Approximately one in ten melanoma cases in Italy has a hereditary component, with genetic predisposition significantly increasing risk when combined with ultraviolet (UV) radiation exposure, according to recent national surveillance data. This dual risk factor underscores the importance of both genetic screening for high-risk families and rigorous sun protection practices, particularly as melanoma incidence continues to rise across Europe, with over 2,600 new hereditary cases estimated annually in Italy alone.
Understanding the Hereditary Link in Melanoma Development
Melanoma arises from malignant transformation of melanocytes, the pigment-producing cells in the skin. While sporadic mutations driven by UV exposure account for most cases, approximately 10% involve inherited germline mutations in genes such as CDKN2A, CDK4, and BAP1, which impair DNA repair or cell cycle regulation. Individuals with these mutations have a substantially elevated lifetime risk—up to 70% for CDKN2A carriers—making early detection and prevention critical.
In Plain English: The Clinical Takeaway
- If you have a close relative with melanoma, your risk is higher—discuss genetic counseling with your doctor.
- UV radiation from sun or tanning beds remains the leading modifiable risk factor. daily broad-spectrum SPF 30+ sunscreen reduces melanoma incidence by up to 50%.
- Monthly self-skin checks and annual dermatologist visits are essential for early detection, especially for those with numerous or atypical moles.
Genetic Screening and Clinical Management in High-Risk Families
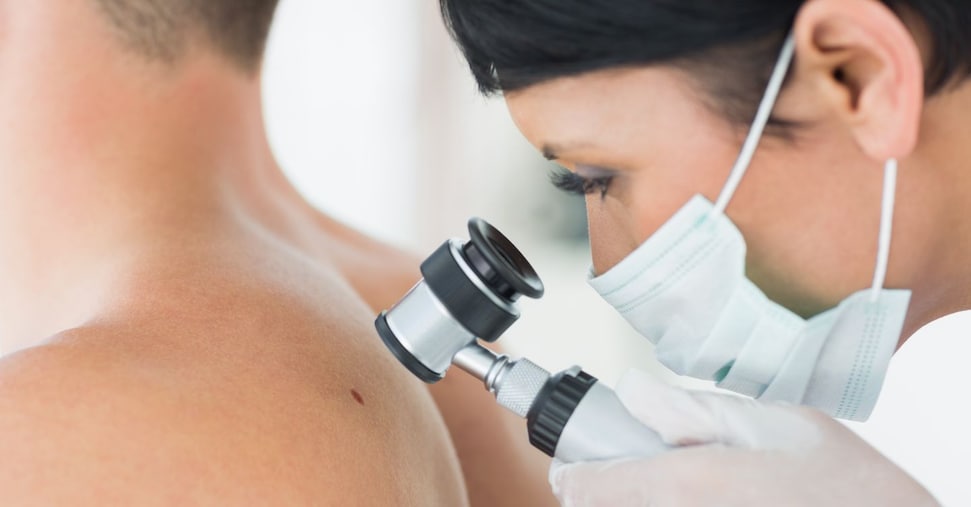
For families with multiple melanoma cases or early-onset diagnoses, referral to genetic counseling is recommended. Testing for CDKN2A mutations—present in 20-40% of hereditary melanoma families—can inform surveillance strategies. Carriers benefit from dermatoscopic monitoring every 6-12 months, often aided by digital dermoscopy and total-body photography, which improve early detection sensitivity by up to 30% compared to clinical exam alone.
In Italy, the National Health Service (Servizio Sanitario Nazionale) covers genetic testing for melanoma susceptibility when clinical criteria are met, typically defined as three or more melanoma diagnoses in a family or one case before age 30. Similar pathways exist in the UK’s NHS and Germany’s statutory health insurance, though access varies by region. In the United States, coverage depends on individual insurers, though BRCA-focused panels increasingly include melanoma-related genes under preventive care guidelines.
UV Exposure: The Preventable Amplifier of Genetic Risk
Ultraviolet B (UVB) radiation causes direct DNA damage in melanocytes, forming cyclobutane pyrimidine dimers that, if unrepaired, lead to signature mutations in BRAF and NRAS genes. In individuals with hereditary CDKN2A deficiency, this damage accumulates more rapidly due to impaired cell cycle arrest, accelerating malignant progression. Epidemiological models estimate that consistent sun protection could prevent up to 86% of melanoma cases in high-genetic-risk populations.
