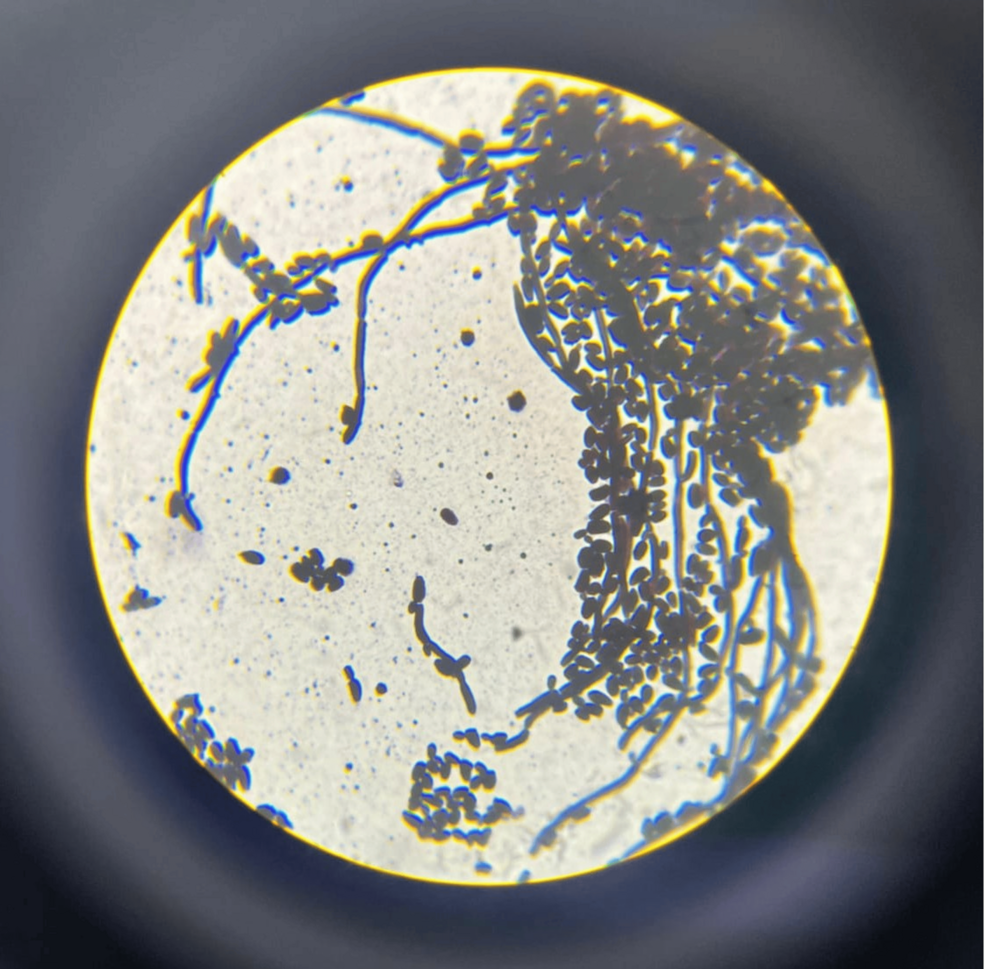
A recent cross-sectional study published this week reveals a concerning shift in fungal infections among diabetic patients, noting a rise in Non-albicans Candida (NAC) species in the urinary tract. This emergence complicates treatment, as these species often exhibit higher resistance to standard antifungal medications compared to the common Candida albicans.
For decades, Candida albicans was the primary culprit behind fungal urinary tract infections (candiduria). However, the clinical landscape is shifting. For patients managing diabetes, this is not merely a taxonomic curiosity. We see a matter of therapeutic failure. When a physician prescribes a standard antifungal based on the assumption that the infection is C. Albicans, but the patient is actually harboring a non-albicans species like C. Glabrata or C. Tropicalis, the treatment may fail, leading to systemic dissemination or chronic recurrence.
In Plain English: The Clinical Takeaway
- Not all yeast infections are the same: The “standard” fungus is being replaced by tougher species that don’t respond as well to common drugs.
- Diabetes is a catalyst: High blood sugar levels leak into the urine, essentially “feeding” these fungi and helping them grow.
