Two years after a significant measles outbreak in central Ohio, vaccination rates in the affected region remain substantially below the herd immunity threshold of 95%, leaving communities vulnerable to preventable resurgence, according to pediatric infectious disease specialists presenting data at the Pediatric Academic Societies annual meeting. Despite effective outbreak containment, sustained improvements in immunization coverage have not materialized, highlighting gaps in long-term public health intervention strategies. This persistent deficit underscores the critical need for ongoing vaccine education, accessible primary care integration, and targeted outreach to address vaccine hesitancy rooted in misinformation and access barriers.
Understanding Measles and the MMR Vaccine Mechanism
Measles is a highly contagious viral infection caused by the measles virus, a member of the Paramyxoviridae family, which spreads through respiratory droplets and can remain infectious in the air for up to two hours. The virus infects immune cells in the respiratory tract, then disseminates systemically, causing characteristic symptoms like high fever, cough, coryza, conjunctivitis, and a maculopapular rash. The measles, mumps, and rubella (MMR) vaccine contains live, attenuated strains of these viruses that stimulate a protective immune response without causing disease in healthy individuals. Upon vaccination, the attenuated virus triggers both humoral and cell-mediated immunity: B cells produce neutralizing antibodies targeting the viral hemagglutinin protein, while cytotoxic T cells eliminate infected cells, establishing immunological memory that prevents future infection upon exposure.
In Plain English: The Clinical Takeaway
- The MMR vaccine is safe and highly effective, preventing measles in about 97% of people after two doses, with protection lasting decades or a lifetime for most.
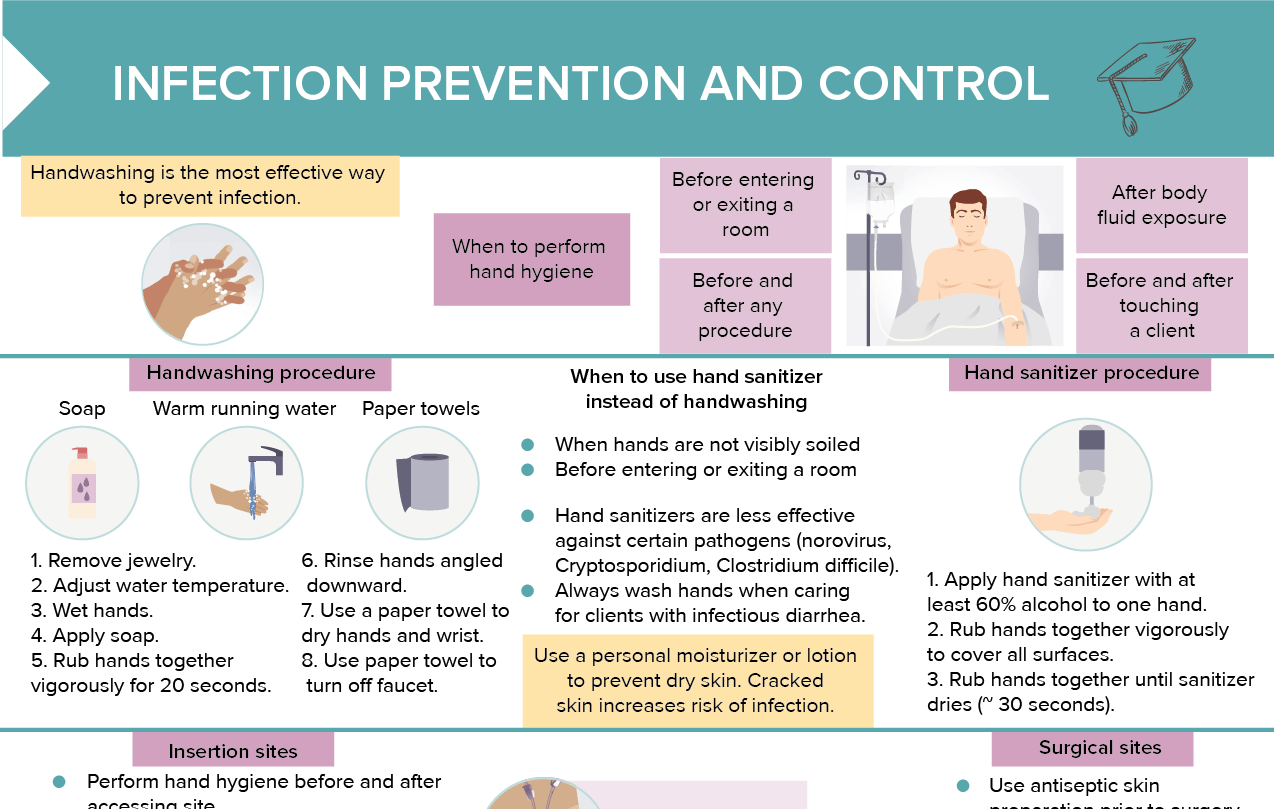
Clinical Control Prevention - Low vaccination rates don’t just risk individual illness—they threaten community immunity, putting infants too young to vaccinate and immunocompromised individuals at grave risk.
- Vaccine hesitancy often stems from misinformation; talking openly with trusted healthcare providers can aid families make informed, evidence-based decisions.
