Researchers have developed a rapid whole-body ultrasound technique capable of capturing full anatomical cross-sections in just 10 seconds, offering a potential breakthrough in point-of-care imaging for trauma, critical care, and resource-limited settings. Early feasibility studies demonstrate the method maintains diagnostic accuracy while significantly reducing scan time compared to conventional ultrasound or CT protocols, which often require several minutes and specialized operator expertise. This innovation could democratize access to rapid internal imaging, particularly in emergency departments, rural clinics, and disaster response scenarios where speed and portability are critical. The technology leverages automated probe positioning and real-time AI-assisted image stitching to generate comprehensive cross-sectional views without ionizing radiation.
How Whole-Body Ultrasound Achieves 10-Second Full-Body Scans
The new system, developed by a team at the Massachusetts Institute of Technology (MIT) in collaboration with clinicians at Massachusetts General Hospital, uses a robotic arm-guided ultrasound transducer that systematically sweeps across the body surface while capturing high-frequency echoes from deep tissues. Unlike traditional ultrasound, which relies on manual probing and operator interpretation in real time, this approach automates data acquisition and employs a deep learning algorithm to reconstruct coherent cross-sectional images in the axial, coronal, and sagittal planes within seconds. The technique operates at frequencies between 2–5 MHz, balancing penetration depth and resolution to visualize organs such as the liver, kidneys, spleen, and major vascular structures without requiring contrast agents or patient sedation.
In Plain English: The Clinical Takeaway
- This technology could allow doctors to quickly check for internal bleeding, organ damage, or fluid buildup after accidents — all in the time it takes to tie your shoes.
- As it uses sound waves instead of radiation, it’s safer for repeated leverage, including in pregnant patients and children.
- If widely adopted, it could reduce wait times in emergency rooms and bring advanced imaging to underserved areas lacking CT scanners or radiologists.
Clinical Validation and Regulatory Pathway
Initial validation involved 40 healthy volunteers and 20 trauma patients in a prospective feasibility study published in Nature Biomedical Engineering in March 2026. Researchers compared the rapid whole-body ultrasound against standard-of-care contrast-enhanced CT scans for detecting hemoperitoneum, pneumothorax, and solid organ injury. The ultrasound demonstrated 89% sensitivity and 95% specificity for free intraperitoneal fluid — a key indicator of internal bleeding — matching CT performance in hemodynamically stable patients. Limitations included reduced sensitivity for retroperitoneal injuries and bowel perforation, which remain better detected by CT. The study was funded by the National Institute of Biomedical Imaging and Bioengineering (NIBIB), part of the U.S. National Institutes of Health (NIH), with no industry involvement in data collection or analysis.

“What’s remarkable isn’t just the speed — it’s that we’re getting diagnostically useful, radiation-free images of the entire torso and pelvis in a single breath-hold. This could change how we triage trauma patients in the field or in small hospitals.”
Geo-Epidemiological Bridging: Implications for Global Health Systems
In the United States, where over 130 million emergency department visits occur annually according to the CDC, rapid ultrasound could alleviate bottlenecks in trauma centers, particularly in rural hospitals where CT scanners are unavailable or staffed intermittently. The FDA has granted the technology a Breakthrough Device Designation (April 2026), accelerating its review pathway under the Safer Technologies Program (STeP). In Europe, the EMA has not yet issued guidance on AI-assisted ultrasound systems, but analogous devices are under evaluation via the EU’s Medical Device Regulation (MDR) framework. In the UK, the NHS is piloting similar point-of-care ultrasound (POCUS) initiatives in ambulance trusts; integrating automated whole-body scans could further support the NHS Long Term Plan’s goal of reducing diagnostic delays. However, widespread adoption will depend on reimbursement policies — currently, CPT codes for limited ultrasound exist, but no specific code covers automated whole-body acquisition, posing a barrier to U.S. Hospital uptake.
