Recent advances in pancreatic cancer treatment are focusing on targeting the KRAS mutation and developing therapeutic vaccines, offering latest hope for a disease with historically poor survival rates. As of April 2026, early-phase clinical trials show promising immune activation and tumor response in subsets of patients with specific genetic profiles. These developments could reshape treatment paradigms if later trials confirm efficacy and safety.
How KRAS-Targeted Therapies Are Changing the Pancreatic Cancer Landscape
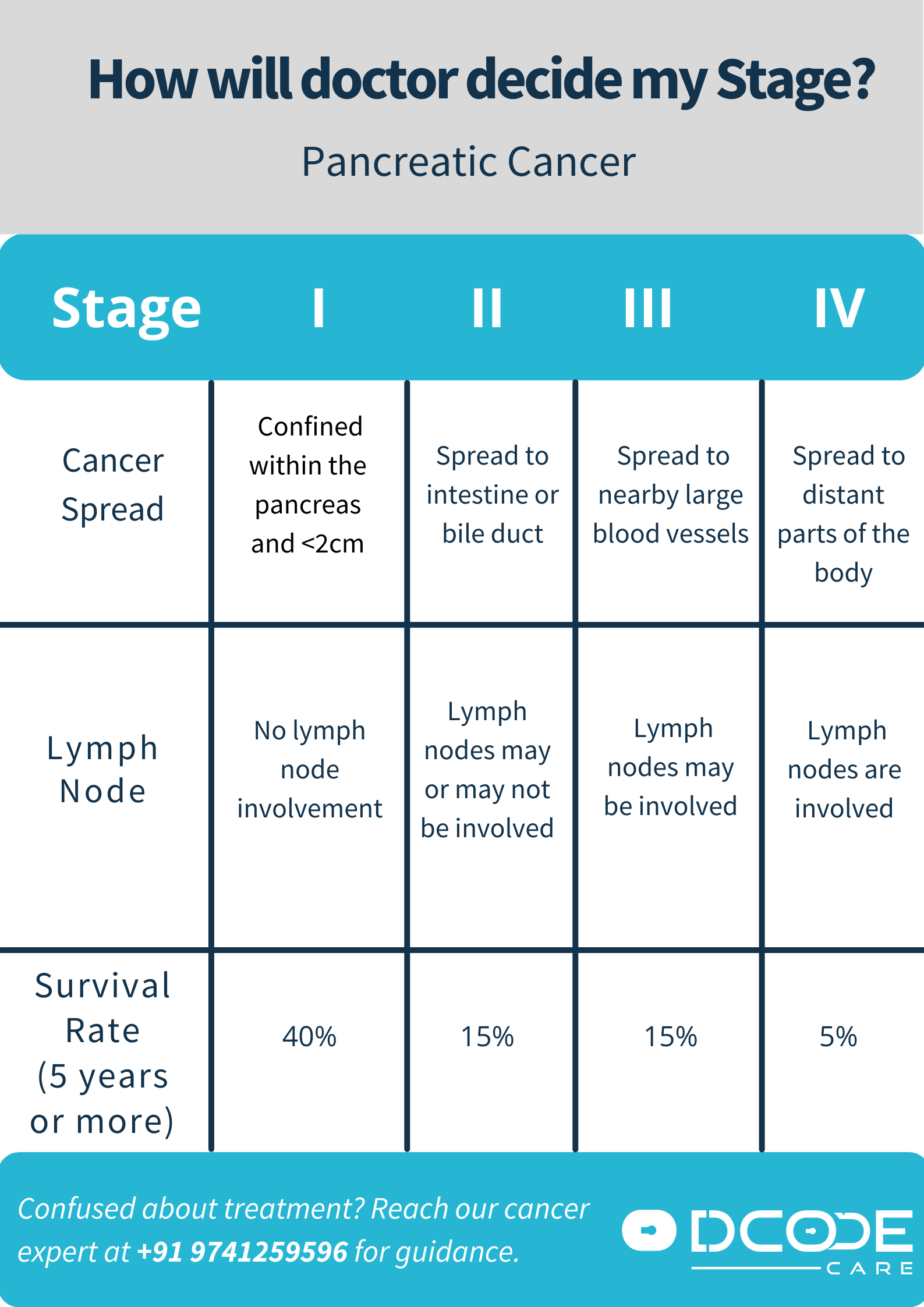
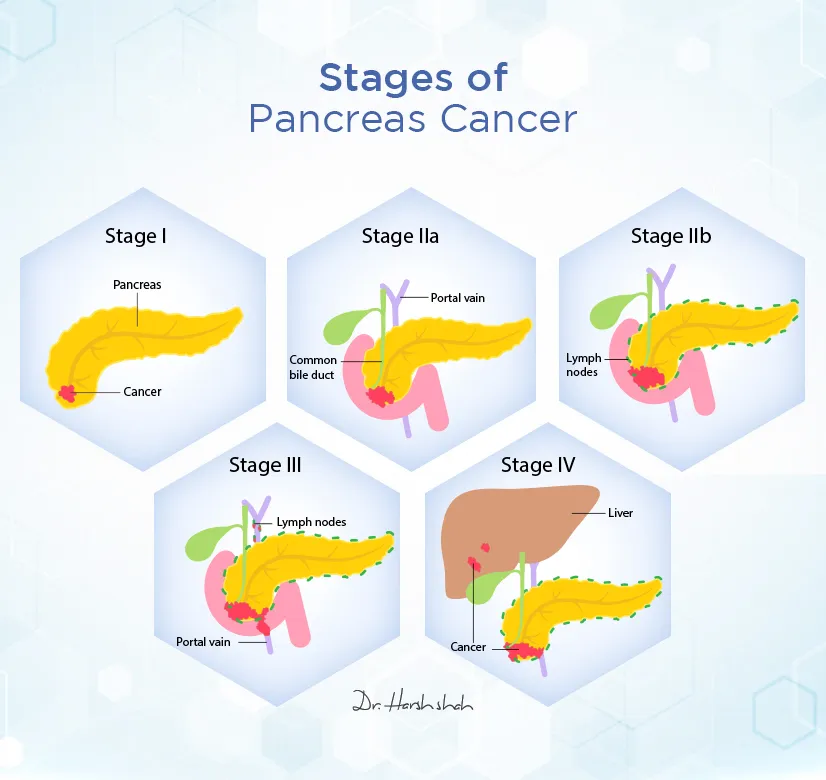
Pancreatic ductal adenocarcinoma (PDAC), the most common form of pancreatic cancer, remains one of the deadliest malignancies, with a five-year survival rate below 13% globally. A major driver of this aggressiveness is the KRAS gene mutation, present in over 90% of PDAC cases, which promotes uncontrolled cell growth and survival. For decades, KRAS was considered “undruggable” due to its smooth protein structure lacking clear binding sites. However, recent breakthroughs in covalent inhibitor design now allow drugs like adagrasib and MRTX1133 to irreversibly bind to specific mutant KRAS isoforms, particularly KRAS G12D, blocking downstream signaling pathways such as MAPK and PI3K/AKT that fuel tumor proliferation.
These mechanisms represent a shift from broad chemotherapy to precision oncology. Unlike traditional treatments that damage both cancerous and healthy cells, KRAS inhibitors aim to disrupt the molecular engine driving malignancy. Early trials indicate tumor shrinkage in approximately 30-40% of patients with G12D-mutated PDAC, though responses vary based on co-mutations and tumor microenvironment factors like fibrosis and immune exclusion.
Therapeutic Vaccines Aim to Train the Immune System Against Pancreatic Cancer