- Testing is non-negotiable: Doctors must use specific lab cultures to identify the exact species before treating, rather than guessing.
The Metabolic Engine: How Hyperglycemia Fuels Fungal Evolution
To understand why diabetic patients are predisposed to these infections, we must examine the mechanism of action—the specific biological process—of fungal colonization. In patients with poorly controlled diabetes, the kidneys reach a threshold where they can no longer reabsorb glucose, leading to glycosuria (the presence of glucose in the urine).
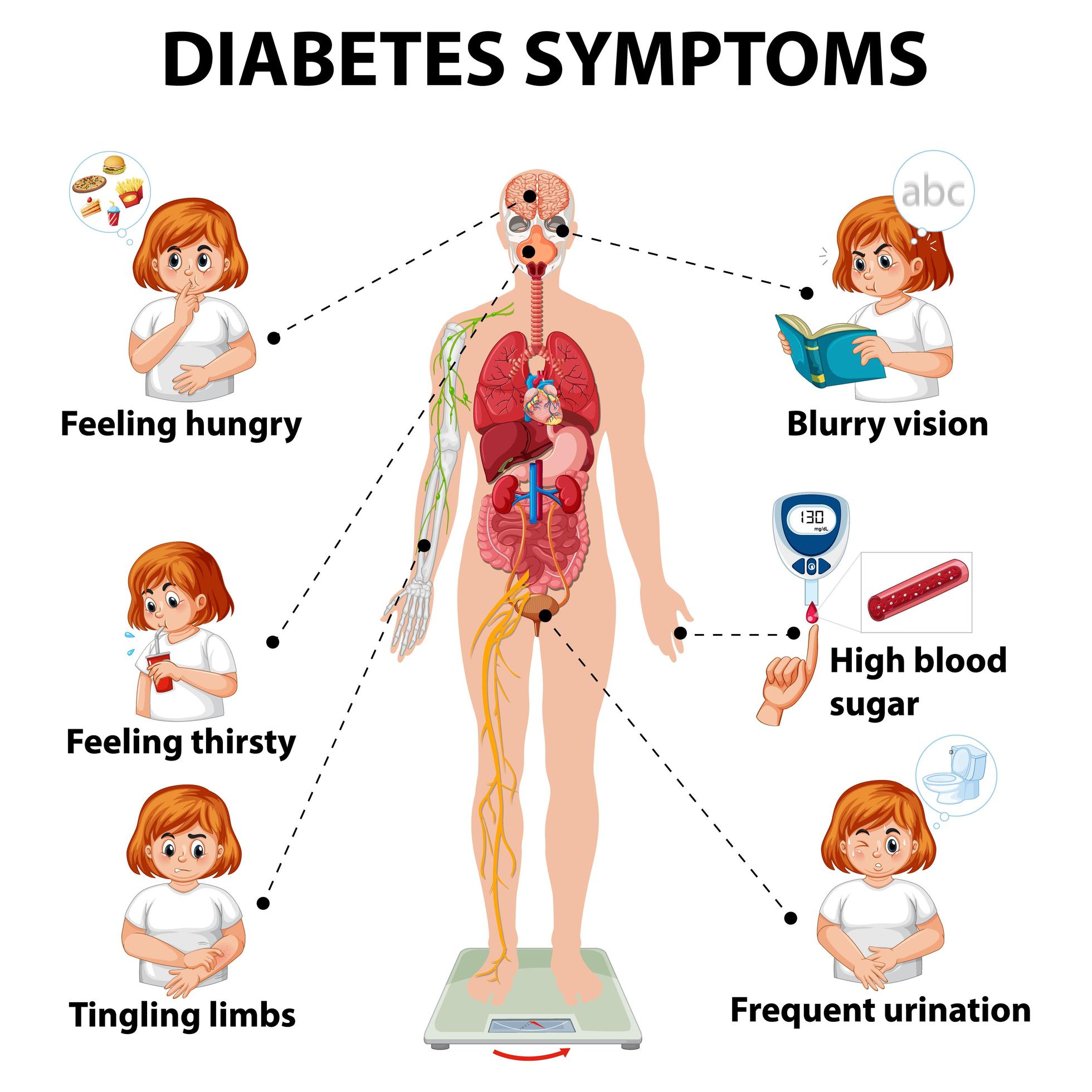
This glucose-rich environment acts as a nutrient broth for Candida species. Beyond simple nutrition, hyperglycemia impairs the host’s immune response, specifically hindering the ability of neutrophils (white blood cells that act as the first line of defense) to engulf and destroy fungal pathogens. This creates a selective pressure environment where Non-albicans Candida (NAC) species, which are often more resilient to environmental stress, can outcompete C. Albicans.
