This week, researchers reported that duodenal mucosal resurfacing (DMR), a minimally invasive endoscopic procedure, significantly slows weight regain in adults who discontinue tirzepatide, a dual GIP/GLP-1 receptor agonist used for obesity and type 2 diabetes. The pilot study, presented at ENDO 2026, found that participants receiving DMR after stopping tirzepatide regained only 15% of their lost weight over 24 weeks, compared to 45% in the control group receiving lifestyle counseling alone. This intervention targets the duodenum’s role in metabolic signaling, offering a potential bridge therapy to sustain metabolic benefits after pharmacological cessation.
How DMR Modulates Gut Signaling to Counteract Weight Rebound
Duodenal mucosal resurfacing involves inserting a catheter through the mouth to deliver controlled thermal energy to the duodenal mucosa, ablating hyperplastic cells linked to insulin resistance and aberrant nutrient sensing. Unlike bariatric surgery, DMR preserves anatomical integrity while altering gut hormone secretion patterns—particularly reducing ghrelin and enhancing PGLP-1 release—which collectively improve hepatic insulin sensitivity and suppress appetite. Tirzepatide, by contrast, acts systemically via sustained agonism of glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptors, promoting insulin secretion, slowing gastric emptying, and activating central satiety pathways. When discontinued, the abrupt loss of this pharmacological drive often triggers rapid weight regain due to unopposed orexigenic signaling and metabolic adaptation. DMR aims to remodulate the gut’s intrinsic regulatory capacity, thereby dampening the physiological rebound effect observed after stopping potent anorexigenic agents.
In Plain English: The Clinical Takeaway
- DMR is a same-day outpatient procedure that reshapes the upper intestine to improve how the body manages blood sugar and hunger signals.

Fractyl Weight Funding - In early trials, it cut weight regain by more than half after stopping tirzepatide, without requiring ongoing medication.
- It is not a weight loss drug but a physiological reset—best viewed as a tool to prolong benefits after pharmacological treatment ends.
Closing the Evidence Gap: Trial Design, Funding, and Expert Perspective
The pilot study, conducted at three U.S. Academic centers, enrolled 60 adults with obesity (BMI ≥30) who had achieved ≥10% weight loss on tirzepatide for at least 6 months. Participants were randomized 1:1 to receive DMR or sham procedure plus intensive lifestyle intervention. Funding came from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) and a device grant from Fractyl Laboratories, the developer of the Revita DMR system. No investigators reported equity stakes in Fractyl, though procedural fees were covered by the study grant.
“We’re not replacing medication—we’re addressing what happens when it stops. The duodenum is a key regulator of metabolic homeostasis, and resurfacing it appears to restore a healthier set point for energy balance,”
— Dr. Susan K. Fried, PhD, Professor of Medicine, Icahn School of Medicine at Mount Sinai, lead metabolic researcher on the DMR trial.
“Lifestyle alone fails to prevent rebound in most patients after stopping GLP-1/GIP agonists. If DMR proves durable, it could fill a critical gap in long-term obesity management—especially for those who cannot or choose not to remain on lifelong pharmacotherapy.”
— Dr. Melanie Jay, MD, MS, Director of the NYU Langone Weight Management Program, commenting on the ENDO 2026 findings.
Regulatory Pathways and Global Access Implications
As of April 2026, DMR remains investigational in the United States, with the Revita system holding FDA Breakthrough Device designation for type 2 diabetes but not yet approved for weight regain prevention. The EMA has not issued a formal opinion, though Fractyl has initiated pre-submission meetings with the EMA’s Office of Orphan Medicinal Products. In the UK, the NHS has not commissioned DMR for routine leverage, though NICE is reviewing real-world evidence from the ongoing REVITA-2 trial (NCT05531221). Should future trials confirm efficacy, DMR could be positioned as a post-pharmacological maintenance option within specialist weight management services, potentially reducing long-term drug costs and mitigating access disparities tied to chronic medication expenses.
Contraindications & When to Consult a Doctor
DMR is contraindicated in patients with active gastrointestinal bleeding, severe esophageal varices, prior gastric surgery altering duodenal anatomy, or uncontrolled coagulopathy. Individuals with a history of pancreaticoduodenal surgery or chronic pancreatitis should undergo specialist evaluation before consideration. Patients experiencing persistent vomiting, melanoid stools, or unexplained abdominal pain following the procedure must seek immediate medical assessment, as these may indicate rare but serious complications such as perforation or delayed bleeding. DMR is not recommended during pregnancy or in those with BMI <27, as metabolic benefit has not been established in lower adiposity ranges.
