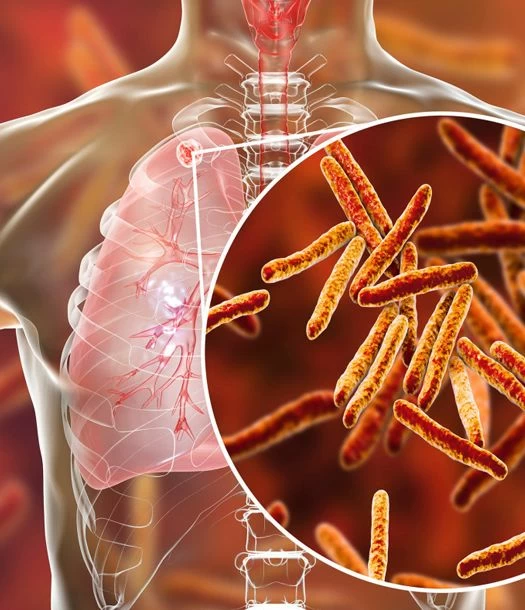
Researchers have identified a specific molecular mechanism—the “invisibility button”—that allows Mycobacterium tuberculosis to evade the human immune system. By suppressing the host’s macrophage response, the bacteria persist in a latent state, complicating treatment and necessitating new therapeutic targets to unmask the infection and accelerate clearance.
For decades, the medical community has struggled with the paradox of tuberculosis (TB): why a pathogen that is theoretically susceptible to antibiotics can linger in the human body for a lifetime. The discovery of this “invisibility” mechanism explains the biological loophole that allows TB to transition from an active infection to a latent state, where it remains dormant and undetectable by the body’s primary defense systems.
This is not merely a laboratory curiosity; We see a public health imperative. With millions of people globally harboring latent TB, the ability to “flip the switch” and produce these bacteria visible to the immune system could revolutionize how we prevent the reactivation of the disease, particularly in immunocompromised populations.
In Plain English: The Clinical Takeaway
- The Hideout: TB bacteria don’t just hide in the lungs; they hide inside the incredibly immune cells (macrophages) that are supposed to destroy them.
- The “Button”: The bacteria utilize a specific molecular signal to stop the immune cell from “digesting” them, effectively becoming invisible to the body’s alarm system.
- The Goal: New drugs are being developed to block this signal, forcing the bacteria out of hiding so the immune system and antibiotics can kill them faster.
The Molecular Mechanism of Immune Evasion
The core of this discovery lies in the mechanism of action—the specific biochemical process through which a drug or pathogen produces its effect—of the Mycobacterium tuberculosis (MTB) cell wall. Specifically, the bacteria manipulate the process of phagosome-lysosome fusion. In a healthy immune response, a macrophage engulfs a pathogen into a vesicle called a phagosome, which then fuses with a lysosome (a sac of digestive enzymes) to destroy the invader.
The “invisibility button” refers to the secretion of specific proteins and lipids that inhibit this fusion. By preventing the lysosome from attaching, the MTB bacterium transforms the phagosome into a protected sanctuary. In this state, the bacteria avoid exposure to the acidic environment and oxidative burst—the chemical weapons the body uses to shred bacterial membranes.
This cellular hijacking allows the bacteria to enter a state of metabolic dormancy. As most current antibiotics target active cell division (the process of a bacterium splitting into two), these dormant “invisible” bacteria are naturally resistant to standard treatment, necessitating the grueling six-to-nine-month drug regimens currently prescribed by the World Health Organization (WHO).
Global Epidemiological Impact and Regulatory Pathways
The implications of this discovery vary significantly by geography. In high-burden regions across Southeast Asia and Sub-Saharan Africa, the prevalence of Multi-Drug Resistant TB (MDR-TB) makes the discovery of “unmasking” agents a critical priority. If a therapeutic can strip the bacteria of its invisibility, it may restore the efficacy of existing antibiotics that previously failed due to the bacteria’s dormant state.
From a regulatory standpoint, the transition from this discovery to clinical application will require rigorous double-blind placebo-controlled trials—studies where neither the patient nor the doctor knows who is receiving the treatment to eliminate bias. The U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) will likely categorize these “unmasking agents” as adjunct therapies, meant to be used alongside traditional rifampicin and isoniazid treatments.
Funding for this research has been predominantly driven by public-private partnerships, including grants from the National Institutes of Health (NIH) and the Bill & Melinda Gates Foundation. This funding structure ensures that the resulting intellectual property remains accessible to low-income countries, avoiding the “pricing-out” effect often seen with orphan drugs.
“The ability to disrupt the intracellular sanctuary of Mycobacterium tuberculosis represents a paradigm shift. We are moving from simply trying to kill the bacteria to first forcing them to reveal themselves to the host immune system.”
Comparative Analysis: Latent vs. Active Tuberculosis
To understand why the “invisibility button” is so dangerous, one must distinguish between the two primary states of TB infection.
| Feature | Latent TB Infection (LTBI) | Active TB Disease |
|---|---|---|
| Immune Status | Bacteria “Invisible” / Contained | Bacteria Active / Uncontrolled |
| Symptom Profile | Asymptomatic | Cough, Fever, Night Sweats |
| Infectivity | Non-infectious | Highly infectious via aerosols |
| Treatment Goal | Prevent reactivation | Eliminate active pathogen |
| Diagnostic Marker | Positive TST/IGRA blood test | Sputum culture/Chest X-ray |
Contraindications & When to Consult a Doctor
While the discovery of the “invisibility button” promises future therapies, current patients must remain vigilant. It is critical to note that any attempt to “stimulate” the immune system without medical supervision can be dangerous. In patients with latent TB, an uncontrolled immune response can lead to hypersensitivity pneumonitis—an inflammation of the lung tissue caused by an overreaction to the bacteria.
Contraindications: Individuals with severe autoimmune disorders or those taking high-dose corticosteroids should exercise extreme caution with any new immunomodulatory TB therapies, as these can either suppress the necessary response or trigger a cytokine storm.
You should consult a healthcare provider immediately if you experience the following “red flag” symptoms, which suggest the bacteria have “flipped the switch” from latent to active:
- A persistent cough lasting more than three weeks, especially if producing blood.
- Unexplained weight loss and loss of appetite.
- Drenching night sweats and low-grade evening fevers.
- Chest pain during breathing or coughing.
The Path Forward: Precision Pulmonology
The identification of this evasion mechanism moves us toward “Precision Pulmonology.” Instead of a one-size-fits-all antibiotic approach, future clinicians may first test a patient’s specific bacterial strain to see which “invisibility” proteins are being expressed. By tailoring the treatment to unmask the specific strain of MTB, One can potentially reduce treatment duration from months to weeks, significantly increasing patient compliance and reducing the emergence of drug-resistant strains.
As we move through 2026, the focus shifts to Phase II clinical trials. The goal is no longer just survival, but the complete eradication of the latent reservoir, finally bringing the world closer to the Lancet-documented goals of global TB elimination.
References
- World Health Organization (WHO). Global Tuberculosis Report 2025/2026.
- The Lancet Infectious Diseases. “Intracellular Survival Mechanisms of M. Tuberculosis.”
- PubMed Central (PMC). “Phagosome-Lysosome Fusion Inhibition in Mycobacterial Infections.”
- Centers for Disease Control and Prevention (CDC). “Latent TB Infection Guidelines.”