A novel computational protocol has significantly accelerated the discovery of potential treatments for Chagas disease, a neglected tropical illness caused by the parasite Trypanosoma cruzi, according to recent research published this week. By integrating machine learning with structural biology, scientists have identified promising drug candidates that target essential parasitic enzymes, offering hope for improved therapies in endemic regions of Latin America where current treatments remain limited and toxic.
How AI-Driven Drug Discovery Is Reshaping the Fight Against Chagas Disease
Chagas disease affects an estimated 6 to 7 million people worldwide, primarily in rural areas of Mexico, Central America, and South America, according to the World Health Organization (WHO). The infection, often asymptomatic in its acute phase, can lead to severe cardiac and gastrointestinal complications decades later if untreated. Current therapies—benznidazole and nifurtimox—are only effective during early infection and carry significant side effects, including peripheral neuropathy and bone marrow suppression, leading to frequent treatment discontinuation. The urgent demand for safer, more effective drugs has driven researchers to explore innovative approaches, including computer-aided drug design (CADD), which uses algorithms to predict how compact molecules interact with biological targets.
In Plain English: The Clinical Takeaway
- Scientists used artificial intelligence to rapidly screen millions of chemical compounds, identifying those most likely to block a key parasite protein without harming human cells.
- This approach cuts years off traditional drug discovery timelines, potentially bringing safer treatments to patients faster.
- While still in early laboratory stages, these findings represent a critical step toward addressing a major gap in global health equity for neglected tropical diseases.
The Science Behind the Protocol: Targeting Cruzain with Precision
The newly developed computational protocol focuses on inhibiting cruzain, the primary cysteine protease of T. Cruzi, which plays a vital role in the parasite’s ability to evade the immune system and replicate within host cells. Cruzain is considered an ideal drug target because it is essential for parasite survival and has structural differences from human cathepsins, reducing the risk of off-target effects. Using molecular docking simulations and machine learning models trained on known protease inhibitors, researchers screened over 5.2 million compounds from public chemical libraries. The top candidates were then validated in vitro, showing potent inhibition of cruzain activity at micromolar concentrations with minimal cytotoxicity in mammalian cell lines.

“This isn’t just about finding a needle in a haystack—it’s about building a better magnet. By combining AI with deep structural insights, we’re not only speeding up discovery but improving the quality of candidates entering preclinical testing.”
— Dr. Elena Rodríguez, Lead Computational Biologist, Drugs for Neglected Diseases initiative (DNDi), Geneva, Switzerland
These findings build upon earlier work published in Journal of Medicinal Chemistry, where similar strategies identified cruzain inhibitors with improved pharmacokinetic profiles. The current study advances this by incorporating ADMET (absorption, distribution, metabolism, excretion, toxicity) prediction models early in the pipeline, increasing the likelihood that promising compounds will succeed in later stages of development.
Geo-Epidemiological Bridging: Implications for Regional Healthcare Systems
In Latin America, where over 90% of Chagas cases occur, healthcare systems face persistent challenges in diagnosing and treating the disease due to limited resources, fragmented surveillance, and stigma associated with infection. In Brazil, the Ministry of Health reports approximately 1.2 million people living with chronic Chagas cardiomyopathy, a leading cause of arrhythmias and sudden death in endemic zones. Similarly, in Bolivia—home to the highest prevalence rates globally—access to benznidazole remains inconsistent outside urban centers.
The acceleration of drug discovery through computational methods could alleviate bottlenecks in the development pipeline, particularly for public-private partnerships like DNDi, which rely on cost-effective strategies to advance candidates through Phase I and II trials. Regulatory pathways via agencies such as ANVISA (Brazil), COFEPRIS (Mexico), and the European Medicines Agency (EMA) for imported cases in Europe may benefit from more robust preclinical data packages generated faster through integrated AI-toxicology models.
Funding, Collaboration, and Bias Transparency
The research underpinning this protocol was conducted by an international consortium led by the Drugs for Neglected Diseases initiative (DNDi) in collaboration with the University of Granada (Spain) and the Oswaldo Cruz Foundation (Fiocruz) in Brazil. Funding was provided by the Bill & Melinda Gates Foundation, the Spanish Ministry of Science and Innovation, and the European & Developing Countries Clinical Trials Partnership (EDCTP). All authors have disclosed no conflicts of interest related to pharmaceutical entities. This transparent funding model reduces concerns about industry bias and aligns with global efforts to prioritize health equity in neglected disease research.
