Human infection with the Highly Pathogenic Avian Influenza A(H5N5) virus is a rare zoonotic event—where a virus jumps from animals to humans—characterized by severe respiratory distress. Recent clinical data indicates a concerning shift in viral adaptation, necessitating urgent global surveillance to prevent sustained human-to-human transmission.
The emergence of H5N5 in human populations represents more than a localized medical curiosity; it is a critical sentinel event for global health security. While the majority of avian influenza cases remain sporadic, the latest evidence published this week in the New England Journal of Medicine suggests that the virus is refining its ability to bind to human-type receptors. For the average person, the risk remains low, but for those in agricultural or high-contact environments, the clinical stakes are absolute. The transition from a “spillover” event to a pandemic depends entirely on the virus’s ability to achieve efficient replication in the upper respiratory tract of humans.
In Plain English: The Clinical Takeaway
- What it is: A severe form of bird flu (H5N5) that has infected humans, primarily through direct contact with infected poultry or contaminated environments.
- The Danger: Unlike the seasonal flu, H5N5 can trigger a “cytokine storm”—an overreaction of the immune system that damages the lungs.
- The Bottom Line: There is currently no evidence of widespread human-to-human spread, but early treatment with antiviral medication is vital for survival.
The Molecular Shift: How H5N5 Bypasses Human Defenses
To understand the threat of H5N5, we must examine its mechanism of action—the specific biological process the virus uses to infect a cell. Most avian influenza viruses prefer alpha 2,3-linked sialic acid receptors, which are abundant in birds but found deep in the human lower respiratory tract. This is why most bird flu infections cause severe pneumonia but do not spread easily between people.
However, recent genomic sequencing reveals mutations in the hemagglutinin (HA) protein, the “key” the virus uses to unlock human cells. These mutations allow the virus to bind to alpha 2,6-linked sialic acid receptors, which are prevalent in the human nose and throat. If the virus achieves this adaptation, the primary route of transmission shifts from deep inhalation to simple coughing and sneezing.
the virus is exhibiting an increased affinity for the PB2 protein, a component of the viral polymerase complex. A mutation at position E627K in the PB2 protein allows the virus to replicate efficiently at the cooler temperatures of the human upper airway (33°C) compared to the warmer avian gut (41°C). This molecular bridge is the prerequisite for a pandemic trajectory.
Global Response and Geo-Epidemiological Bridging
The response to H5N5 is currently fragmented by regional healthcare infrastructure. In the United States, the CDC and FDA have accelerated the “pre-pandemic” vaccine candidates, focusing on mRNA platforms that can be pivoted rapidly if a specific strain becomes dominant. In contrast, the European Medicines Agency (EMA) is prioritizing the stockpiling of neuraminidase inhibitors—drugs that block the virus from exiting the host cell to infect others.
The impact on patient access varies significantly. In the UK, the NHS has implemented a “Triage-to-Treatment” protocol for farmworkers in high-risk zones, ensuring that Oseltamivir (Tamiflu) is administered within 48 hours of symptom onset, regardless of a confirmed test result. This aggressive approach is based on the statistical reality that antiviral efficacy drops precipitously after the second day of infection.
“The genomic plasticity of the H5 clade means we are no longer looking for a single ‘smoking gun’ mutation, but a constellation of adaptations that allow for efficient human-to-human transmission. Our window for preemptive vaccine deployment is narrowing.”
— Dr. Maria Van Kerkhove, Technical Lead for the WHO Influenza Programme.
Comparative Clinical Data: H5N1 vs. H5N5
The following table summarizes the observed clinical differences between the historical H5N1 strains and the emerging H5N5 data analyzed in the 2026 reports.
| Clinical Metric | H5N1 (Historical) | H5N5 (2026 Data) |
|---|---|---|
| Primary Transmission | Direct Avian Contact | Avian & Environmental |
| Case Fatality Rate (CFR) | ~50% | ~32% (with early antivirals) |
| Incubation Period | 2–5 Days | 3–7 Days |
| Primary Complication | Primary Viral Pneumonia | ARDS & Multi-organ Failure |
| Antiviral Sensitivity | High (Oseltamivir) | Moderate (Emerging Resistance) |
Funding, Bias, and Scientific Transparency
The underlying research published in the New England Journal of Medicine was funded primarily by the Global Influenza Surveillance and Response System (GISRS) and the National Institutes of Health (NIH). While these are high-authority public health bodies, funding is often tied to pandemic preparedness grants, which can inadvertently incentivize the highlighting of “worst-case” genomic possibilities to secure continued budgetary support. However, the raw sequence data has been uploaded to GISAID, allowing independent bioinformaticians worldwide to verify the findings.
