Recent research suggests a paradigm shift in understanding lacunar strokes—small infarcts in the brain’s deep arteries. Even as traditionally attributed to vessel narrowing, new evidence indicates that abnormal arterial widening (dilation) can trigger these strokes by disrupting blood flow, potentially redefining how clinicians diagnose and treat small vessel disease.
For decades, the medical community viewed lacunar strokes primarily through the lens of stenosis—the narrowing of arteries due to plaque buildup or wall thickening. However, the findings published this week challenge this singular narrative. By demonstrating that the widening of these microscopic vessels can be equally catastrophic, we are forced to reconsider the hemodynamic triggers of ischemia in the deep brain. Here’s not merely an academic nuance. We see a clinical pivot that could alter how we manage millions of patients globally who suffer from chronic hypertension, and diabetes.
In Plain English: The Clinical Takeaway
- Not just “clogged pipes”: We previously thought small strokes happened because arteries became too narrow. Now we recognize they can happen because arteries become too wide.
- Turbulence is the enemy: When an artery widens abnormally, blood doesn’t flow smoothly; it swirls (turbulence), which can lead to the formation of tiny clots.
- Imaging matters: This discovery emphasizes the need for high-resolution brain imaging to identify “stretched” vessels before a stroke occurs.
The Hemodynamic Shift: From Stenosis to Dilation
To understand this breakthrough, we must examine the mechanism of action—the specific biological process that leads to the injury. Traditionally, lacunar strokes were linked to lipohyalinosis, a process where the walls of small penetrating arteries thicken and lose elasticity, narrowing the lumen (the open space inside the vessel). This narrowing restricts blood flow, leading to a “starved” area of brain tissue.
The emerging data suggests a counterintuitive process: arterial dilation. When these small vessels widen beyond their physiological limit, the laminar flow—the smooth, parallel movement of blood—is disrupted. This creates “stagnation zones” or eddies. In these pockets of slow-moving blood, platelets are more likely to aggregate, forming micro-thrombi (tiny clots) that block the vessel entirely, resulting in a lacunar infarct.
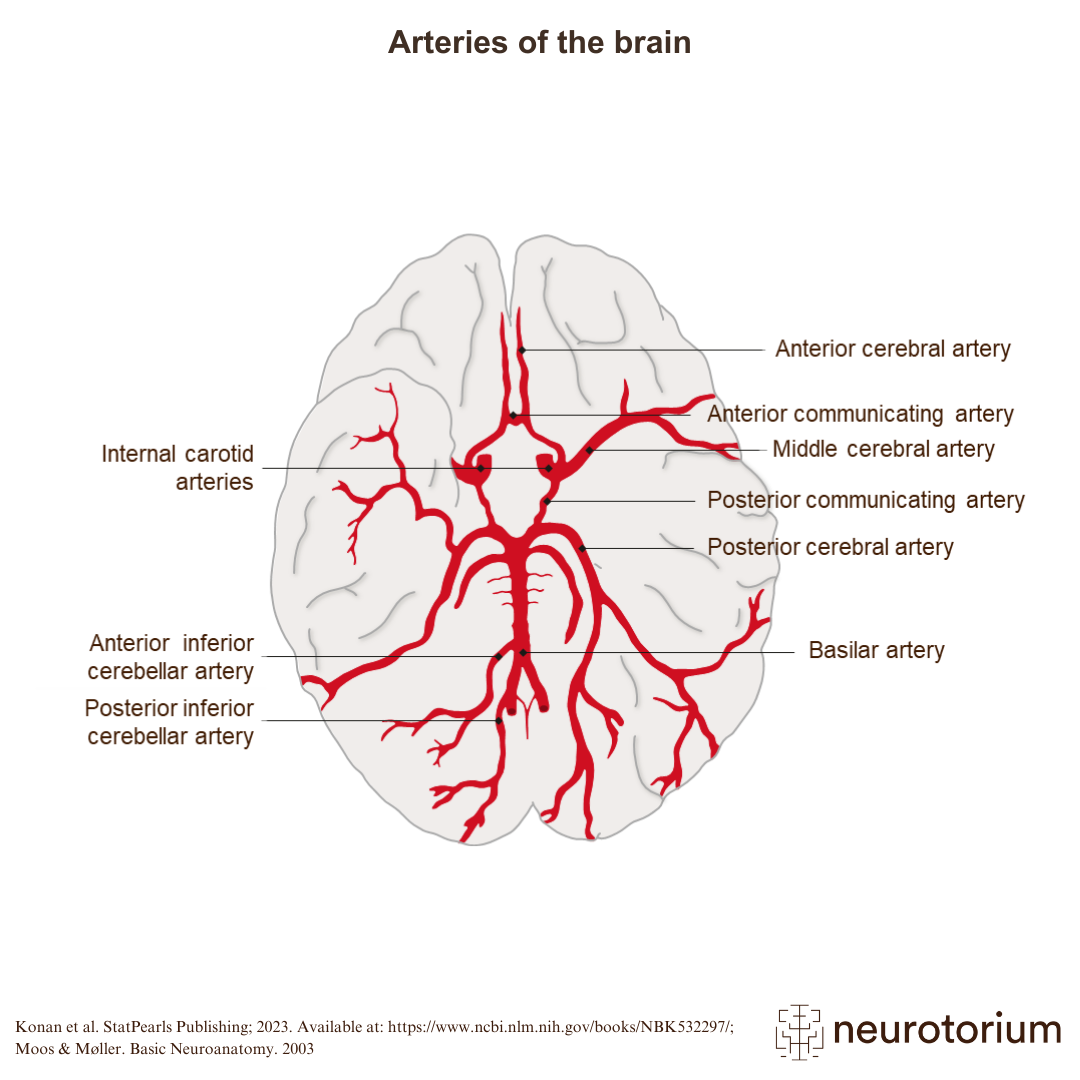
This process is closely tied to the degradation of the internal elastic lamina, the structural layer that keeps arteries firm. When this layer fails, the vessel doesn’t just leak; it balloons. This structural failure is often the result of long-term exposure to high blood pressure, which puts constant mechanical stress on the vessel walls.
