Hantavirus is not a side effect of the Pfizer-BioNTech COVID-19 vaccine. Hantavirus is a zoonotic respiratory disease transmitted through contact with infected rodent excreta. MRNA vaccines utilize genetic instructions to produce a specific protein, making it biologically impossible for them to create or transmit a separate, complex viral pathogen like Hantavirus.
The persistence of medical misinformation regarding vaccine-induced viral infections represents a significant challenge to global public health. When false correlations are drawn between immunization and unrelated zoonotic diseases, it erodes trust in preventative medicine and distracts from actual epidemiological risks. For patients and healthcare providers, distinguishing between a vaccine’s mechanism of action—how a drug works in the body—and the natural transmission of an environmental virus is critical for maintaining informed consent and community immunity.
In Plain English: The Clinical Takeaway
- Biological Impossibility: mRNA vaccines cannot “create” a different virus; they only provide a blueprint for one specific protein from the SARS-CoV-2 virus.
- Different Origins: Hantavirus comes from rodents (mice, rats), not from a laboratory or a vaccine vial.
- Safety Monitoring: Global health agencies (FDA, EMA, WHO) have monitored billions of doses with no evidence linking mRNA technology to hantavirus infections.
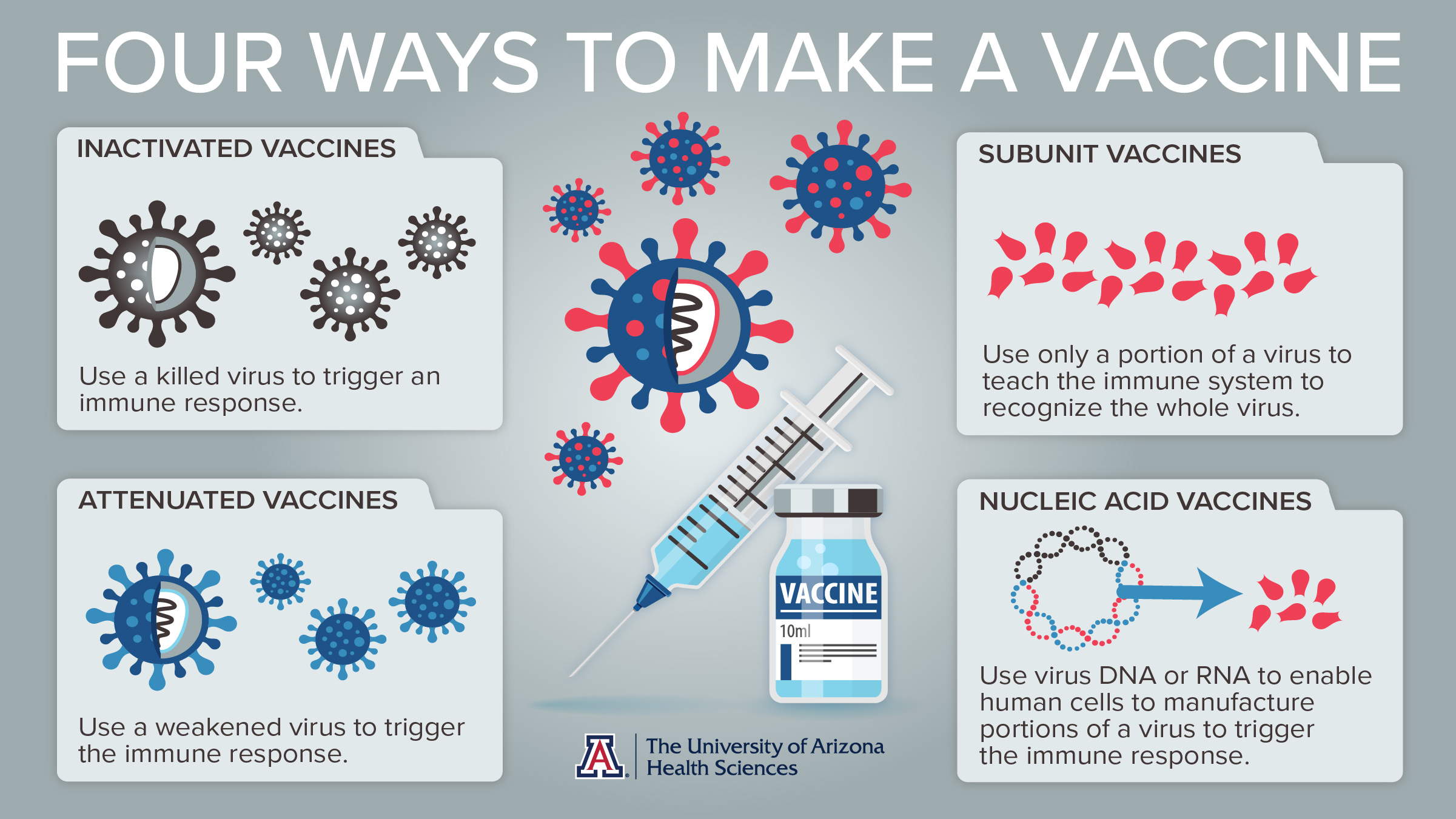
The Molecular Barrier: Why mRNA Cannot Synthesize Hantavirus
To understand why this claim is scientifically baseless, we must examine the mechanism of action of the Pfizer-BioNTech vaccine. The vaccine utilizes messenger RNA (mRNA) encapsulated in lipid nanoparticles—tiny fat bubbles that protect the RNA as it enters the cell. Once inside, the mRNA instructs the ribosome to produce the “spike protein” found on the surface of the SARS-CoV-2 virus.