many of these NAC species are adept at forming biofilms. A biofilm is a protective, slimy layer that the fungi secrete to shield themselves from both the patient’s immune system and antifungal medications. This structural defense is a primary reason why candiduria in diabetic patients often becomes chronic.
Comparing the Pathogens: Albicans vs. Non-Albicans
The clinical challenge lies in the varying sensitivity profiles of these species. While C. Albicans generally responds well to azoles—a common class of antifungal drugs—many NAC species have evolved intrinsic resistance.
| Candida Species | Typical Prevalence | Fluconazole Sensitivity | Clinical Association |
|---|---|---|---|
| C. Albicans | Highest (Historically) | High | General population/Diabetics |
| C. Glabrata | Increasing | Low (Often Resistant) | Elderly/Long-term antifungal use |
| C. Tropicalis | Moderate | Moderate | Strongly linked to Diabetes |
| C. Parapsilosis | Low to Moderate | Moderate | Catheter-associated infections |
Global Health Implications and Regulatory Response
This shift in fungal epidemiology is not uniform globally, but it is a rising trend across major healthcare systems. In the United States, the CDC has flagged antifungal-resistant Candida auris as an urgent threat, a trend that mirrors the broader rise of NACs in diabetic populations. In Europe, the EMA has seen a corresponding increase in reports of azole resistance, prompting a push for more precise diagnostic stewardship.
The impact on patient access is significant. When first-line, affordable generics like fluconazole fail, patients must move to second-line therapies such as echinocandins. These drugs are significantly more expensive and often require intravenous administration, placing a heavier burden on the NHS in the UK and similar public health systems worldwide.
“The transition toward non-albicans species represents a significant challenge for empirical therapy. People can no longer afford to treat ‘yeast’ as a monolith; we must treat the specific species identified in the lab to prevent the acceleration of antifungal resistance,” says Dr. Aris Thorne, a leading mycologist specializing in opportunistic infections.
Regarding the transparency of this research, this cross-sectional study was funded through institutional academic grants. Notice no reported conflicts of interest from pharmaceutical entities, which reinforces the objectivity of the findings regarding drug resistance patterns.
Contraindications & When to Consult a Doctor
Antifungal treatments are not without risk. Patients should be aware of the following contraindications—conditions or factors that serve as a reason to withhold a certain treatment:
- Hepatic Impairment: Many azole antifungals are processed by the liver. Patients with pre-existing liver disease must be monitored closely for hepatotoxicity (liver damage).
- Drug Interactions: Antifungals can interfere with the metabolism of other critical medications, including certain blood thinners and statins.
- Pregnancy: Certain antifungal agents are contraindicated during the first trimester due to potential teratogenic effects (birth defects).
Seek immediate medical attention if you experience:
- High fever accompanying urinary urgency or burning.
- Lower back or flank pain (indicating the infection may have ascended to the kidneys).
- Confusion or extreme lethargy, which may signal systemic candidemia (fungus in the bloodstream).
The Path Forward: Precision Mycology
The emergence of NACs in diabetic patients underscores the necessity of “precision mycology.” We are moving away from the era of empirical prescribing—where a doctor guesses the pathogen—and toward a model of targeted therapy. For the diabetic patient, the most effective “antifungal” is often rigorous glycemic control. By reducing the glucose load in the urine, we eliminate the primary fuel source for these opportunistic fungi.
As we look toward 2027, the focus will likely shift toward the development of new antifungal classes that bypass the biofilm defenses of C. Glabrata and C. Tropicalis, ensuring that a manageable condition like diabetes does not lead to an untreatable infection.