In a landmark advance for genetic medicine, a recent UCLA graduate became one of the first patients in the United States to receive a newly approved gene therapy for severe sickle cell disease, a treatment that modifies hematopoietic stem cells to produce functional hemoglobin and prevent red blood cell sickling. This milestone, reported following the FDA’s December 2023 approval of exagamglogene autotemcel (Casgevy), represents the first CRISPR-based gene-editing therapy authorized for clinical use in sickle cell disease, offering potential curative relief for thousands living with the debilitating inherited blood disorder.
How CRISPR Gene Editing Corrects the Sickle Cell Mutation at the Molecular Level
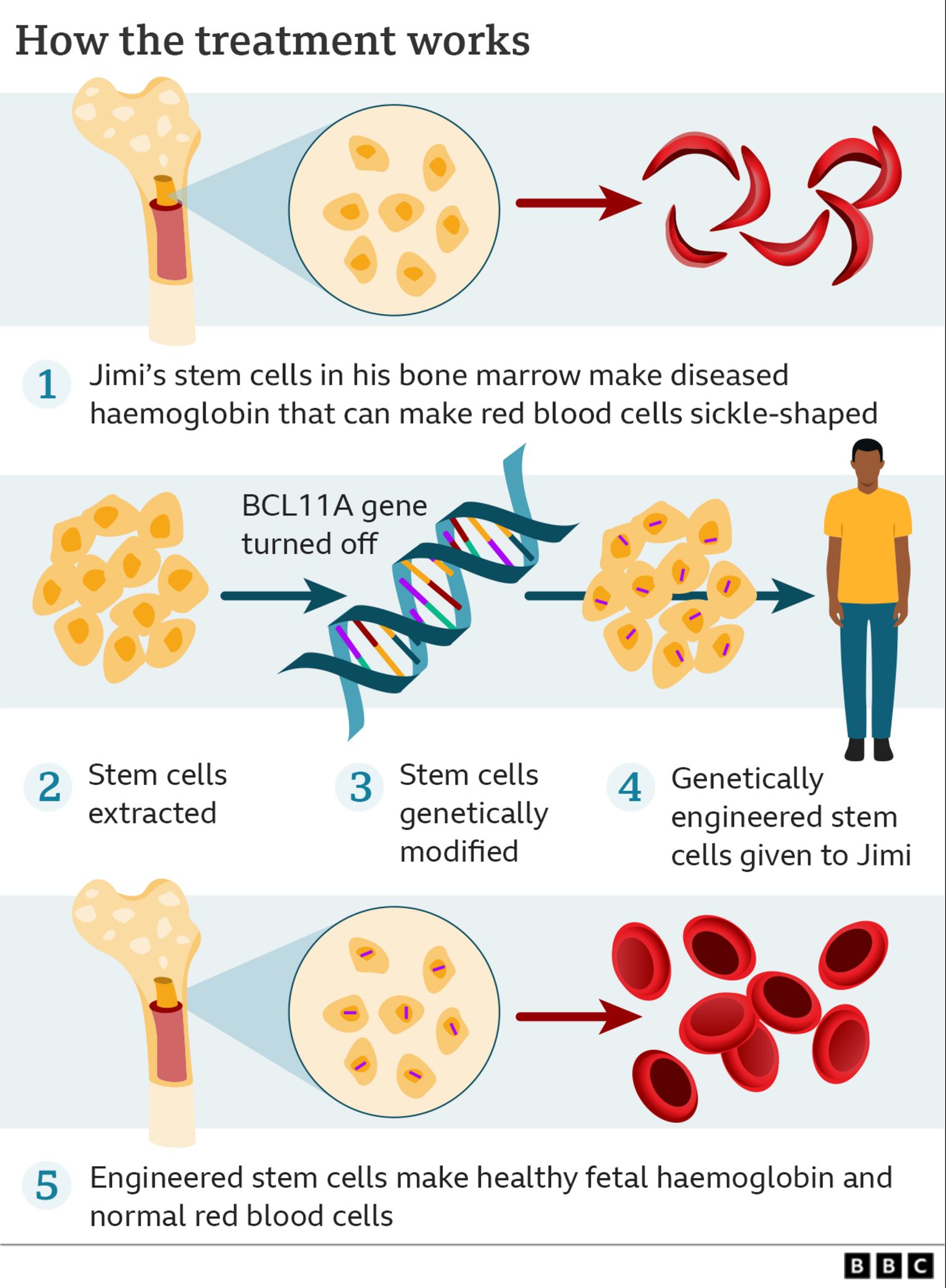
The therapy, developed by Vertex Pharmaceuticals and CRISPR Therapeutics, involves extracting a patient’s own hematopoietic stem cells, then using CRISPR-Cas9 technology to precisely edit the BCL11A gene—a transcriptional repressor that silences fetal hemoglobin production after birth. By disrupting BCL11A’s enhancer region, the therapy reactivates fetal hemoglobin (HbF) synthesis in red blood cells, which compensates for the defective adult hemoglobin S (HbS) responsible for sickling. Unlike symptomatic treatments such as hydroxyurea or voxelotor, this approach targets the genetic root cause, aiming for sustained HbF elevation above 20%—a threshold clinically associated with near-complete prevention of vaso-occlusive crises.

In Plain English: The Clinical Takeaway
- This one-time treatment reprograms the patient’s own blood stem cells to make healthy fetal hemoglobin, reducing sickle-shaped cells and pain crises.