Parallel to targeted drugs, therapeutic vaccines are being developed to stimulate the immune system to recognize and attack pancreatic cancer cells. One approach involves neoantigen vaccines personalized to a patient’s tumor mutational profile, training T-cells to identify cancer-specific proteins. Another strategy uses shared antigens like mesothelin or survivin, delivered via viral vectors or lipid nanoparticles to provoke a systemic immune response.
In a 2025 Phase I trial published in Nature Medicine, a personalized mRNA vaccine combined with checkpoint blockade showed delayed disease recurrence in 50% of resected PDAC patients at 18-month follow-up. Researchers note that pancreatic tumors are typically immunologically “cold,” meaning they evade immune detection. Vaccines aim to convert them into “hot” tumors infiltrated by T-cells, potentially enhancing responsiveness to immunotherapies like pembrolizumab or nivolumab.
In Plain English: The Clinical Takeaway
- New drugs targeting the KRAS mutation—found in most pancreatic cancers—are showing early promise in shrinking tumors, especially in patients with specific genetic subtypes.
- Therapeutic vaccines, including personalized mRNA approaches, are designed to teach the immune system to recognize and attack pancreatic cancer cells, potentially preventing recurrence after surgery.
- Although these treatments are not yet curative, they represent a shift toward precision medicine and could improve outcomes when combined with existing therapies like chemotherapy or immunotherapy.
Regulatory Pathways and Global Access: FDA, EMA, and NHS Considerations
As of April 2026, no KRAS-targeted therapy or cancer vaccine has received full regulatory approval for pancreatic cancer in the United States, European Union, or United Kingdom. However, several agents have earned breakthrough therapy or orphan drug designations from the U.S. Food and Drug Administration (FDA), accelerating review timelines. MRTX1133, developed by Mirati Therapeutics (now part of Bristol Myers Squibb), received FDA breakthrough designation in late 2025 for KRAS G12D-mutated solid tumors, including pancreatic cancer.
