Longevity science is shifting from simply extending lifespan to optimizing “healthspan”—the period of life spent in good health. Driven by breakthroughs in cellular senescence and metabolic regulation, the medical consensus now emphasizes a synergy of pharmacological interventions, rigorous lifestyle modifications, and social connectivity to delay age-related chronic diseases.
The global discourse on longevity has evolved beyond the vanity of “anti-aging” creams into a serious clinical pursuit of biological age deceleration. As healthcare systems worldwide face an unprecedented demographic shift toward an aging population, the goal is no longer just the avoidance of death, but the prevention of the frailty and cognitive decline that typically characterize the final decades of life. This transition is critical because extending life without maintaining function increases the systemic burden on global healthcare infrastructures and diminishes the individual’s quality of life.
In Plain English: The Clinical Takeaway
- Healthspan over Lifespan: The goal is to stay functionally young and disease-free for longer, rather than simply adding years of illness to the end of life.
- Lifestyle is the Foundation: No supplement or drug can override the fundamental biological requirements of deep sleep, nutrient-dense caloric restriction, and strong social bonds.
- Caution with “Bio-hacking”: Many longevity supplements lack large-scale, double-blind placebo-controlled trials (studies where neither the patient nor the doctor knows who is getting the treatment) and should not replace standard medical care.
The Cellular Machinery: Understanding Senescence and Autophagy
To understand longevity, we must examine the “Hallmarks of Aging.” At the center of This represents cellular senescence—a state where cells stop dividing but refuse to die, becoming “zombie cells” that secrete pro-inflammatory cytokines. This process triggers chronic low-grade inflammation, often termed “inflammaging,” which accelerates the degradation of tissues, and organs.
The clinical objective is to promote autophagy, the body’s internal “recycling” mechanism. Autophagy is the metabolic process where a cell breaks down its own damaged components to maintain homeostasis (biological balance). When autophagy fails, cellular debris accumulates, leading to the protein misfolding seen in neurodegenerative diseases like Alzheimer’s and Parkinson’s.
Emerging research into senolytics—a class of drugs designed to selectively induce death in senescent cells—is currently in various clinical trial phases. By clearing these zombie cells, researchers aim to improve the mechanism of action (the specific biochemical interaction through which a drug produces its effect) of the body’s own regenerative processes.
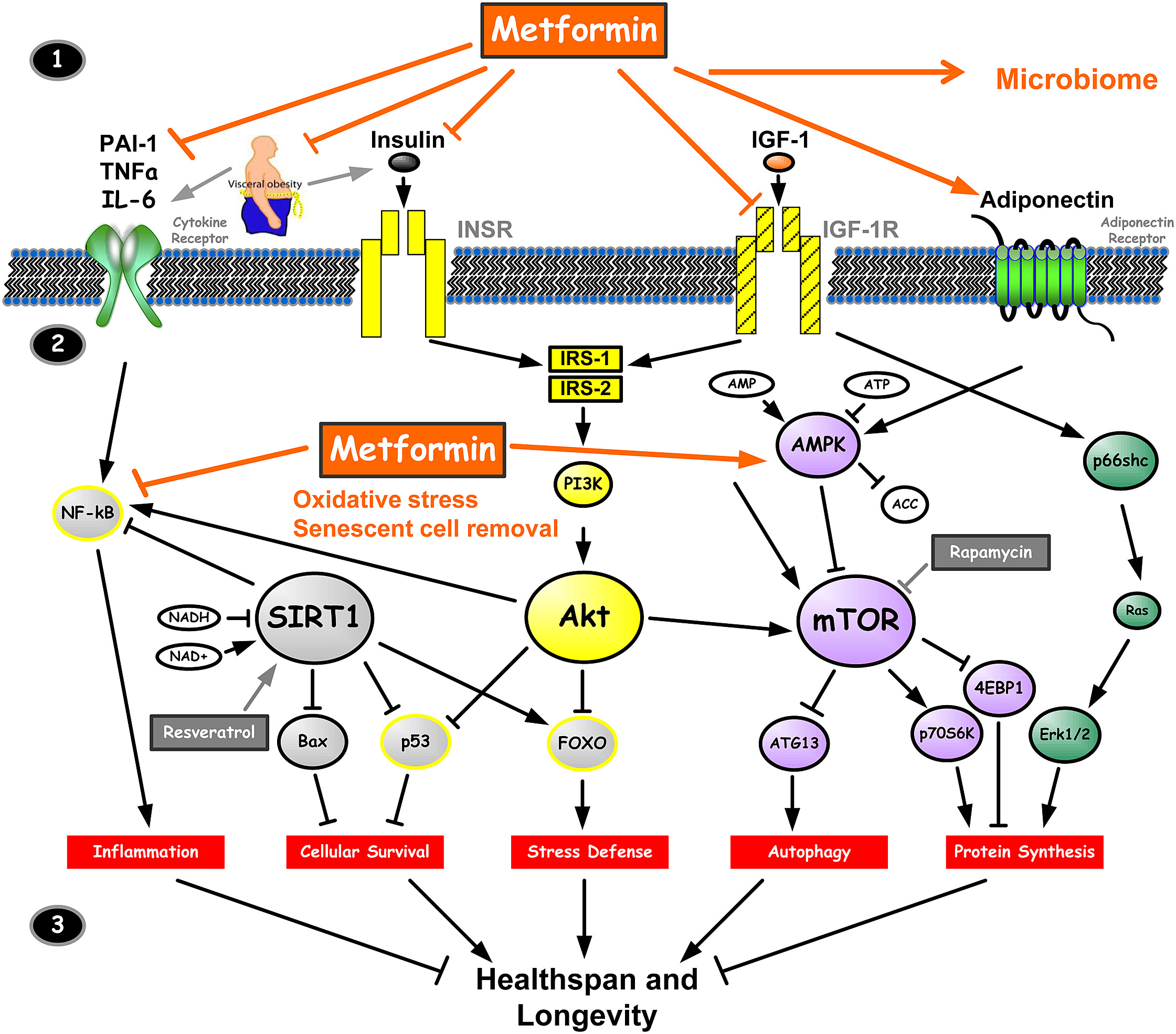
Pharmacological Frontiers: From Metformin to Rapamycin
The current pharmaceutical interest in longevity focuses heavily on metabolic pathways, specifically the mTOR (mammalian target of rapamycin) and AMPK (AMP-activated protein kinase) pathways. These act as the body’s “fuel gauges,” determining whether the cell should grow or enter a state of repair and survival.
