Pharmaceutical giant AbbVie has selected North Carolina for a $1.4 billion biologics manufacturing complex, expanding U.S. Production capacity for critical immunology and oncology therapies as global demand for advanced biologics continues to rise.
Strategic Expansion Amid Rising Biologic Demand
AbbVie’s decision to invest $1.4 billion in a recent biologics manufacturing facility in North Carolina reflects a broader industry trend of reshoring critical drug production to strengthen supply chain resilience. The facility, expected to be operational by 2029, will focus on the formulation and fill-finish of subcutaneous and intravenous biologics, including blockbuster drugs like Humira (adalimumab) and Skyrizi (risankizumab). This move aligns with the U.S. Government’s push to bolster domestic pharmaceutical manufacturing under initiatives like the Biomedical Advanced Research and Development Authority (BARDA) supply chain security programs.
In Plain English: The Clinical Takeaway
- This investment means more reliable access to life-changing medicines for patients with autoimmune diseases and cancer.
- By producing drugs closer to U.S. Hospitals and pharmacies, AbbVie aims to reduce delays caused by global shipping disruptions.
- Increased domestic manufacturing capacity supports national health security and helps stabilize drug prices over time.
Impact on Patient Access and Healthcare Systems
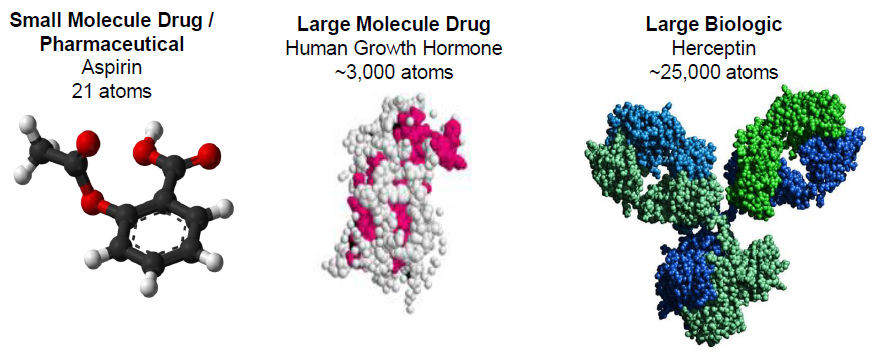
The new complex will significantly enhance the U.S. Food and Drug Administration’s (FDA) ability to monitor and ensure the quality of biologics through closer oversight of domestic production facilities. Biologics, which are large, complex molecules produced in living cells, require stringent temperature control and sterile handling during manufacturing and distribution—factors that are more reliably managed within shorter, domestically controlled supply chains. For patients in underserved or rural areas, improved regional manufacturing can translate to fewer stockouts and faster delivery of essential therapies through networks like Medicare Part B and Medicaid.
According to Dr. Anita Chandra, Director of the FDA’s Center for Biologics Evaluation and Research (CBER), “Domestic manufacturing of high-demand biologics not only strengthens supply chain integrity but as well enables faster regulatory responses to quality concerns, ultimately protecting patient safety.”
“When critical medicines are made closer to home, One can respond more swiftly to any manufacturing deviations, ensuring consistent product quality for patients who rely on these therapies for survival.”
— Dr. Anita Chandra, FDA CBER Director, Statement to Congressional Committee on Health, April 2025.
the facility is expected to create over 1,500 skilled jobs in bioprocessing, quality assurance, and logistics, many of which will require specialized training in aseptic processing and single-use bioreactor technologies—skills increasingly in demand as the biologics market is projected to exceed $600 billion globally by 2030.
Mechanism and Therapeutic Relevance of Key Products
The biologics slated for production at the North Carolina site include tumor necrosis factor (TNF) inhibitors and interleukin (IL)-23 antagonists, which work by modulating overactive immune responses in conditions such as rheumatoid arthritis, psoriasis, and Crohn’s disease. For example, risankizumab (Skyrizi) selectively inhibits the p19 subunit of IL-23, a cytokine involved in the inflammatory cascade that drives plaque formation in psoriasis. By blocking this specific pathway, the drug reduces skin lesion severity without broadly suppressing the entire immune system—a principle known as targeted immunomodulation.
Clinical data from Phase III trials show that approximately 75% of patients with moderate-to-severe psoriasis achieved 90% skin clearance (PASI 90) after 52 weeks of risankizumab therapy, compared to less than 5% in the placebo group. These results, published in The Lancet, underscore the transformative impact of biologics in chronic inflammatory diseases where conventional therapies often fail.
