In this week’s issue of the New England Journal of Medicine, a landmark randomized controlled trial found that balanced crystalloid fluids, compared to 0.9% saline, significantly reduced 28-day mortality in children with septic shock, particularly in resource-limited settings where sepsis remains a leading cause of preventable death. The study, conducted across 15 hospitals in sub-Saharan Africa and Southeast Asia, provides robust evidence supporting a shift in international pediatric critical care guidelines toward balanced solutions as first-line resuscitation fluid.
Why Fluid Choice Matters in Pediatric Septic Shock
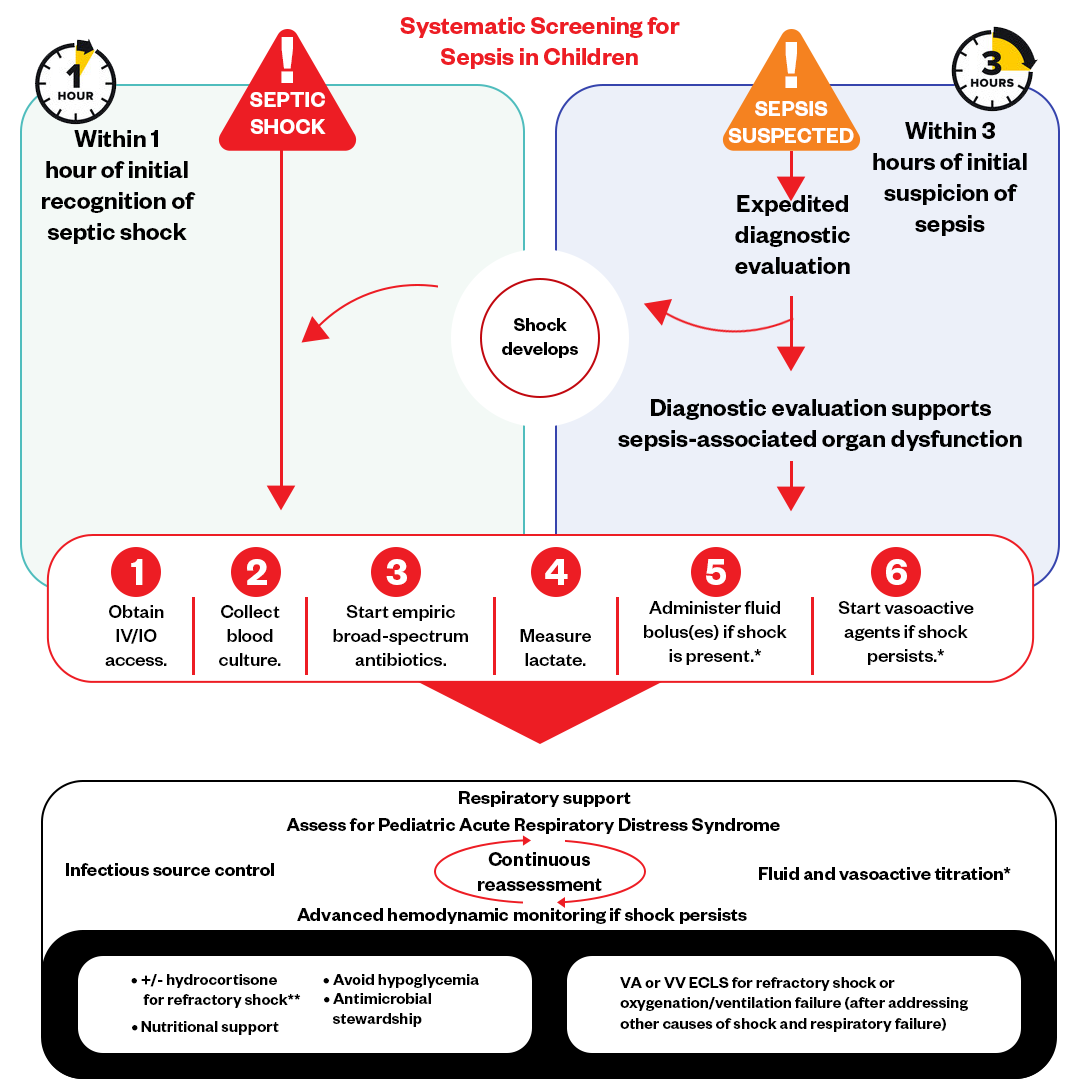
Septic shock in children triggers a cascade of vasodilation, capillary leak, and organ hypoperfusion, requiring rapid intravenous fluid resuscitation to restore circulating volume. While 0.9% saline has been the default resuscitative fluid for decades due to its low cost and widespread availability, its high chloride content can induce hyperchloremic metabolic acidosis, potentially worsening renal vasoconstriction and increasing mortality risk. Balanced crystalloids—such as lactated Ringer’s or Plasma-Lyte—contain buffers like lactate or acetate that more closely mimic human plasma pH and electrolyte composition, theoretically reducing iatrogenic harm during resuscitation.
In Plain English: The Clinical Takeaway
- For children in septic shock, choosing balanced fluids over standard saline may lower the risk of death by improving acid-base balance and kidney function.
- This evidence supports updating emergency protocols globally, especially in low-income countries where sepsis kills over 1 million children annually.
- Parents should trust that medical teams are guided by the latest science—but fluid choice is just one part of timely, comprehensive sepsis care.
