Researchers have identified inconsistent endpoints across clinical trials for metabolic dysfunction-associated steatohepatitis (MASH) with cirrhosis, creating challenges in comparing treatment efficacy and delaying regulatory approvals for therapies targeting this advanced form of fatty liver disease, which affects an estimated 5-7% of adults globally and is rising fastest in regions with high rates of obesity and type 2 diabetes.
Why Endpoint Inconsistency Hinders Progress in MASH Cirrhosis Treatment
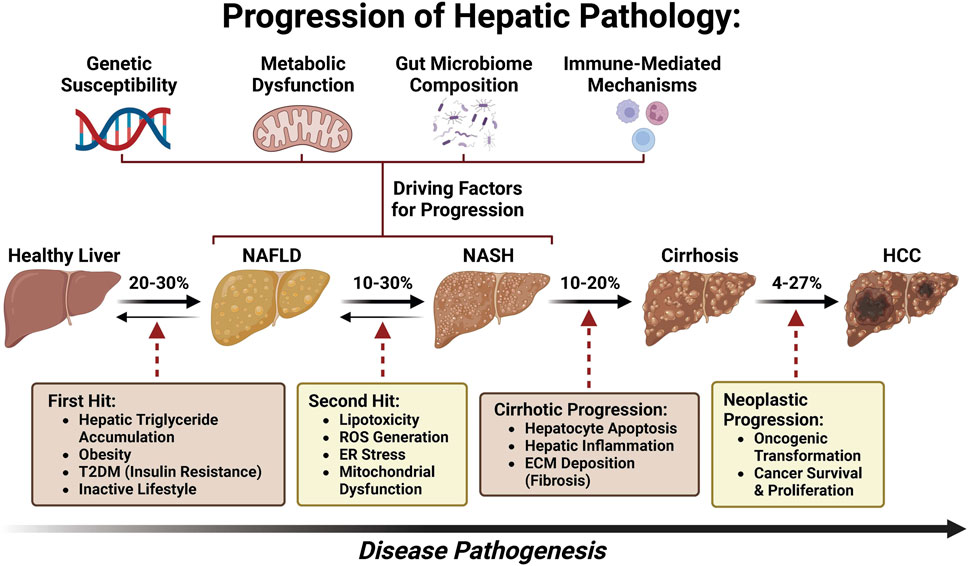
The lack of standardized endpoints in MASH cirrhosis trials complicates cross-study comparisons, as some trials prioritize histologic improvement whereas others focus on clinical outcomes like mortality or liver transplantation. This fragmentation impedes meta-analyses and prolongs the timeline for regulatory agencies such as the FDA and EMA to evaluate recent therapies. Given that MASH cirrhosis represents the most severe stage of metabolic dysfunction-associated fatty liver disease (MAFLD), where liver scarring progresses to irreversible fibrosis and portal hypertension, establishing universally accepted endpoints is critical for accelerating access to effective treatments.
In Plain English: The Clinical Takeaway
- MASH is a serious liver condition caused by fat buildup linked to obesity and diabetes, which can lead to cirrhosis and liver failure if untreated.
- Without agreed-upon ways to measure treatment success, it’s harder to compare new medicines and bring them to patients faster.
- Patients should focus on managing metabolic health through weight control, exercise, and diabetes care, as no therapy is yet approved specifically for MASH cirrhosis.
Clinical Trial Design Gaps and Regulatory Implications
Current Phase II and III trials for MASH cirrhosis investigate various mechanisms, including farnesoid X receptor (FXR) agonists, peroxisome proliferator-activated receptor (PPAR) agonists, and antibody-based therapies targeting inflammatory pathways. Still, endpoint heterogeneity persists: some studies use the NAFLD Activity Score (NAS) or fibrosis staging via biopsy, while others rely on clinical endpoints such as hepatic decompensation events. This divergence complicates regulatory decision-making, as agencies require consistent, clinically meaningful outcomes to assess benefit-risk profiles. The American Association for the Study of Liver Diseases (AASLD) has advocated for composite endpoints combining histologic and clinical measures, but adoption remains uneven across industry-sponsored trials.
Geoeconomic Impact on Patient Access
In the United States, where an estimated 24 million adults have MASH and 2-3% progress to cirrhosis, the FDA’s cautious approach due to endpoint variability may delay approvals, disproportionately affecting underserved populations in states with limited hepatology specialists. In Europe, the EMA has encouraged adaptive trial designs but faces similar fragmentation. In contrast, the NHS in the UK has prioritized early detection through liver fibrosis screening in diabetes clinics, yet treatment access remains constrained without approved therapies. Bridging this gap requires harmonized endpoints that reflect real-world outcomes meaningful to patients and payers alike.