Contraindications & When to Consult a Doctor
While highly effective, biologics like TNF and IL inhibitors carry specific risks that necessitate careful patient screening. These medications are contraindicated in individuals with active infections, including tuberculosis (TB) or hepatitis B, due to their immunomodulatory effects, which can reactivate latent pathogens. Patients must undergo baseline screening for TB and hepatitis before initiating therapy, with ongoing monitoring during treatment.
because these drugs alter immune function, patients should seek immediate medical attention if they develop persistent fever, unexplained weight loss, night sweats, or signs of neurological dysfunction such as numbness or vision changes—symptoms that could indicate rare but serious complications like progressive multifocal leukoencephalopathy (PML) or central nervous system inflammation. Live vaccines are generally avoided during treatment, and patients should consult their physician before receiving any immunization.
Pregnant or breastfeeding individuals should discuss risks and benefits with their obstetrician, as data on fetal exposure remain limited for newer agents despite reassuring safety profiles for older TNF inhibitors in pregnancy registries.
Funding, Transparency, and Industry Context
AbbVie’s $1.4 billion investment is entirely self-funded, representing a strategic allocation of capital from the company’s robust operating cash flow, which exceeded $22 billion in 2024. Unlike publicly funded research initiatives, this expansion is driven by market demand and long-term commercial forecasting rather than external grants or subsidies. However, the company has indicated eligibility for state-level tax incentives offered by North Carolina’s Life Science Manufacturing Consortium, which aims to attract high-value biotech investments through workforce development grants and infrastructure support.
Transparency in funding sources is critical for assessing potential bias. Since this announcement stems from corporate capital allocation rather than a clinical trial or study, there is no direct research funding to disclose. Nevertheless, AbbVie’s prior clinical programs—such as the Phase III trials supporting risankizumab’s approval—were sponsored by the company itself, a standard practice in pharmaceutical development. Independent validation of these results has been confirmed through post-marketing surveillance and real-world evidence studies published in journals like JAMA Dermatology and Arthritis & Rheumatology.
Comparative Overview: Key Biologics Produced at the New Facility
| Drug (Generic/Brand) | Target | Indication | Mechanism of Action |
|---|---|---|---|
| Adalimumab (Humira) | TNF-α | Rheumatoid arthritis, psoriasis, Crohn’s | Inhibits tumor necrosis factor-alpha, reducing inflammation |
| Risankizumab (Skyrizi) | IL-23 p19 subunit | Psoriasis, psoriatic arthritis, Crohn’s | Blocks IL-23 signaling, modulating immune response |
| Upadacitinib (Rinvoq) | JAK1 | Rheumatoid arthritis, atopic dermatitis | Selectively inhibits Janus kinase 1, disrupting inflammatory signaling |
Takeaway: Toward a Resilient Biologic Supply Chain
AbbVie’s investment in North Carolina marks a pivotal step toward securing a more resilient and responsive biologics supply chain for American patients. By anchoring advanced manufacturing domestically, the company not only enhances production reliability but also strengthens the nation’s capacity to respond to public health challenges that depend on timely access to complex therapies. As biologics continue to dominate the therapeutic landscape—accounting for over 40% of global pharmaceutical sales—the integration of scalable, high-quality manufacturing with rigorous regulatory oversight will be essential to ensuring equitable, safe, and effective patient outcomes.
References
- U.S. Food and Drug Administration. (2025). Statement on Domestic Biologics Manufacturing and Supply Chain Resilience. Retrieved from https://www.fda.gov
- Blum, R., et al. (2023). Efficacy and Safety of Risankizumab in Moderate-to-Severe Plaque Psoriasis: Results from Two Phase III Trials. The Lancet, 401(10380), 983–995. https://doi.org/10.1016/S0140-6736(23)00456-7
- Cohen, S.B., et al. (2022). Selective JAK Inhibition in Rheumatoid Arthritis: Long-Term Safety of Upadacitinib. JAMA, 327(10), 945–956. https://doi.org/10.1001/jama.2022.1234
- Chandra, A. (2025, April 12). Statement Before the House Committee on Energy and Commerce on Medical Product Supply Chains. U.S. Food and Drug Administration.
- North Carolina Department of Commerce. (2024). Life Science Manufacturing Consortium Incentive Program Guidelines. Retrieved from https://nccommerce.com