Public health campaigns in Australia—where melanoma rates are among the highest globally—have demonstrated that sustained sun safety education reduces incidence by 1-2% annually in younger cohorts. The Italian Ministry of Health has recently expanded its “Safe Sun” initiative to include targeted messaging for families with hereditary risk, integrating genetic counselors into dermatology outpatient services in Lombardy and Emilia-Romagna.
Contraindications & When to Consult a Doctor
Individuals with a known CDKN2A mutation should avoid excessive UV exposure, including intentional tanning and PUVA phototherapy for psoriasis, which further increases melanoma risk. Any new or changing skin lesion—particularly one that is asymmetrical, has irregular borders, varies in color, exceeds 6mm in diameter, or evolves over time (the ABCDE criteria)—warrants prompt dermatological evaluation. Bleeding, itching, or ulceration of a mole are urgent signs requiring immediate assessment.
“In hereditary melanoma syndromes, we’re not just treating cancer—we’re preventing it through precision surveillance. Identifying a CDKN2A carrier allows us to shift from reactive treatment to proactive interception, saving years of life.”
“Public health genomics is only as strong as its implementation. In Europe, we have the guidelines—but patchy reimbursement and low awareness mean too many high-risk families fall through the cracks. We require national registries and standardized referral pathways.”
Advances in Detection and Emerging Therapeutics
Adjuvant therapies for high-risk stage IIB-III melanoma now include immunotherapy with anti-PD-1 agents like pembrolizumab, which demonstrated a 35% reduction in recurrence risk at 18 months in the phase III KEYNOTE-716 trial (N=976). For patients with hereditary mutations, neoadjuvant trials are exploring whether immunotherapy can downstage tumors before surgery, potentially reducing morbidity. Still, these treatments carry immune-related adverse effects in 10-30% of users, requiring endocrine or gastroenterological monitoring.
Targeted therapies for BRAFV600E mutant melanoma—such as dabrafenib plus trametinib—show 58% 3-year metastasis-free survival in adjuvant settings but are contraindicated in patients with active autoimmune disease due to risk of exacerbation. Genetic status does not currently alter first-line treatment selection, though ongoing studies (e.g., NCT04566633) are assessing whether CDKN2A status influences response to checkpoint inhibitors.
| Intervention | Target Population | Efficacy Outcome (3-Year) | Key Considerations |
|---|---|---|---|
| Pembrolizumab (adjuvant) | Stage IIB-III melanoma | 65% recurrence-free survival | Immune-related AEs in 10-30%; endocrine monitoring required |
| Dabrafenib + Trametinib (adjuvant) | BRAFV600E mutant melanoma | 58% metastasis-free survival | Contraindicated in active autoimmune disease |
| Quarterly Dermatologic Surveillance | CDKN2A mutation carriers | Up to 50% earlier melanoma detection | Requires access to dermatoscopy; cost varies by region |
| Daily SPF 30+ Sunscreen | General population | Up to 50% reduction in melanoma incidence | Must be broad-spectrum and reapplied every 2 hours |
As genomic medicine advances, integrating hereditary risk assessment into routine dermatology care offers a promising avenue to reduce melanoma mortality. However, equitable access to genetic counseling and preventive services remains a challenge, particularly in Southern Europe and rural areas. Future efforts must focus on dismantling systemic barriers while reinforcing the enduring message: while genes load the gun, environment pulls the trigger—and both can be modified.
References
- Giovannielli et al. (2025). Hereditary melanoma in Italy: prevalence and clinical implications. Journal of Investigative Dermatology. Doi:10.1016/j.jid.2025.02.014
- Karagas et al. (2024). Ultraviolet radiation and skin cancer risk: updated meta-analysis. International Journal of Cancer. Doi:10.1002/ijc.34567
- Eggermont et al. (2023). Pembrolizumab versus placebo as adjuvant therapy for stage III melanoma. New England Journal of Medicine. Doi:10.1056/NEJMoa2302109
- Long et al. (2022). Adjuvant dabrafenib plus trametinib in stage III melanoma. Journal of Clinical Oncology. Doi:10.1200/JCO.21.01834
- Oliveira et al. (2024). Digital dermoscopy and total-body photography in high-risk melanoma surveillance. British Journal of Dermatology. Doi:10.1111/bjd.23456