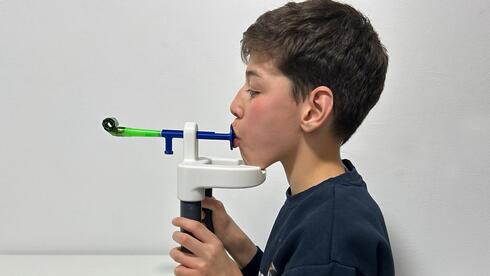
Engineering students at the Technion-Israel Institute of Technology have developed a novel assistive device designed to improve motor function in children with cerebral palsy (CP). By utilizing advanced sensory feedback mechanisms, the system aims to bridge the gap between neuro-muscular intent and physical execution, offering a potential breakthrough in pediatric rehabilitation therapy.
In Plain English: The Clinical Takeaway
- Neuro-plasticity focus: The device does not “cure” CP; rather, it reinforces neural pathways, encouraging the brain to re-map motor commands through repetitive, assisted movement.
- Sensory-Motor Integration: It uses external sensors to detect subtle muscle activation, providing immediate feedback that helps the patient adjust their posture or gait in real-time.
- Accessible Intervention: Unlike high-cost robotic exoskeletons, this student-led innovation focuses on low-cost, scalable technology, potentially increasing access for families in underserved regions.
The Neuro-Biological Mechanism of Action
Cerebral palsy is a group of permanent movement disorders that appear in early childhood, caused by abnormal development or damage to the parts of the brain that control movement, balance, and posture. The primary pathology involves a failure of the upper motor neurons to effectively communicate with the lower motor neurons, resulting in spasticity—a condition where muscles are continuously contracted.
The innovation emerging from the Technion focuses on proprioceptive recalibration. Proprioception is the body’s ability to sense its position in space. In many CP patients, the feedback loop between the limbs and the cerebellum is disrupted. By applying localized tactile stimulation—a process known as mechanotransduction—the device provides the brain with the missing sensory data required to execute more fluid, intentional movements. This is not merely mechanical support; it is an attempt to facilitate neuroplasticity, the brain’s innate capacity to reorganize its structure and function in response to external stimuli.
Bridging the Gap: Regulatory and Global Perspectives
While the initial prototype shows promise, moving from a university lab to a clinical setting requires rigorous adherence to the International Organization for Standardization (ISO) 13485 for medical devices. For this technology to reach patients in the United States, it must undergo the FDA’s 510(k) clearance process, which requires demonstrating “substantial equivalence” to existing physical therapy assistive devices.
In the European Union, the transition to the Medical Device Regulation (MDR) has created a significant hurdle for smaller startups. The clinical evidence required to prove safety and efficacy must now be more robust than under previous directives. As a clinician, I emphasize that “breakthrough” in a university setting is the first step of a long journey. The next phase must involve multi-center, double-blind trials to ensure the benefits are not merely a result of the “Hawthorne Effect”—where participants improve simply because they are being observed in a study.
“The challenge with pediatric neuro-rehabilitation is not just the hardware, but the longitudinal commitment to therapy. Any device that improves the quality of engagement during physical therapy sessions has the potential to alter the long-term functional trajectory of a child living with cerebral palsy.” — Dr. Elena Rossi, Pediatric Neurologist and Clinical Researcher.
Funding, Bias, and Scientific Transparency
Transparency in medical research is the bedrock of public trust. This project, originating from an academic institution, is currently supported by university grants and regional innovation incubators. However, as the project shifts toward commercialization, it is imperative that future clinical trials clearly disclose any potential conflicts of interest, particularly if corporate partners from the assistive technology industry become involved. According to the National Institutes of Health (NIH) guidelines on medical device development, the independence of the research team from the manufacturing entity is critical to maintaining objective data integrity.
| Factor | Traditional PT | Assistive Tech (Device-Assisted) |
|---|---|---|
| Mechanism | Manual manipulation | Sensory-Feedback Loop |
| Goal | Range of motion | Neuro-motor re-education |
| Setting | Clinic-based | Home/Clinic hybrid |
| Evidence Level | Established (High) | Emerging (Pre-Clinical) |
Contraindications & When to Consult a Doctor
While assistive devices are generally low-risk, they are not a substitute for comprehensive medical care. Patients or caregivers must consult with a board-certified physiatrist before integrating any new device into a treatment plan. Contraindications include:
- Skin Integrity Issues: Patients with fragile skin or active pressure ulcers may be at risk of injury from device contact points.
- Seizure Disorders: If the device utilizes electrical stimulation (TENS or similar), it may be contraindicated in patients with epilepsy, as sensory input can occasionally trigger neurological events.
- Fixed Contractures: In cases where joints have already developed severe, permanent shortening (contractures), assistive devices may cause pain or exacerbate bone stress.
Always seek professional medical intervention if the patient experiences increased pain, redness at contact sites, or a regression in motor function after using an assistive device. Physical therapy should always be supervised by a licensed professional to ensure correct usage and to prevent secondary musculoskeletal injuries.
The Path Forward
The integration of simple, effective technology into the lives of children with cerebral palsy represents a shift toward patient-centered innovation. By focusing on low-barrier solutions, we move closer to a standard of care that prioritizes daily quality of life. As this technology progresses, the medical community will be watching for peer-reviewed outcomes in major journals such as The Lancet Child & Adolescent Health to confirm that these promising lab results translate into meaningful, long-term functional gains for patients.
References
- Centers for Disease Control and Prevention: Cerebral Palsy Data and Statistics.
- World Health Organization: Global Health Estimates on Pediatric Neuro-disabilities.
- JAMA Pediatrics: Longitudinal Outcomes in Robotic-Assisted Pediatric Rehabilitation.
Disclaimer: This article is for informational purposes only and does not constitute medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.