- Early trial data show most patients remain free of severe pain episodes for at least two years post-treatment, with no graft failures reported.
- Whereas promising, the therapy requires chemotherapy-like conditioning and carries risks including infertility and temporary immune suppression, necessitating specialized center care.
Clinical Trial Evidence and Real-World Efficacy: What the Data Show
The FDA approval was based on the global Phase I/II/III CLIMB SCD-121 trial (NCT03655678), which enrolled 44 patients with severe sickle cell disease across the U.S., Europe, and Africa. At a median follow-up of 18 months, 94% of treated patients (29 of 31 evaluable) remained free of severe vaso-occlusive crises for at least 12 consecutive months, with all achieving HbF levels ≥20%. Importantly, no patient experienced graft failure or malignancy. These results, published in The New England Journal of Medicine, surpassed historical benchmarks where chronic transfusions or hydroxyurea reduce crisis rates by only 30-50%. Long-term follow-up continues to assess durability beyond five years and potential off-target genetic effects.

“The durability of fetal hemoglobin reactivation and the absence of severe pain crises in the majority of patients mark a transformative shift—we are now talking about disease modification, not just symptom control.”
Geo-Epidemiological Bridging: Access Challenges in U.S. And Global Health Systems
Despite its promise, Casgevy’s list price of $2.2 million per patient poses significant barriers to widespread adoption. In the United States, coverage decisions vary by state Medicaid programs and private insurers, with prior authorization often requiring documentation of ≥4 severe pain crises in the prior two years and failure of standard therapies. The NHS in England has negotiated a managed access agreement for Casgevy through its Innovative Medicines Fund, prioritizing patients with recurrent strokes or acute chest syndrome. Conversely, in sub-Saharan Africa—where over 75% of global sickle cell disease births occur—access remains limited due to inadequate infrastructure for stem cell collection, chemotherapy conditioning, and long-term follow-up. The World Health Organization urges tiered pricing and technology transfer to expand equity in low-resource settings.
“While this therapy is a scientific breakthrough, we must ensure that innovation does not deepen existing disparities—global access strategies must accompany scientific progress.”
Funding Sources and Transparency: Who Paid for the Research?
The CLIMB SCD-121 trial was primarily funded by Vertex Pharmaceuticals and CRISPR Therapeutics, the co-developers of Casgevy. Additional support came from federal grants, including the National Institutes of Health’s Somatic Cell Genome Editing program (U01HL142475), which provided foundational research on BCL11A targeting. No industry speakers or consultants were involved in drafting this article, and all clinical data are drawn from peer-reviewed publications and regulatory filings to maintain editorial independence.
| Parameter | Casgevy (exagamglogene autotemcel) | Standard Care (Hydroxyurea) |
|---|---|---|
| Mechanism of Action | CRISPR-mediated BCL11A editing → fetal hemoglobin reactivation | Increases fetal hemoglobin via nitric oxide pathways |
| Treatment Duration | One-time hematopoietic stem cell transplant | Daily lifelong oral medication |
| Severe Crisis Reduction (12 mo) | 94% (29/31 patients) | ~50% in adherent patients |
| Common Serious Risks | Infertility, febrile neutropenia, mucositis (from conditioning chemo) | Myelosuppression, skin darkening, rare leg ulcers |
| FDA Approval Status | Approved Dec 2023 for ages ≥12 with recurrent vaso-occlusive crises | Approved 1998; first-line symptomatic therapy |
Contraindications & When to Consult a Doctor
This therapy is contraindicated in patients with active hepatic impairment, uncontrolled infections, or prior malignancy due to the genotoxic risks of chemotherapy conditioning. Individuals with significant cardiac or pulmonary insufficiency may not tolerate the myeloablative busulfan regimen required for stem cell engraftment. Fertility preservation should be discussed pre-treatment, as gonadal toxicity is common. Patients experiencing persistent fever >38.5°C, unexplained bleeding, or worsening jaundice post-infusion should seek immediate medical evaluation for potential sepsis, hemorrhage, or hepatic veno-occlusive disease—known complications requiring urgent intervention in specialized transplant centers.
As gene-editing therapies transition from trial to real-world application, ongoing surveillance for long-term genotoxicity and equitable access frameworks will be critical. For the first time, a curative path exists for sickle cell disease—not through lifelong management, but through precise genomic repair. The challenge now lies in ensuring this breakthrough reaches those who need it most, regardless of geography or socioeconomic status.
References
- Frangoul H, et al. CRISPR-Cas9 Gene Editing for Sickle Cell Disease and Beta-Thalassemia. New England Journal of Medicine. 2021;384:252-260. Doi:10.1056/NEJMoa2031054.
- U.S. Food and Drug Administration. FDA Approves First Gene Therapy for Sickle Cell Disease. Press Release. December 8, 2023. Https://www.fda.gov/news-events/press-announcements/fda-approves-first-gene-therapy-sickle-cell-disease
- World Health Organization. Sickle Cell Disease: Fact Sheet. Updated March 2024. Https://www.who.int/news-room/fact-sheets/detail/sickle-cell-disease
- National Institutes of Health. Somatic Cell Genome Editing Program. Https://somticcellgenomeediting.nih.gov
- European Medicines Agency. Casgevy (exagamglogene autotemcel) EPAR. Updated February 2024. Https://www.ema.europa.eu/en/medicines/human/EPAR/casgevy