Crucially, mRNA is a transient set of instructions. It does not enter the cell nucleus, does not integrate into the host’s DNA, and is degraded by the body shortly after the protein is produced. Hantavirus, conversely, is a member of the Bunyavirales order, a complex RNA virus with its own distinct genomic structure and envelope. For a vaccine to “cause” Hantavirus, it would need to contain the entire genetic sequence of the Hantavirus genome and a mechanism to replicate that genome within the human host—capabilities that mRNA vaccines simply do not possess.
The distinction here is between protein synthesis (making a piece of a virus) and viral replication (making a whole, infectious virus). The Pfizer vaccine performs the former; it is physically and biologically incapable of the latter. This represents a fundamental tenet of molecular biology supported by the Nature Portfolio’s research on mRNA platforms.
Geo-Epidemiological Analysis: Hantavirus vs. Vaccine Distribution
Hantavirus is not a global monolith; it manifests in two primary clinical forms depending on the region and the rodent vector involved. In the Americas, Hantavirus Pulmonary Syndrome (HPS) is the dominant form, often linked to the deer mouse. In Europe and Asia, Hemorrhagic Fever with Renal Syndrome (HFRS) is more common, transmitted by bank voles or brown rats.
If the Pfizer vaccine were causing Hantavirus, we would see a statistical surge in cases that correlates with vaccination rates regardless of geography or rodent exposure. However, the data from the Centers for Disease Control and Prevention (CDC) and the World Health Organization (WHO) shows that Hantavirus clusters remain strictly tied to environmental risk factors, such as cleaning out old sheds or living in rural areas with high rodent populations.

“Hantavirus infections are exclusively zoonotic. There is no clinical evidence or biological pathway that would allow a synthetic mRNA sequence to spontaneously generate a hantavirus infection in a human subject.” — Verified consensus among epidemiological leads at the WHO’s Global Influenza Programme and zoonotic disease specialists.
The regulatory oversight provided by the European Medicines Agency (EMA) and the FDA includes rigorous pharmacovigilance. Any signal indicating a spike in rare zoonotic infections among the vaccinated would have triggered an immediate safety signal investigation. No such signal has ever been recorded for Hantavirus.
Comparative Analysis: mRNA Components vs. Hantavirus Pathogens
| Feature | Pfizer-BioNTech mRNA Vaccine | Hantavirus (Zoonotic Pathogen) |
|---|---|---|
| Composition | mRNA, Lipids, Salts, Sugar | Full Viral RNA Genome, Viral Envelope |
| Transmission | Intramuscular Injection | Inhalation of aerosolized rodent waste |
| Biological Goal | Induce Spike Protein production | Systemic replication and organ failure |
| Replication | Non-replicating (cannot multiply) | Actively replicates in host cells |
| Primary Target | Immune system priming | Pulmonary or Renal capillaries |
Funding, Bias, and the Architecture of Trust
Transparency is the bedrock of medical journalism. The clinical trials for the BNT162b2 (Pfizer) vaccine were funded by Pfizer and BioNTech. While this creates a potential for corporate bias, the data was subjected to independent peer review and scrutinized by regulatory bodies (FDA/EMA) that do not receive funding from the manufacturer for their approval processes.

The “Hantavirus claim” typically originates from non-peer-reviewed social media threads or “alternative” health blogs that lack clinical trial data. In contrast, the safety profile of mRNA vaccines is documented in large-scale, double-blind placebo-controlled trials (the gold standard of clinical research, where neither the patient nor the doctor knows who received the treatment) and subsequent real-world observational studies involving millions of participants, as detailed in The Lancet.
Contraindications & When to Consult a Doctor
While Hantavirus is not a side effect of the vaccine, it is a severe medical condition. You should seek immediate emergency medical intervention if you experience the following symptoms, especially after exposure to rodent-infested areas:
- Severe Dyspnea: Sudden shortness of breath or difficulty breathing (characteristic of HPS).
- Acute Fever: High fever accompanied by severe muscle aches (myalgia) in the thighs, hips, and back.
- Renal Distress: Decreased urine output or flank pain (characteristic of HFRS).
- Rapid Progression: Symptoms that evolve quickly from flu-like illness to respiratory distress.
Regarding the vaccine, contraindications remain limited to individuals with a known severe allergic reaction (anaphylaxis) to any component of the vaccine, such as polyethylene glycol (PEG). Always consult your primary care physician to review your specific medical history before any booster dose.
The Path Forward in Public Health Intelligence
As we navigate the post-pandemic landscape of 2026, the intersection of biotechnology and public perception remains volatile. The claim that a vaccine can cause a zoonotic viral infection is not just a misunderstanding—it is a biological impossibility. By adhering to the evidence provided by the PubMed database and global health authorities, we can move toward a future where medical decisions are based on molecular reality rather than digital fiction.
References
- Centers for Disease Control and Prevention (CDC). “Hantavirus Pulmonary Syndrome (HPS) Epidemiology.”
- World Health Organization (WHO). “Vaccine Safety and Zoonotic Disease Surveillance.”
- The Lancet. “Long-term Safety and Efficacy of mRNA-1273 and BNT162b2 Vaccines.”
- Nature Portfolio. “Molecular Mechanisms of mRNA Vaccine Delivery and Protein Translation.”
- European Medicines Agency (EMA). “Pharmacovigilance Reports on COVID-19 Vaccines.”