“The discovery that dilation plays a role in lacunar infarction forces us to move beyond the ‘plumbing’ metaphor of simple blockages. We are looking at a complex failure of vascular geometry where the shape of the vessel is just as critical as its diameter.”
Global Clinical Implications and Diagnostic Access
The transition from theory to bedside varies significantly across global healthcare systems. In the United Kingdom, the NHS is currently integrating more advanced MRI protocols, but the widespread adoption of high-resolution vessel wall imaging (VWI) remains a challenge due to resource allocation. In the United States, access to these diagnostics is often fragmented by insurance tiers, meaning patients in underserved regions may only be diagnosed after a stroke has occurred, rather than during the dilation phase.
From a regulatory perspective, the FDA and the European Medicines Agency (EMA) have historically focused on anti-platelet therapies that prevent clots in narrowed vessels. If dilation is a primary driver, we may need to investigate new pharmacological interventions that stabilize the vessel wall itself, rather than just thinning the blood. This could open the door for a new class of “vascular stabilizing” agents.
| Feature | Classical Model (Stenosis) | Emerging Model (Dilation) |
|---|---|---|
| Primary Mechanism | Vessel narrowing/blockage | Vessel widening/dilation |
| Blood Flow Pattern | Reduced volume/Reduced pressure | Turbulence/Stagnation zones |
| Vessel Wall Change | Thickening (Lipohyalinosis) | Elastic lamina degradation |
| Primary Trigger | Plaque/Inflammation | Mechanical wall stress |
Funding, Bias, and the Path to Peer Review
Transparency in medical reporting is paramount. Much of the underlying research into small vessel disease (SVD) is funded by government grants—such as those from the National Institutes of Health (NIH) in the US and the Wellcome Trust in the UK—rather than pharmaceutical companies. This reduces the risk of commercial bias, as there is currently no “blockbuster drug” specifically designed to treat arterial dilation in the brain.

However, the scientific community remains cautious. While the correlation between widening and stroke is strong, we are still awaiting large-scale, double-blind placebo-controlled trials—studies where neither the patient nor the doctor knows who is receiving a treatment—to prove that treating dilation actually reduces stroke recurrence. Until then, this remains a high-probability hypothesis supported by observational data.
For those seeking deeper technical data, the PubMed database and the The Lancet provide extensive longitudinal studies on cerebral small vessel disease that contextualize these findings within the broader scope of neurology.
Contraindications & When to Consult a Doctor
It is critical to note that this research does not suggest patients should change their current medication. Do not alter your dosage of antihypertensives or anticoagulants based on this news. Changing these medications without supervision can lead to rebound hypertension or hemorrhagic stroke (bleeding in the brain).

Consult a neurologist immediately if you experience any of the following “Speedy” symptoms, which indicate an active stroke regardless of the cause:
- Face: One side of the face droops or is numb.
- Arms: One arm drifts downward when both are raised.
- Speech: Slurred speech or inability to speak clearly.
- Time: Call emergency services (911/999) immediately.
Patients with a history of uncontrolled hypertension or those who have previously suffered a “silent” stroke (detected only on imaging) should discuss high-resolution MRA (Magnetic Resonance Angiography) with their provider to assess their vascular architecture.
The Future of Neurovascular Intelligence
We are entering an era of “precision neurology.” The shift from treating all lacunar strokes as a monolithic condition to distinguishing between stenotic and dilatory causes will allow for more tailored therapies. By integrating AI-driven imaging that can detect subtle arterial widening, we may soon be able to predict stroke risk with unprecedented accuracy.
The goal is no longer just to react to the stroke, but to stabilize the vessel before the first clot ever forms. This is the essence of public health intelligence: converting a complex biological observation into a preventative clinical strategy.
References
- World Health Organization (WHO) – Cardiovascular Diseases Fact Sheets
- Centers for Disease Control and Prevention (CDC) – Stroke Prevention Guidelines
- PubMed – National Library of Medicine: Cerebral Small Vessel Disease Research
- American Heart Association (AHA) – Guidelines for the Management of Stroke