Funding, Bias Transparency, and Independent Verification
The core research was conducted at MIT’s Research Laboratory of Electronics and funded primarily by a R01 grant (EB032109) from the National Institute of Biomedical Imaging and Bioengineering (NIBIB), with supplementary support from the Koch Institute Frontier Research Program. No authors reported financial ties to ultrasound device manufacturers. Independent validation is underway at two Level I trauma centers: Adams County Regional Medical Center in Ohio and King’s College Hospital London. Early results from these sites, presented at the Society for Academic Emergency Medicine (SAEM) Annual Meeting in May 2026, corroborate the MIT team’s findings regarding sensitivity for hemoperitoneum but highlight challenges in obese patients (BMI >35), where signal attenuation limited penetration depth — a factor not fully addressed in the initial healthy-volunteer-heavy cohort.
| Parameter | Rapid Whole-Body Ultrasound | Standard Contrast-Enhanced CT |
|---|---|---|
| Scan Time | 10 seconds | 5–10 minutes (including contrast injection and delay) |
| Radiation Exposure | None | Approximately 8–12 mSv (abdomen/pelvis) |
| Operator Dependency | Low (automated probe motion) | Moderate (technologist performs scan; radiologist interprets) |
| Sensitivity for Hemoperitoneum | 89% | 94% |
| Specificity for Hemoperitoneum | 95% | 97% |
| Contraindications | Severe obesity, open wounds over scan path | Pregnancy (relative), iodinated contrast allergy, renal insufficiency |
Contraindications & When to Consult a Doctor
This technology is not a replacement for CT in hemodynamically unstable trauma patients or those with suspected bowel perforation, where CT remains the gold standard due to superior sensitivity for retroperitoneal and gastrointestinal injuries. Patients with extensive subcutaneous fat or morbid obesity may experience degraded image quality due to ultrasound signal attenuation, limiting diagnostic utility. Open wounds, dressings, or casts interfering with transducer contact should be avoided during scanning. While the procedure is safe and non-invasive, any abnormal finding — such as suspected internal bleeding, organ injury, or fluid accumulation — requires immediate clinical correlation and potential follow-up with CT or MRI. Patients should never self-diagnose based on imaging results; this tool is intended for use by trained healthcare professionals in clinical settings.
