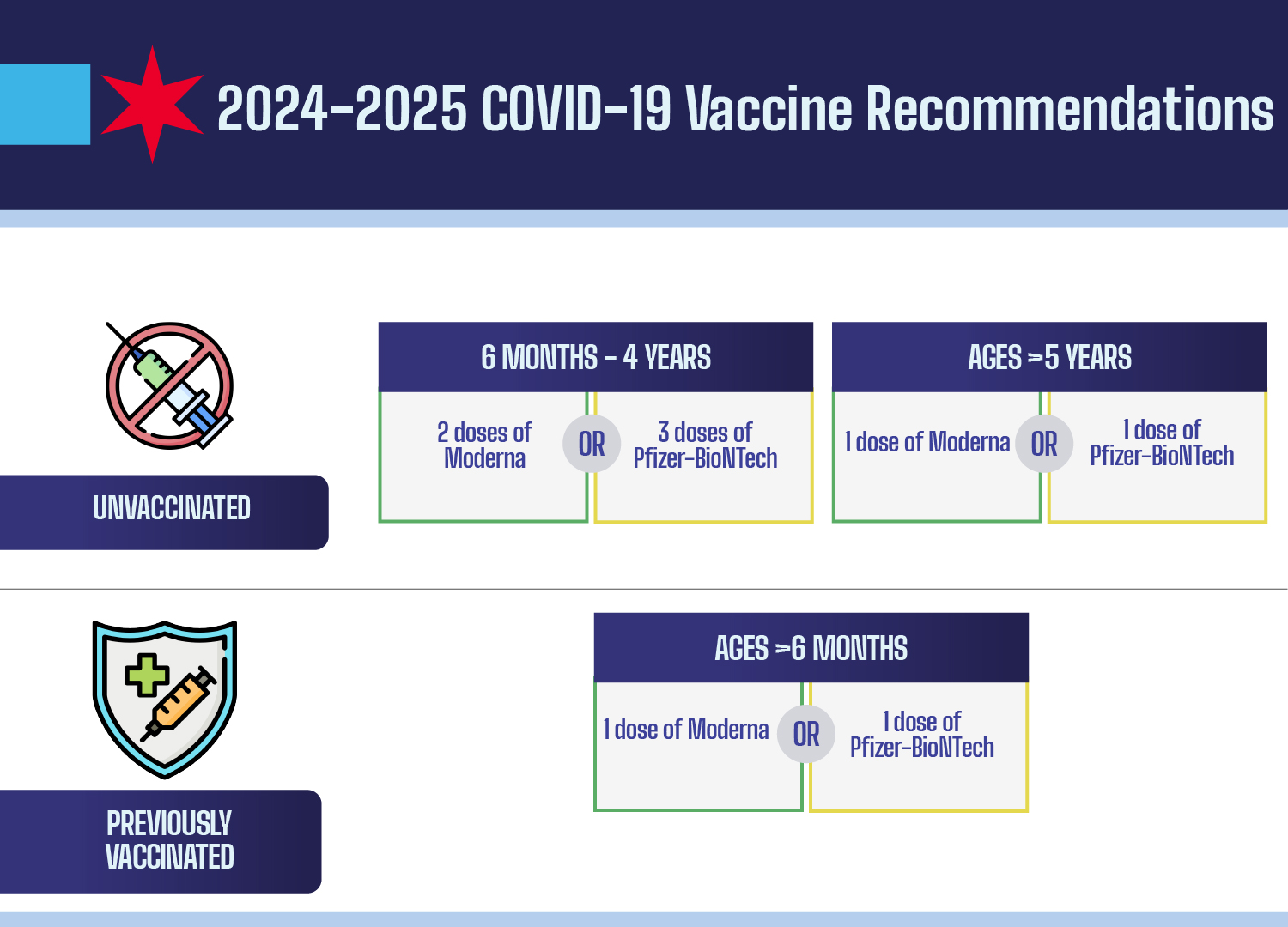
The Korea Disease Control and Prevention Agency (KDCA) has released effectiveness data for the 2024-2025 COVID-19 vaccination cycle, confirming that current boosters significantly reduce severe illness and hospitalization. Reported adverse events remain predominantly mild, consisting primarily of localized injection-site pain and transient myalgia (muscle aches).
This data arrives at a critical juncture as global health authorities transition toward a seasonal vaccination model, similar to the annual influenza shot. For patients, In other words the focus has shifted from preventing every single “sniffle” to preventing the catastrophic collapse of pulmonary function and the long-term neurological sequelae associated with severe COVID-19. Understanding the delta between “infection prevention” and “severe disease prevention” is essential for maintaining public trust in immunization programs.
In Plain English: The Clinical Takeaway
- Protection Focus: The vaccine is designed to keep you out of the hospital, not necessarily to stop you from catching a mild cold-like version of the virus.
- Common Side Effects: Sore arms and muscle aches are signs that your immune system is “practicing” its response; they are normal and temporary.
- Timing Matters: Staying updated with the latest seasonal formula is key because the virus mutates (changes its shape), making older vaccines less effective.
