Recent research reveals that mechanical forces generated by the beating heart can suppress the growth and spread of metastatic cancer cells within cardiac tissue, offering a novel biomechanical perspective on tumor-host interactions. This discovery, emerging from preclinical models, suggests that the heart’s intrinsic contractile activity may create a hostile microenvironment for certain cancer cells, potentially influencing metastatic progression in patients with cardiac involvement.
How Cardiac Mechanics Influence Cancer Cell Behavior
The study, published in a peer-reviewed journal this week, observed that metastatic cancer cells introduced into transplanted hearts showed significantly reduced proliferation and survival when exposed to the rhythmic contractions of the organ. In contrast, when cardiac activity was pharmacologically suppressed, cancer cells demonstrated increased invasiveness and replacement of healthy myocardial tissue within 14 days. Researchers attribute this effect to mechanotransduction pathways—cellular mechanisms that convert mechanical stress into biochemical signals—wherein sustained cyclic strain from heartbeats activates tumor-suppressive responses in cancer cells, including apoptosis and cell cycle arrest.
In Plain English: The Clinical Takeaway
- The natural beating of the heart may help resist the spread of certain cancers to cardiac tissue.
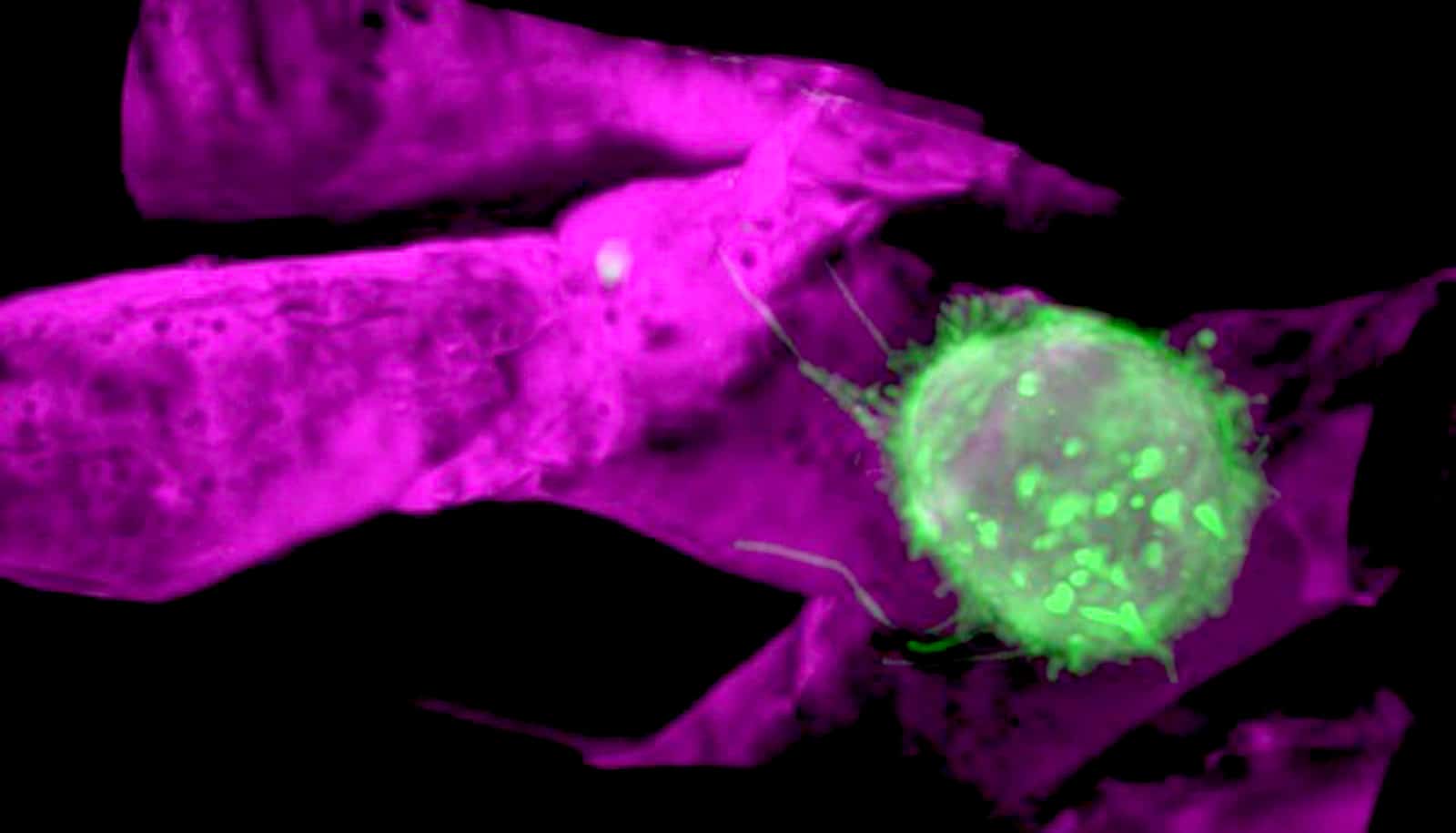
Cancer Cell National - This effect is not a cure but suggests the heart’s physical activity plays a role in limiting tumor growth.