Epidemiological Gaps and Regional Health System Impact
The Ohio outbreak, which began in late 2022 and peaked in early 2023, ultimately infected 85 individuals, primarily unvaccinated children, according to the Ohio Department of Health. While aggressive public health response—including isolation, contact tracing, and targeted vaccination clinics—contained transmission within months, subsequent data from the Centers for Disease Control and Prevention (CDC) show that as of early 2026, MMR vaccination coverage in the affected counties remains at approximately 82%, well below the 95% threshold needed to prevent sustained transmission. This gap is particularly concerning in Franklin County, where pediatric clinic data reveal persistent delays in well-child visits and vaccine administration, exacerbated by provider shortages and transportation barriers in rural areas. In contrast, states with stronger school immunization enforcement and integrated immunization information systems, such as Massachusetts and Connecticut, have maintained MMR coverage above 94% despite national trends of declining trust in vaccines.

These disparities reflect broader challenges in the U.S. Public health infrastructure. The Centers for Medicare & Medicaid Services (CMS) reimbursement model often underfunds vaccine counseling and outreach, while state-level variation in vaccine exemption policies creates uneven protection. The Advisory Committee on Immunization Practices (ACIP) recommends routine MMR vaccination at 12–15 months and again at 4–6 years, but implementation depends heavily on state Medicaid policies and local health department capacity. Unlike the UK’s National Health Service (NHS), which uses a centralized call-and-recall system for childhood vaccinations, the U.S. Relies on fragmented private and public systems, increasing the likelihood of missed opportunities.
Funding Sources and Research Transparency
The data presented at the Pediatric Academic Societies meeting were derived from an ongoing observational study conducted by researchers at Boston Children’s Hospital and Harvard Medical School, led by Dr. Rosemary A. Martoma. The study was funded by the National Institutes of Health (NIH) through the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) under grant R01 HD105501, with additional support from the Harvard Clinical and Translational Science Center. No pharmaceutical companies were involved in funding or data interpretation, minimizing potential conflicts of interest. The research leveraged electronic health record data from multiple urban pediatric networks to assess vaccination trends before, during, and after the outbreak, adjusting for socioeconomic status, race, and geographic mobility.
Expert Perspectives on Vaccine Confidence and Access
“We’ve seen that outbreak response alone doesn’t fix the underlying issues—whether it’s distrust in medical institutions, logistical hurdles for working families, or persistent myths about vaccine safety. Real progress requires investing in community health workers and embedding vaccine conversations into every well-child visit.”
“When vaccination rates dip below herd immunity, it’s not a failure of individual choice—it’s a system failure. We need policies that make vaccination the easy, default choice: school requirements without loopholes, reminders in electronic health records, and equitable access regardless of zip code.”
Contraindications & When to Consult a Doctor
The MMR vaccine is contraindicated in individuals with a history of severe allergic reaction (e.g., anaphylaxis) to a previous dose or any vaccine component, such as gelatin or neomycin. It should also be avoided in people with severe immunodeficiency, including those undergoing chemotherapy, with congenital immunodeficiencies, or advanced HIV/AIDS with low CD4 counts. Pregnant individuals should not receive the MMR vaccine due to theoretical risk to the fetus, though vaccination is recommended before conception or immediately postpartum. Moderate or severe acute illness is a precaution—not a contraindication—but vaccination should be delayed until recovery. Anyone experiencing symptoms suggestive of measles—high fever, cough, runny nose, red eyes, or a spreading rash—should seek medical evaluation immediately, particularly if unvaccinated or exposed to an known case, as early diagnosis supports isolation and prevents further spread.

| Metric | Value | Source |
|---|---|---|
| Herd immunity threshold for measles | 95% vaccination coverage | CDC |
| MMR vaccine effectiveness (2 doses) | 97% prevention of measles | CDC |
| Ohio outbreak cases (2022–2023) | 85 confirmed | Ohio Department of Health |
| Post-outbreak MMR coverage in affected Ohio counties (2026) | ~82% | CDC Immunization Data |
| National average MMR coverage (2025) | 90.8% | CDC National Immunization Survey |
Moving Forward: Systems-Level Solutions
Addressing stagnant vaccination rates requires more than public service announcements—it demands structural reform. Successful models include Vermont’s elimination of non-medical vaccine exemptions and Louisiana’s use of immunization information systems to trigger automated reminders for overdue vaccinations. Integrating vaccine status checks into electronic health records with clinical decision support, as piloted in Kaiser Permanente networks, has improved timely completion by up to 20%. Globally, the World Health Organization’s Immunization Agenda 2030 emphasizes equity, calling for reduced disparities in vaccine access by 2030. In the U.S., reauthorizing and expanding the Section 317 Immunization Program could provide critical funding to under-resourced health departments tasked with outreach and vaccine procurement. Rebuilding trust means meeting communities where they are—through faith-based organizations, schools, and trusted local leaders—while upholding the scientific consensus that vaccines are among the safest and most effective public health tools ever developed.
References
- Centers for Disease Control and Prevention. Measles (Rubeola) Vaccination. CDC.gov. Updated 2025.
- Martoma RA, et al. Post-outbreak vaccination trends in pediatric populations. Presented at: Pediatric Academic Societies Annual Meeting; 2026 May; Baltimore, MD.
- O’Leary ST, et al. Vaccine hesitancy and refusal in the United States. Acad Pediatr. 2024;24(3):456-465.
- Ohio Department of Health. Measles Outbreak Summary Report. 2023.
- World Health Organization. Immunization Agenda 2030: A Global Strategy to Exit No One Behind. WHO.int. 2020.