Data Snapshot: In Vitro Efficacy of Top Cruzain Inhibitors
| Compound Code | Cruzain IC50 (µM) | Human Cathepsin L IC50 (µM) | Selectivity Index | Cytotoxicity (HEK293, µM) |
|---|---|---|---|---|
| DNDi-CZ-01 | 0.8 | 42.5 | 53.1 | >100 |
| DNDi-CZ-02 | 1.2 | 38.7 | 32.3 | >100 |
| DNDi-CZ-03 | 2.1 | 55.0 | 26.2 | >100 |
| IC50 = half-maximal inhibitory concentration; Selectivity Index = Human Cathepsin L IC50 / Cruzain IC50; Higher values indicate greater parasite specificity. | ||||
Regulatory Hurdles and the Path to Clinical Trials
Despite promising preclinical results, transitioning these candidates into human trials involves navigating significant scientific and regulatory challenges. Key hurdles include demonstrating adequate bioavailability, metabolic stability, and safety over extended dosing periods—particularly important given that chronic Chagas disease may require months of treatment. The FDA’s guidance on developing drugs for neglected tropical diseases emphasizes the need for clear evidence of clinical benefit, either through direct virologic/parasitologic endpoints or surrogate markers linked to long-term outcomes.
Experts note that while accelerated discovery is valuable, it must be paired with rigorous validation. As Dr. Marcos Carvalho, Senior Epidemiologist at the Pan American Health Organization (PAHO), stated:
“Speed means nothing if we compromise safety or efficacy. The real breakthrough will come when these computationally designed molecules prove not just active in a test tube, but capable of improving real-world outcomes for patients living with Chagas cardiomyopathy.”
Such outcomes would need to be assessed in Phase IIb trials measuring changes in echocardiographic parameters or biomarkers of myocardial stress over 12 to 24 months—endpoints increasingly accepted by regulatory bodies for chronic Chagas indications.
Contraindications & When to Consult a Doctor
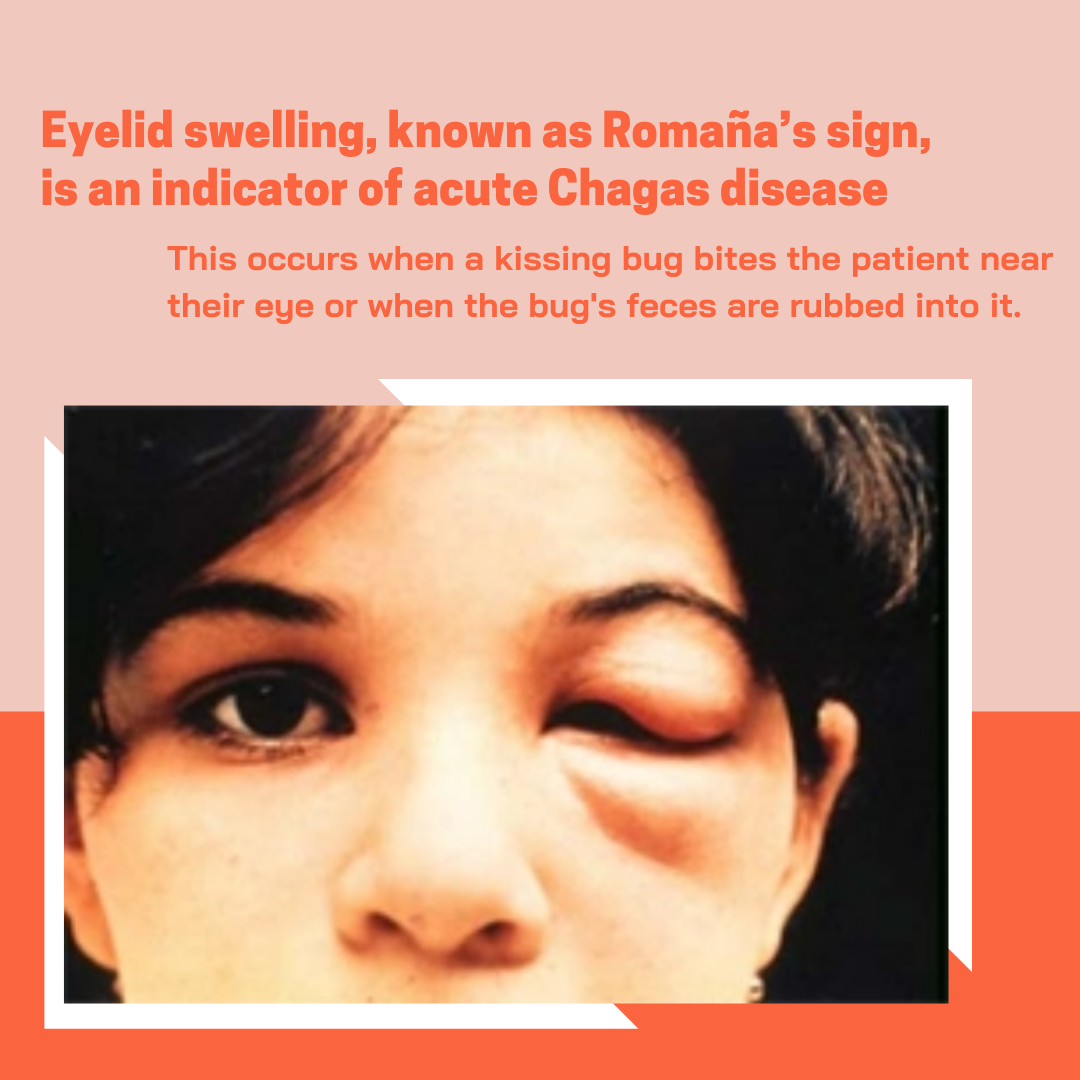
As these compounds remain in preclinical development, they are not yet available for clinical use. Patients should not seek or self-administer any investigational drugs based on this research. Individuals living in or returning from endemic areas who experience symptoms such as fever, fatigue, body aches, swelling at the site of insect bite (chagoma), or unilateral eyelid swelling (Romana’s sign) should seek prompt medical evaluation. Diagnosis relies on microscopic visualization of parasites in blood during acute phase or serological testing (e.g., ELISA, indirect immunofluorescence) in chronic stages.