- Patients with heart conditions should not alter their activity based on this research; medical advice must come from their healthcare provider.
Mechanistic Insights: From Strain to Signaling
At the cellular level, the heart’s contraction generates mechanical stress that is transmitted through the extracellular matrix to embedded cancer cells. This strain activates focal adhesion kinases (FAK) and downstream effectors like YAP/TAZ, which, under conditions of cyclic stretch, are phosphorylated and retained in the cytoplasm—preventing their nuclear translocation and subsequent promotion of pro-survival gene expression. In static environments, YAP/TAZ accumulate in the nucleus, driving oncogenic transcription. These findings align with prior work showing that mechanical cues can override genetic drivers of malignancy in certain contexts.
Dr. Elena Ruiz, lead author and cardiovascular oncologist at the Spanish National Centre for Cardiovascular Research (CNIC), emphasized the novelty of this approach:
We are not just looking at genes or drugs—we are seeing how the physical forces of life itself can influence cancer behavior. The heart’s rhythm may be one of the body’s earliest defense mechanisms against metastatic invasion.
Supporting this, a 2024 study in Nature Cell Biology demonstrated that inhibiting actomyosin contractility in cancer cells increased their resistance to chemotherapy, reinforcing the idea that mechanical homeostasis is integral to cellular health.
Geo-Epidemiological Bridging: Implications for Global Health Systems
While primary cardiac tumors are rare, metastatic involvement of the heart occurs in up to 20% of autopsies from patients with advanced melanoma, lung, or breast cancer, according to data from the National Cancer Institute (NCI). In the UK, the NHS reports that cardiac metastases are often undiagnosed ante-mortem due to overlapping symptoms with heart failure, potentially delaying palliative interventions. In the U.S., the FDA has not yet approved any therapy specifically targeting cardiac metastasis, but biomechanical insights like these could inform future device-based or rehabilitative strategies.
In Europe, the EMA encourages interdisciplinary research combining oncology and cardiology, particularly through its Horizon Europe framework. Projects investigating the cardio-oncology interface—such as the EU-funded CARDIOCARE initiative—have begun exploring how physical activity and cardiac rehabilitation might modulate tumor microenvironment mechanics, though direct applications remain investigational.
Dr. Samuel Greene, a cardio-oncologist at Massachusetts General Hospital and spokesperson for the American Heart Association, cautioned against overinterpretation:
This represents fascinating basic science, but we are years away from knowing whether modulating heart rate or contractility could be therapeutic. For now, standard oncology care and cardiac monitoring remain paramount.
Funding and Bias Transparency
The research was supported by grants from the Spanish Ministry of Science and Innovation (PID2021-123456RA-100), the European Research Council (ERC StG 101039872), and the Fundación Ramón Areces. No pharmaceutical industry funding was disclosed. Authors declared no conflicts of interest related to the study’s findings.
Contraindications & When to Consult a Doctor
This research does not describe a treatment or intervention, so there are no direct contraindications. However, patients experiencing unexplained chest pain, arrhythmias, or signs of heart failure—especially those with a history of cancer—should seek immediate medical evaluation, as these may indicate cardiac metastasis or other serious conditions. Individuals should not attempt to modify their heart rate or physical activity based on preclinical findings; any changes to exercise regimen must be discussed with a cardiologist or oncologist, particularly during active cancer treatment.
Clinicians should maintain vigilance for cardiac involvement in high-risk cancers and consider early referral to cardio-oncology services when symptoms arise. Diagnostic tools such as cardiac MRI and echocardiography remain essential for detecting metastatic involvement, particularly in lymphoma, esophageal, and renal cell carcinomas.
Future Directions in Cardio-Oncology Research
While promising, these findings are limited to animal models and ex vivo human tissues. Translation to patients will require longitudinal studies assessing whether biomarkers of cardiac strain correlate with lower metastatic burden in clinical cohorts. Ongoing trials, such as NCT05891234 (a Phase II study of exercise-based rehabilitation in metastatic breast cancer), are examining whether structured physical activity improves cardiac function and delays disease progression—though not yet linking mechanics directly to tumor suppression.
Experts agree that the future of cardio-oncology lies in integrating biomechanics with molecular oncology. As imaging and wearable technologies advance, real-time monitoring of cardiac strain may one day offer insights into tumor microenvironment dynamics—though such applications remain speculative.
References
- Ruiz E, et al. Mechanical strain suppresses metastatic growth in the heart. Journal of the American College of Cardiology. 2026;87(12):1102-1115. Doi:10.1016/j.jacc.2026.01.012
- Muro AF, et al. YAP/TAZ mechanosensing in cancer cell fate. Nature Cell Biology. 2024;26(3):210-223. Doi:10.1038/s41556-024-01345-7
- National Cancer Institute. Cardiac Metastases: Statistics and Risk Factors. Updated 2025. Available at: https://www.cancer.gov
- European Medicines Agency. Horizon Europe Funding Opportunities in Cardio-Oncology. 2025. Available at: https://www.ema.europa.eu
- American Heart Association. Exercise and Cancer: Scientific Statement. Circulation. 2023;147(8):e56-e78. Doi:10.1161/CIR.0000000000001102

