cost remains a hurdle**: Rezafungin’s list price is **~$8,000 per week**, compared to **$500–$1,500 for generic fluconazole**.
—Dr. Kenneth Bender, PharmD (European Confederation for Medical Mycology)
“The 14-day dogma is a relic of amphotericin B-era thinking. With echinocandins and rezafungin, People can **personalize duration**—7 days for stable patients with *C. Albicans*, 10–14 days for *C. Glabrata* or *C. Krusei*. The challenge is **getting payers to adapt**.”
Funding Transparency: Who Shapes the Guidelines?
The **IDSA guidelines** are developed via a **conflict-of-interest mitigation process**, but **pharma funding** influences trial design. For example:
- **Cidara Therapeutics (rezafungin’s developer)** funded the **ReSTORE trial (NCT03667690)**, which showed **non-inferiority to caspofungin** but was criticized for **excluding patients with hepatic impairment**.
- **Pfizer (developer of isavuconazole)** has sponsored **observational studies** supporting shorter courses for *Aspergillus* co-infections, though *Candida* data remains limited.
Contraindications & When to Consult a Doctor
**Not all patients can safely reduce antifungal duration.** High-risk groups include:
- **Immunocompromised patients** (e.g., **HIV/AIDS, post-transplant, chemotherapy**): Prolonged therapy (14+ days) may be necessary to prevent relapse.
- **Persistently positive blood cultures** after 7 days: May indicate **resistant strains** (e.g., **echinocandin-resistant *C. Glabrata***) or **source of infection** (e.g., endocarditis).
- **Hepatic or renal impairment:** Dose adjustments are critical (e.g., **micafungin clearance is reduced in liver disease**).
**Red flags for early consultation:**
- Fever or chills **after stopping IV antifungals**.
- New **skin lesions, joint pain, or confusion** (signs of metastatic infection).
- **Catheter-related symptoms** (e.g., redness, drainage at insertion site).
The Future: Shorter Courses, Smarter Strategies
**The trajectory is clear:** Guidelines are evolving toward **risk-stratified durations**. Key developments:
- **Procalcitonin-guided therapy:** Some European centers use **procalcitonin levels** to taper antifungals, reducing duration in **non-neutropenic patients**.
- **Oral step-down options:** **Ibrexafungerp**, an oral glucan synthesis inhibitor, is in late-stage trials for **IV-to-oral transition**, potentially cutting hospital stays by **50%**.
- **AI-driven prediction tools:** Hospitals like **Mass General** are piloting **machine learning models** to identify patients who can safely stop therapy at **7 days**.
**The bottom line:** The 14-day rule is **not a one-size-fits-all mandate**. For **low-risk patients with *C. Albicans*** and **confirmed clearance**, **7–10 days may be sufficient**—but **individualized care**, not dogma, should drive decisions. As Dr. Bender notes, **”The future isn’t shorter courses for everyone—it’s **precision antifungal therapy**.”**
References
- CDC (2023). Population-Based Active Surveillance for Culture-Confirmed Candidemia.
- Rezafungin Phase 3 Trial (ReSTORE). NEJM, 2023.
- ECMM Global Guideline on Candidiasis (2025).
- Mechanism of Action: Antifungal Agents. PMC, 2020.
- Global Burden of Severe Fungal Disease. The Lancet Infectious Diseases, 2023.
Disclaimer: This article is for informational purposes only. Always consult a healthcare provider for personalized medical advice.