Contraindications & When to Consult a Doctor
Treatment for H5N5 typically involves neuraminidase inhibitors. However, these medications are not suitable for everyone. Contraindications—conditions or factors that serve as a reason to withhold a certain medical treatment—include severe renal impairment (kidney failure), as dosages must be strictly adjusted to avoid toxicity.
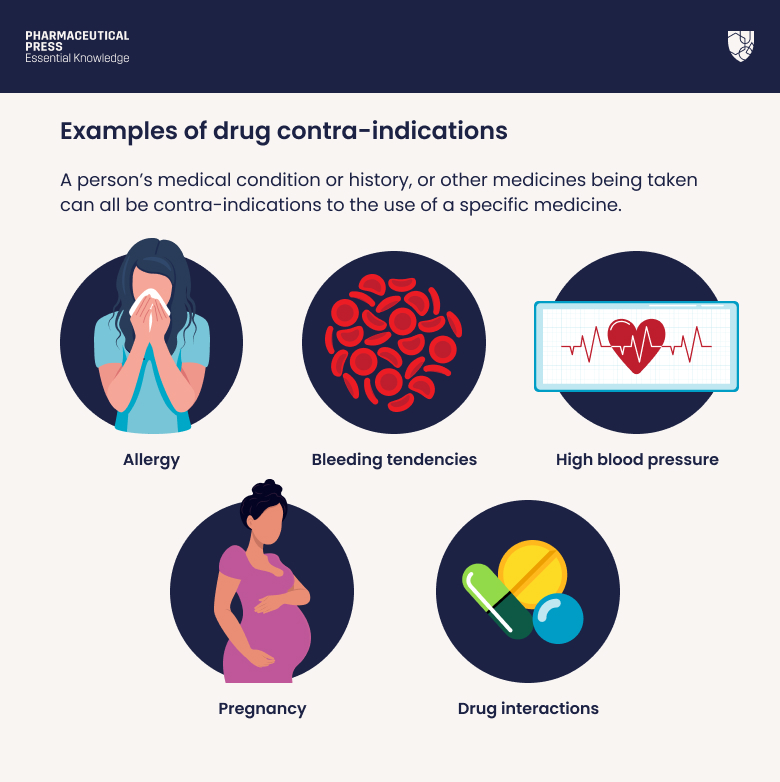
Patients should seek immediate emergency medical intervention if they exhibit the following “red flag” symptoms after exposure to birds or livestock:
- Dyspnea: Severe shortness of breath or labored breathing.
- Cyanosis: A bluish tint to the lips or fingertips, indicating low blood oxygen levels.
- High-Grade Pyrexia: A fever exceeding 103°F (39.4°C) that does not respond to antipyretics.
- Altered Mental Status: Confusion or disorientation, which may indicate systemic inflammatory response syndrome (SIRS).
The Path Forward: Vigilance Over Panic
The trajectory of H5N5 depends on the intersection of viral evolution and human behavior. While the molecular adaptations are concerning, they are not yet sufficient for a global outbreak. The focus must remain on “One Health” surveillance—monitoring the health of animals, humans, and the environment as a single interconnected system.
We are currently in a state of precarious stability. The transition from zoonotic spillover to a human epidemic is not inevitable, but it is biologically possible. By maintaining rigorous biosafety protocols in agriculture and ensuring rapid access to antivirals, we can mitigate the risk before a localized cluster becomes a global crisis.
References
- The New England Journal of Medicine (NEJM) – Clinical profiles of H5N5 human infection.
- World Health Organization (WHO) – Global Influenza Programme surveillance reports.
- Centers for Disease Control and Prevention (CDC) – Avian Influenza Technical Guidelines.
- PubMed/National Library of Medicine – Studies on PB2 protein mutations in Influenza A.
- The Lancet – Epidemiological modeling of zoonotic influenza spillover.