Metformin, a first-line medication for Type 2 Diabetes, has gained notoriety in longevity circles due to observational data suggesting a lower incidence of cancer and cardiovascular disease in users. However, the TAME (Targeting Aging with Metformin) trial remains the gold standard for determining if metformin can actually delay the onset of age-related diseases in non-diabetic populations.

Similarly, Rapamycin, an immunosuppressant, is being studied for its ability to inhibit mTOR, thereby mimicking the effects of caloric restriction. While promising, the risk of immunosuppression—reducing the body’s ability to fight infections—makes it a high-risk intervention that requires strict clinical oversight.
“The transition from treating single diseases to treating the biological process of aging itself represents the most significant paradigm shift in medicine since the discovery of antibiotics. We are moving toward a preventative model where the ‘patient’ is anyone currently aging.” — Dr. Nir Barzilai, Director of the Albert Einstein College of Medicine Longevity Research program.
The Geo-Epidemiological Divide and Regulatory Hurdles
The accessibility of longevity interventions varies wildly by region. In the United States, the FDA (Food and Drug Administration) does not currently recognize “aging” as a disease, which complicates the approval process for drugs specifically designed to extend healthspan. This has led to a surge in “off-label” prescriptions, where doctors prescribe approved drugs for non-approved uses.
In Europe, the EMA (European Medicines Agency) maintains a similarly cautious stance, focusing on the treatment of specific geriatric syndromes rather than aging as a whole. Meanwhile, in regions like Japan and Singapore, public health initiatives are more integrated, focusing on “Active Aging” policies that combine urban design with metabolic screening to preserve the elderly integrated into the workforce and community.
Funding for this research is increasingly shifting from government grants to private venture capital and “billionaire-funded” institutes. While this accelerates innovation, it introduces a significant bias toward high-cost, high-tech interventions (like stem cell therapy) over low-cost, scalable public health interventions (like improving nutrition and social connectivity).
Comparing Longevity Intervention Strategies
The following table summarizes the current evidence-based approaches to extending healthspan, categorized by their clinical reliability and risk profile.
| Intervention Type | Primary Mechanism | Evidence Level | Risk Profile |
|---|---|---|---|
| Caloric Restriction | AMPK Activation / Autophagy | High (Cross-species) | Low (Risk of malnutrition) |
| Social Integration | Cortisol Reduction / Oxytocin | High (Epidemiological) | Negligible |
| Metformin (Off-label) | Insulin Sensitivity / mTOR inhibition | Moderate (Observational) | Moderate (GI distress, Lactic acidosis) |
| Rapamycin | mTOR Inhibition | Moderate (Animal models) | High (Immunosuppression) |
| Senolytics | Selective Apoptosis of Senescent Cells | Low/Emerging (Phase I/II) | Unknown (Long-term) |
The Social Determinant: Why Connection is Biological
The mention of living to 140 through socialization is not mere sentimentality; it is rooted in neurobiology. Chronic loneliness triggers a systemic stress response, increasing the production of cortisol, which in turn accelerates telomere attrition (the shortening of the protective caps on the ends of chromosomes). Shortened telomeres are a primary marker of biological aging.

Data from “Blue Zones”—regions where people consistently live past 100—show that social cohesion is as predictive of longevity as diet or exercise. Strong social ties modulate the immune system and reduce the prevalence of systemic inflammation, effectively acting as a biological buffer against the stressors of aging.
Contraindications & When to Consult a Doctor
Longevity protocols are not universal. Certain interventions can be dangerous depending on your medical history:
- Metformin: Contraindicated for individuals with severe renal impairment (kidney failure) or acute heart failure due to the risk of lactic acidosis.
- mTOR Inhibitors (Rapamycin): Should be avoided by those with compromised immune systems or active infections, as they can suppress the body’s natural defense mechanisms.
- Extreme Fasting: Contraindicated for individuals with a history of eating disorders, Type 1 Diabetes, or those who are underweight (BMI < 18.5).
Consult a physician immediately if you experience: Unexplained rapid weight loss, sudden onset of cognitive confusion, or persistent fatigue after starting a new longevity supplement or dietary regimen. Never alter prescription dosages to “mimic” longevity trials without a licensed provider’s supervision.
The future of longevity lies not in a single “miracle pill,” but in the precision application of biology. By combining the molecular precision of senolytics and metabolic regulators with the proven efficacy of social and lifestyle interventions, we are moving toward an era where the final years of life are defined by vitality rather than decay.