Funding Sources and Potential Bias
Much of the ongoing research into MASH cirrhosis therapeutics is funded by pharmaceutical companies developing investigational agents, raising concerns about outcome selection bias. For example, trials sponsored by firms developing FXR agonists may prioritize histologic endpoints that favor their mechanism, while those testing PPAR agonists might emphasize metabolic improvements. Independent funding from institutions like the National Institutes of Health (NIH) or the European Union’s Horizon Europe program is essential to generate unbiased data. Transparency in funding disclosures, as mandated by the International Committee of Medical Journal Editors (ICMJE), helps mitigate perceived conflicts of interest.

Expert Perspectives on Standardization Efforts
“Without consensus on what constitutes a meaningful clinical benefit in MASH cirrhosis, we risk approving therapies based on surrogate markers that don’t translate to improved survival or quality of life.”
“Regulatory science must evolve to accept clinically relevant endpoints that reflect patient-centered outcomes, especially in chronic liver diseases where histologic improvement alone may not suffice.”
Key Trial Design Comparisons: Endpoint Heterogeneity in MASH Cirrhosis Studies
| Trial Focus | Primary Endpoint Type | Example Agents | Regulatory Feedback |
|---|---|---|---|
| Histologic Improvement | ≥1-stage fibrosis improvement without NASH worsening | FXR agonists (e.g., obeticholic acid) | FDA: Requires confirmation with clinical outcomes |
| Metabolic Response | Reduction in liver fat + ALT normalization | PPAR agonists (e.g., lanifibranor) | EMA: Encourages inclusion of histologic data |
| Clinical Outcomes | Time to first hepatic decompensation event | Anti-inflammatory monoclonal antibodies | FDA/EMA: Acceptable if statistically powered |
Contraindications & When to Consult a Doctor
Patients with suspected or confirmed MASH cirrhosis should avoid alcohol entirely, as even minimal consumption accelerates fibrosis progression. Those with decompensated cirrhosis (evidenced by ascites, variceal bleeding, or hepatic encephalopathy) are typically excluded from clinical trials and require urgent specialist care. Individuals experiencing unexplained fatigue, abdominal swelling, or jaundice should seek immediate medical evaluation, as these may indicate liver failure. Routine monitoring via FibroScan, serum biomarkers (e.g., FIB-4, ELF test), and ultrasound is recommended for high-risk patients with obesity, type 2 diabetes, or metabolic syndrome.

While lifestyle intervention remains the cornerstone of MASH management, ongoing efforts to standardize trial endpoints offer hope for faster development of effective therapies. Until then, multidisciplinary care involving hepatologists, endocrinologists, and dietitians provides the best opportunity to slow disease progression and improve long-term outcomes in this growing public health challenge.
References
- Sanyal AJ, et al. Obeticholic acid for the treatment of non-alcoholic steatohepatitis: interim analysis from a multicenter, randomized, placebo-controlled trial. Lancet. 2019;394(10208):1653-1662.
- Loomba R, et al. Lanifibranor in non-alcoholic steatohepatitis: a randomized, placebo-controlled phase 2b trial. Nat Med. 2021;27(5):849-857.
- Friedman SL, et al. Mechanisms of NAFLD progression and therapeutic strategies. Cell Metab. 2021;33(4):683-701.
- Younossi ZM, et al. Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. J Hepatol. 2023;78(6):1159-1172.
- FDA Guidance for Industry: Noncirrhotic Non-alcoholic Steatohepatitis With Liver Fibrosis: Developing Drugs for Treatment. 2023.