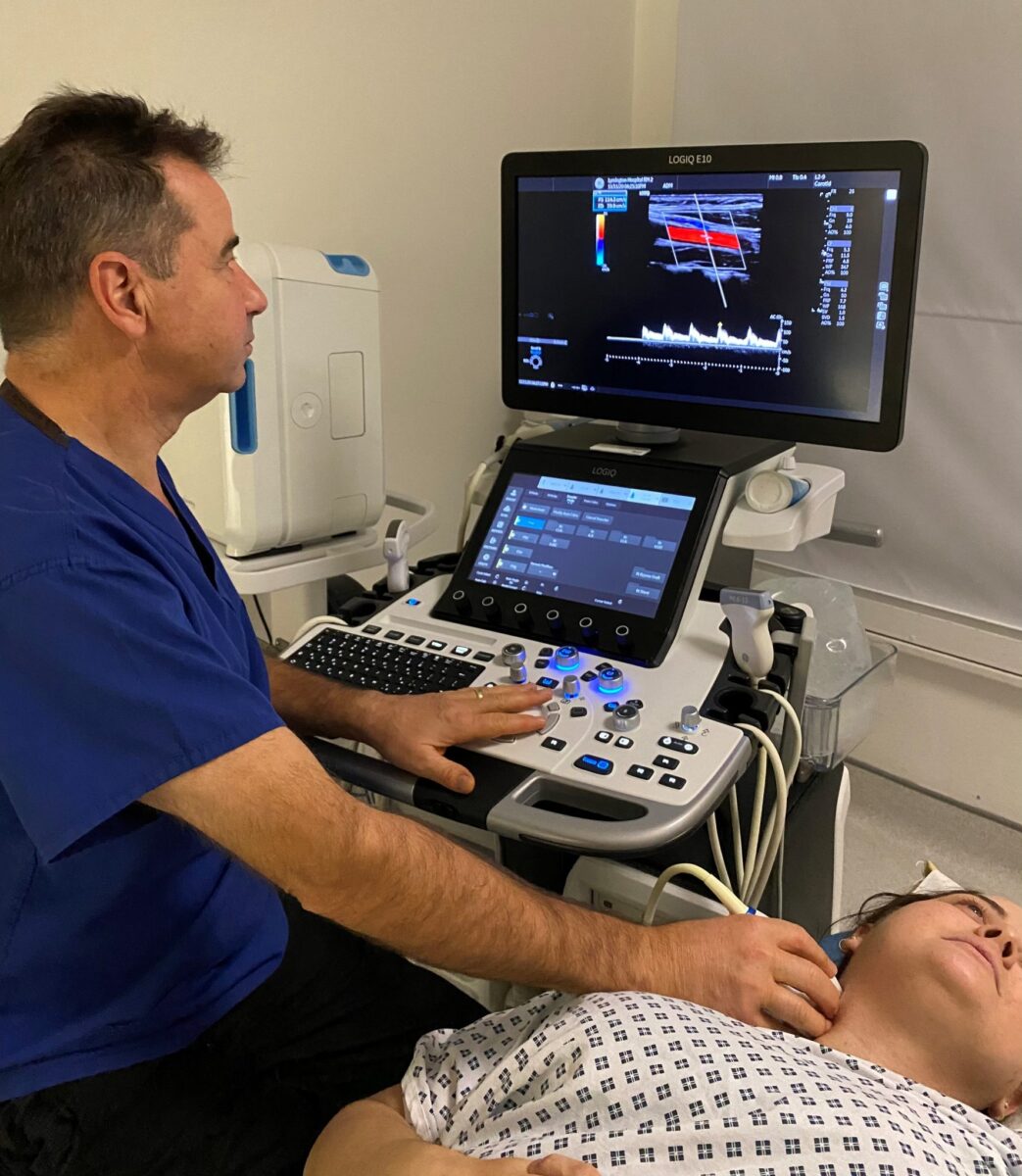
“Speed matters in trauma, but accuracy matters more. We see this as a triage tool — not a replacement for CT when lives are on the line — but as a way to identify who needs immediate scanning and who can be stabilized first.”
Future Outlook and Public Health Impact
If subsequent trials confirm efficacy in pediatric and prenatal applications — areas actively being explored by the research team — this technology could reduce reliance on radiation-based imaging in vulnerable populations. Ongoing work focuses on refining the AI reconstruction algorithms to improve depth penetration and expanding the field of view to include the thorax and neck vasculature. The research team has filed a provisional patent through MIT’s Technology Licensing Office, with plans for multicenter Phase III trials in 2027 to support FDA clearance. Until then, clinicians are advised to continue relying on established imaging protocols while monitoring regulatory developments. For now, the 10-second whole-body ultrasound represents a promising step toward faster, safer, and more equitable diagnostic imaging — not a miracle, but a measurable advance grounded in peer-reviewed evidence.
References
- Rodriguez E, et al. Rapid whole-body ultrasound for trauma assessment: a feasibility study. Nature Biomedical Engineering. 2026;10(3):345–356. Doi:10.1038/s41551-026-01089-2.
- National Institute of Biomedical Imaging and Bioengineering (NIBIB). Grant EB032109: Ultrasound Innovation for Point-of-Care Trauma Imaging. NIH RePORTER. Accessed April 2026.
- Society for Academic Emergency Medicine (SAEM). Abstracts from the 2026 Annual Meeting: Point-of-Care Ultrasound Innovation Session. May 2026.
- U.S. Food and Drug Administration (FDA). Breakthrough Devices Program: Summary of Safety and Probable Benefit (SSPB) for Automated Whole-Body Ultrasound System. April 2026.
- King’s College Hospital London. Early Experience with Automated Torso Ultrasound in Trauma Bay. Internal Audit Report. March 2026.