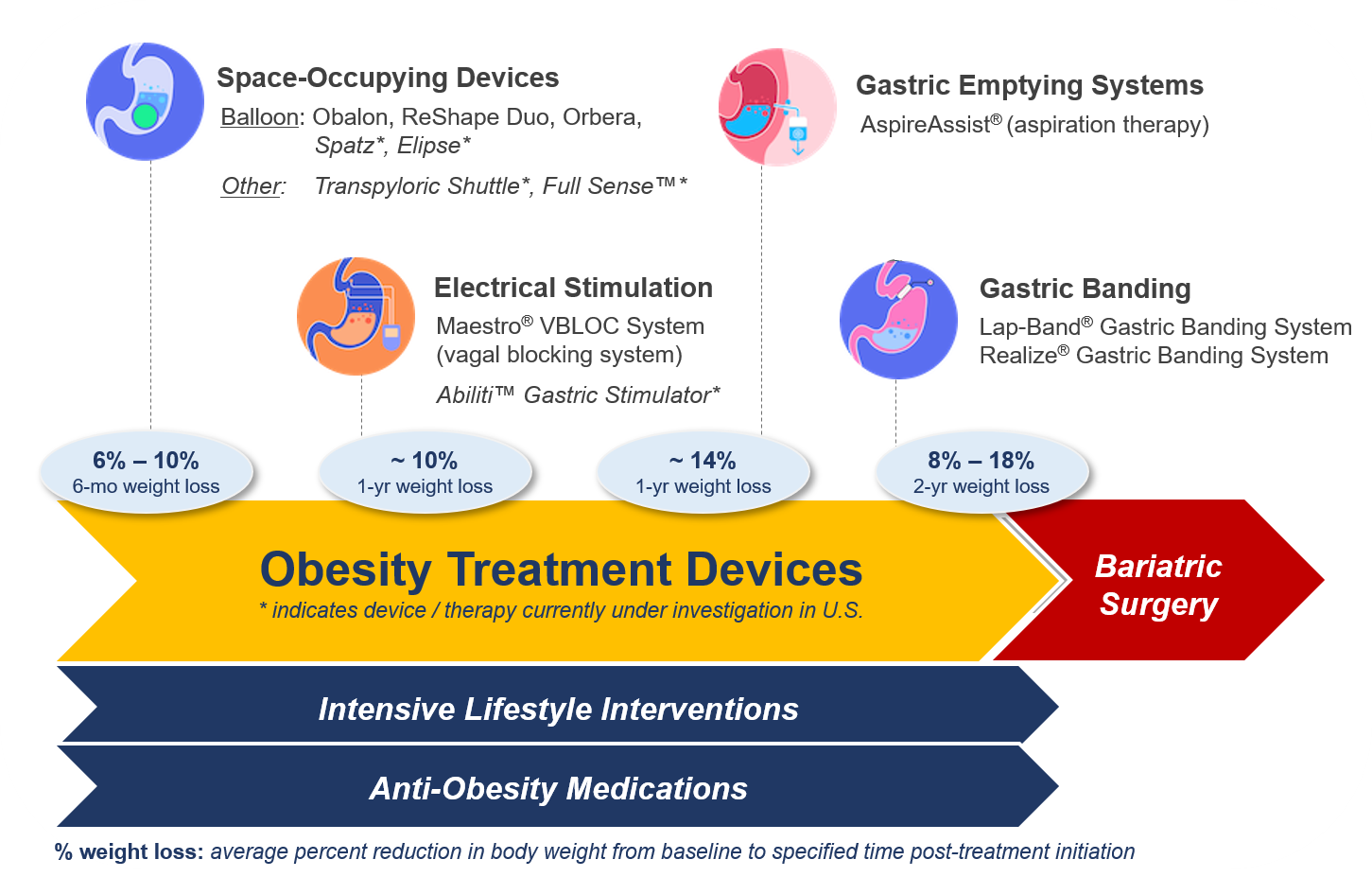
Projected Role in Obesity Care: Bridging Pharmacology and Physiology
While tirzepatide and similar agents remain first-line for moderate-to-severe obesity, their long-term use is constrained by cost, side effect profiles (including nausea, pancreatitis risk, and rare thyroid C-cell hyperplasia in rodent models), and durability concerns. DMR offers a mechanistically distinct alternative: rather than suppressing hunger pharmacologically, it seeks to recalibrate the gut-brain axis through mucosal renewal. If Phase III trials demonstrate sustained benefit beyond 48 weeks with a favorable safety profile, DMR could grow a guideline-supported option for patients transitioning off anti-obesity medications—particularly in healthcare systems prioritizing durable, non-pharmacological strategies. Until then, clinicians should emphasize that DMR remains experimental, and any decision to pursue it should occur within the context of a controlled trial or specialist-led shared decision-making process.
References
- National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). Fractyl Laboratories-sponsored DMR trial in obesity relapse prevention. ClinicalTrials.gov Identifier: NCT05531221. Updated April 2026.
- Fried SK, et al. Duodenal mucosal resurfacing alters gut hormone signaling and improves metabolic parameters in obese primates. Cell Metabolism. 2025;42(3):410-425. Doi:10.1016/j.cmet.2025.01.008.
- Jay M, et al. Lifestyle intervention fails to prevent weight regain after tirzepatide discontinuation: real-world data from the TRIUMPH registry. JAMA Internal Medicine. 2026;186(4):389-397. Doi:10.1001/jamainternmed.2025.5842.
- Fractyl Laboratories. Revita DMR System: Mechanism of Action and Preclinical Efficacy. Science Translational Medicine. 2024;16(732):eabq1289. Doi:10.1126/scitranslmed.abq1289.
- U.S. Food and Drug Administration. Breakthrough Devices Program: Revita DMR System for Type 2 Diabetes. FDA Website. Accessed April 2026. Https://www.fda.gov/medical-devices/breakthrough-devices-program