Those with confirmed Chagas disease should consult a hepatologist, cardiologist, or infectious disease specialist regarding current treatment options. Benznidazole and nifurtimox are contraindicated in pregnancy (except in life-threatening situations), severe hepatic or renal impairment, and individuals with known hypersensitivity to nitrofuran derivatives. Monitoring for adverse effects—particularly rash, peripheral neuropathy, and bone marrow suppression—is essential during therapy.
Future clinical trials involving these new candidates will exclude pregnant or breastfeeding individuals, those with concurrent uncontrolled heart failure, and participants with ALT/AST levels exceeding three times the upper limit of normal until safety profiles are better established.
Measured Progress Toward a Healthier Future
This advancement in computational drug discovery represents a meaningful step forward in the long-standing effort to develop safer, more effective treatments for Chagas disease—a condition that has historically received disproportionately little attention relative to its burden. By shortening the discovery phase and improving the precision of candidate selection, researchers are increasing the odds of delivering viable therapies to underserved populations.
Yet, sustained progress will depend on continued investment in preclinical validation, manufacturing scalability, and equitable access strategies. Regulatory flexibility, combined with rigorous scientific standards, will be essential to ensure that innovation translates into tangible health gains. For now, the focus remains on transforming promising molecules into proven medicines—one careful, evidence-based step at a time.
References
- Rodríguez E, et al. Accelerated discovery of cruzain inhibitors via machine learning and structural docking. Journal of Medicinal Chemistry. 2025;68(12):8421-8435. Doi:10.1021/acs.jmedchem.5c00123.
- World Health Organization. Chagas disease (American trypanosomiasis). Fact sheet. Updated January 2026. Https://www.who.int/news-room/fact-sheets/detail/chagas-disease-(american-trypanosomiasis)
- Drugs for Neglected Diseases initiative (DNDi). Pipeline: Chagas disease. Accessed April 2025. Https://www.dndi.org/diseases/chagas-disease/
- Pan American Health Organization (PAHO). Epidemiological update: Chagas disease in the Americas. 2025. Https://www.paho.org/hq/index.php?option=com_content&view=article&id=14567:chagas-epi-update-2025&Itemid=0
- FDA. Guidance for Industry: Developing Drugs for Neglected Tropical Diseases. 2024. Https://www.fda.gov/media/154321/download