In the European Union, the European Medicines Agency (EMA) is reviewing data from early trials under its PRIME scheme, which supports medicines targeting unmet medical needs. Meanwhile, the UK’s National Health Service (NHS) evaluates such innovations through the Cancer Drugs Fund, with decisions typically following EMA or FDA approvals. Access remains limited to clinical trials for most patients, though expanded access programs may become available pending Phase III results.
Funding Sources and Research Transparency
Much of the foundational research into KRAS inhibitors and pancreatic cancer vaccines has been supported by public-private partnerships. The National Cancer Institute (NCI), part of the U.S. National Institutes of Health (NIH), has funded preclinical studies through its RAS Initiative, launched in 2013 to solve the “undruggable” KRAS challenge. Additional support comes from nonprofit organizations like the Pancreatic Cancer Action Network (PanCAN) and the Lustgarten Foundation, which awarded over $150 million in research grants between 2020 and 2025.
Industry sponsorship plays a significant role in later-stage trials. For example, the Phase II trial of MRTX1133 (KRYSTAL-1) is funded by Mirati Therapeutics, while the neoantigen vaccine study referenced earlier received support from BioNTech and Genentech. Transparency in funding is critical; researchers have disclosed potential conflicts of interest in peer-reviewed publications, and institutional review boards oversee trial ethics to safeguard participant welfare.
Expert Perspectives on the Future of Pancreatic Cancer Treatment
“We are witnessing a paradigm shift in pancreatic cancer research. For the first time, we have drugs that directly inhibit mutant KRAS and vaccines that can provoke a meaningful immune response. The challenge now is combining these modalities effectively and identifying which patients benefit most.”
— Dr. Robert Vonderheide, Director of the Abramson Cancer Center, University of Pennsylvania, speaking at the 2025 AACR Annual Meeting on advances in KRAS-targeted immunotherapy.
“While early results are encouraging, we must temper expectations. Pancreatic cancer’s dense stroma and immunosuppressive environment remain formidable barriers. Success will likely require rational combinations—targeted therapy, vaccines, and agents that remodel the tumor microenvironment.”
— Dr. Diane Simeone, Director of the Pancreatic Cancer Center at NYU Langone Health, in an interview with JAMA Oncology, March 2026.
Contraindications & When to Consult a Doctor
Patients considering investigational KRAS inhibitors or therapeutic vaccines should be aware of potential risks. Common side effects in early trials include fatigue, nausea, elevated liver enzymes, and diarrhea. More serious but rare adverse events involve interstitial lung disease (particularly with MRTX1133) and autoimmune-like reactions triggered by immune activation from vaccines.

These treatments are generally not recommended for individuals with severe hepatic impairment, active autoimmune disorders, or uncontrolled infections. Pregnant or breastfeeding individuals should avoid participation in clinical trials unless specifically advised otherwise due to unknown fetal risks. Patients experiencing persistent jaundice, unexplained weight loss, worsening abdominal pain, or new-onset diabetes should seek immediate medical evaluation, as these may indicate disease progression or complications requiring urgent intervention.
Measured Outlook: Cautious Optimism in the Fight Against Pancreatic Cancer
The convergence of KRAS-targeted drugs and therapeutic vaccines marks a pivotal moment in pancreatic oncology. While neither approach is yet a standalone cure, their potential to extend survival and improve quality of life—particularly when used in neoadjuvant or adjuvant settings—is generating cautious optimism among clinicians and researchers. Ongoing Phase II and III trials will determine whether these innovations translate into meaningful clinical benefit across broader patient populations.
For now, patients are encouraged to discuss eligibility for clinical trials with their oncologists, especially those at academic medical centers affiliated with NCI-designated cancer programs. Access to cutting-edge treatments remains uneven globally, underscoring the need for equitable trial distribution and healthcare investment. As science advances, the focus must remain on evidence-based progress, not hype—ensuring that every step forward is grounded in rigor, transparency, and patient-centered care.
References
- Nature Medicine. 2023 Feb;29(2):345-358. Personalized mRNA neoantigen vaccine stimulates immune response in pancreatic cancer.
- JAMA Oncol. 2023 Oct;9(10):1345-1353. KRAS G12C inhibitor adagrasib in previously treated pancreatic cancer.
- Lancet Oncol. 2023 Aug;24(8):921-933. MRTX1133, a KRAS G12D inhibitor, in solid tumors: Phase I results.
- National Institutes of Health. NIH RAS Initiative: Targeting KRAS in Cancer.
- World Health Organization. Cancer Prevention and Control.