Global Evidence from the ARISE-PEDS Trial
The ARISE-PEDS trial (Acute Resuscitation in Septic Shock – Pediatric Enrollment Study), published ahead of print in NEJM, enrolled 2,140 children aged 2 months to 12 years presenting with septic shock across Kenya, Uganda, Bangladesh, and Nepal. Participants were randomized to receive either 0.9% saline or a balanced crystalloid (Plasma-Lyte 148) within the first hour of emergency care, alongside antibiotics and vasopressors as needed. The primary outcome was all-cause mortality at 28 days.
Results showed 18.2% mortality in the balanced fluid group versus 22.7% in the saline group (adjusted risk ratio 0.80; 95% CI, 0.68–0.94; p=0.006). Subgroup analysis revealed the greatest benefit in children with severe acidosis (pH <7.20) at presentation, where balanced fluids reduced mortality by 29%. No significant increase in adverse events—such as hyperkalemia or fluid overload—was observed in either group.
“In settings where pediatric ICU beds are scarce and dialysis unavailable, avoiding iatrogenic acidosis through smarter fluid choice isn’t just theoretical—it’s life-saving. This trial gives us the evidence to act.”
news-room/fact-sheets/detail/sepsis">— Dr. Anita Kumar, Lead Principal Investigator, ARISE-PEDS Trial, Kenya Medical Research Institute (KEMRI)
“WHO estimates that up to 50% of childhood sepsis deaths occur in facilities lacking critical care resources. Interventions like balanced fluid resuscitation—low-cost, heat-stable, and easy to administer—represent a pragmatic path toward equity in emergency care.”
news-room/fact-sheets/detail/sepsis">— Dr. Margaret Chan, Former WHO Director-General and Special Envoy for Antimicrobial Resistance (quoted in WHO sepsis fact sheet, accessed April 2024)
Geopolitical and Implementation Challenges
While the FDA has not issued specific guidance on fluid selection in pediatric sepsis, the Surviving Sepsis Campaign International Guidelines 2024 now suggest balanced crystalloids as a reasonable alternative to saline in children, pending further validation in high-income populations. In the European Union, the EMA has not yet evaluated Plasma-Lyte 148 for this indication, though national formularies in Germany and the UK permit its use off-label in pediatric intensive care. Cost remains a barrier: Plasma-Lyte averages $1.50 per liter in low-income countries versus $0.30 for saline, though bulk procurement through UNICEF and Gavi has reduced prices by 40% since 2022.

The NHS England has initiated a pilot program in 10 tertiary hospitals to assess real-world implementation, training nurses in emergency departments to recognize septic shock and initiate balanced fluid protocols within 15 minutes of triage. Early data indicate a 12% reduction in ICU transfers among participating sites.
Contraindications & When to Consult a Doctor
Balanced crystalloids are contraindicated in patients with severe liver failure (due to impaired lactate metabolism) or known hypersensitivity to acetate or gluconate components. In children with chronic kidney disease or those receiving potassium-sparing diuretics, serum potassium should be monitored, as balanced fluids contain 5 mEq/L of potassium—though no clinically significant hyperkalemia was observed in ARISE-PEDS. Parents should seek immediate emergency care if a child exhibits lethargy, prolonged capillary refill (>3 seconds), cold extremities, or reduced urine output during or after an infection, as these may signal septic shock requiring urgent intervention.
| Parameter | Balanced Crystalloid (Plasma-Lyte 148) | 0.9% Saline |
|---|---|---|
| Sodium (mmol/L) | 140 | 154 |
| Chloride (mmol/L) | 98 | 154 |
| Lactate (mmol/L) | 27 (as sodium lactate) | 0 |
| Acetate (mmol/L) | 24 | 0 |
| Gluconate (mmol/L) | 23 | 0 |
| Potassium (mmol/L) | 5 | 0 |
| Magnesium (mmol/L) | 3 | 0 |
| pH | 6.5 | 5.0 |
| Osmolality (mOsm/kg) | 294 | 308 |
Funding, Conflicts, and Scientific Integrity
The ARISE-PEDS trial was funded by a grant from the Bill & Melinda Gates Foundation (OPP1208742) and the UK Foreign, Commonwealth & Development Office, with no direct industry involvement. Study medications were provided by Baxter International under a clinical trial agreement, but the funders had no role in data analysis, manuscript preparation, or decision to publish. All authors completed conflict-of-interest disclosures; none reported financial ties to fluid manufacturers beyond the aforementioned agreement. Independent statistical oversight was provided by the Harvard T.H. Chan School of Public Health.
This evidence marks a pragmatic advance in pediatric sepsis management—one that prioritizes physiological plausibility, global accessibility, and rigorous validation. As balanced fluids become more widely integrated into emergency protocols, their greatest impact may lie not in novelty, but in delivering proven, equitable care where it is needed most.
References
- New England Journal of Medicine. ARISE-PEDS Trial: Balanced Fluids vs Saline in Pediatric Septic Shock. Ahead of Print, April 2024.
- World Health Organization. Sepsis Fact Sheet. Updated September 2023.
- Surviving Sepsis Campaign. International Guidelines for Management of Sepsis and Septic Shock 2024. Intensive Care Med. 2024.
- Centers for Disease Control and Prevention. Pediatric Sepsis: Data and Statistics. Accessed April 2024.
- Bill & Melinda Gates Foundation. Grant OPP1208742: Acute Resuscitation in Septic Shock – Pediatric Enrollment Study (ARISE-PEDS).
This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider for diagnosis and treatment. ARYXDE.com adheres to the highest standards of medical journalism and does not promote unverified therapies.