The Mechanism of Action: How Updated Boosters Target Viral Drift
The efficacy of the 2024-2025 vaccines relies on targeting “viral drift”—the gradual accumulation of mutations in the spike protein of the SARS-CoV-2 virus. Most current vaccines utilize mRNA (messenger RNA) technology. This acts as a set of instructions telling your cells to produce a harmless piece of the spike protein, which triggers an immune response.

When the KDCA reports “preventative effects,” they are measuring the reduction in viral load and the presence of neutralizing antibodies. These antibodies bind to the virus, preventing it from entering human cells via the ACE2 receptor, which is the primary gateway the virus uses to infect the lungs and vascular system.
However, the “information gap” in many public reports is the distinction between sterilizing immunity (stopping infection entirely) and clinical immunity (preventing severe disease). Because the virus evolves rapidly, sterilizing immunity is short-lived, but clinical immunity—provided by T-cells that remember the virus—lasts much longer and protects the lower respiratory tract.
“The goal of seasonal vaccination is not the eradication of the virus, but the management of its impact on healthcare infrastructure and the prevention of individual mortality.” — Dr. Maria Van Kerkhove, Technical Lead on COVID-19, World Health Organization (WHO).
Global Regulatory Alignment and Patient Access
The KDCA’s findings mirror trends observed by the Centers for Disease Control and Prevention (CDC) in the United States and the European Medicines Agency (EMA). All three agencies have converged on the strategy of “variant-updated” boosters.
In the US, the FDA’s approval process for these updates has been streamlined to ensure that the vaccine formula matches the circulating strains (such as the JN.1 or KP.2 lineages) as closely as possible. In the UK, the NHS has prioritized high-risk groups—the elderly and immunocompromised—recognizing that while healthy adults may experience mild illness, the risk of “Long COVID” or systemic inflammation remains high for vulnerable populations.
Funding for these large-scale surveillance programs is typically a hybrid of government public health budgets and pharmaceutical research grants. The KDCA’s monitoring is government-funded, which ensures a level of objectivity in reporting adverse events, as their primary mandate is population health rather than profit.
Comparative Analysis of Vaccine Outcomes
The following table summarizes the general clinical expectations for the 2024-2025 vaccine cycle based on aggregated data from the KDCA and global health partners.
| Clinical Metric | Unvaccinated / Outdated Booster | Updated 2024-2025 Booster | Clinical Significance |
|---|---|---|---|
| Risk of Severe Pneumonia | Moderate to High | Significantly Reduced | Prevents respiratory failure |
| Hospitalization Rate | Baseline | 30-70% Lower | Reduces ICU burden |
| Common Side Effects | N/A | Localized Pain / Fever | Transient immune activation |
| Symptomatic Infection | High Risk | Moderate Risk | Vaccine prevents death, not all colds |
Addressing the “Myalgia” and Localized Inflammation
The KDCA mentions “local pain and muscle aches” (myalgia). From a clinical perspective, this is an innate immune response. When the vaccine is administered, the body releases cytokines—signaling proteins that recruit white blood cells to the area. This causes temporary inflammation, which we perceive as pain or swelling.
This process is a positive indicator that the body is recognizing the antigen. It is fundamentally different from an allergic reaction (anaphylaxis), which is rare and occurs almost immediately after injection. The statistical probability of a severe adverse reaction remains extremely low compared to the risk of multi-organ failure associated with an uncontrolled COVID-19 infection in high-risk individuals.
Contraindications & When to Consult a Doctor
While the vaccine is safe for the vast majority of the population, certain individuals must exercise caution. You should consult a healthcare provider if you have:
- A history of severe allergic reactions (anaphylaxis) to any component of the vaccine, such as polyethylene glycol (PEG).
- Acute febrile illness: If you have a high fever or a severe active infection, vaccination should be postponed until recovery.
- Severe Immunocompromised Status: Patients on high-dose corticosteroids or chemotherapy may have a diminished response to the vaccine and may require a different dosing schedule.
Seek immediate medical attention if you experience: Shortness of breath, chest pain, or swelling of the face and throat within six hours of vaccination.
The Path Forward: Toward Endemic Stability
As we move through 2026, the trajectory of COVID-19 is shifting from a pandemic crisis to an endemic management phase. The data from the KDCA reinforces the necessity of the “booster” strategy. By maintaining a baseline of antibody protection, we prevent the healthcare system from becoming overwhelmed during winter surges.
The objective remains clear: we cannot stop the virus from existing, but through scientifically literate vaccination strategies, we can stop it from being lethal. The transition to a seasonal cadence is not a failure of the vaccine, but a victory of adaptation in the face of a mutating pathogen.
References
- PubMed Central (National Library of Medicine) – Clinical trial data on mRNA efficacy.
- World Health Organization (WHO) – Global surveillance and variant tracking reports.
- The Lancet – Peer-reviewed longitudinal studies on vaccine durability.
- Journal of the American Medical Association (JAMA) – Analysis of adverse event probabilities.