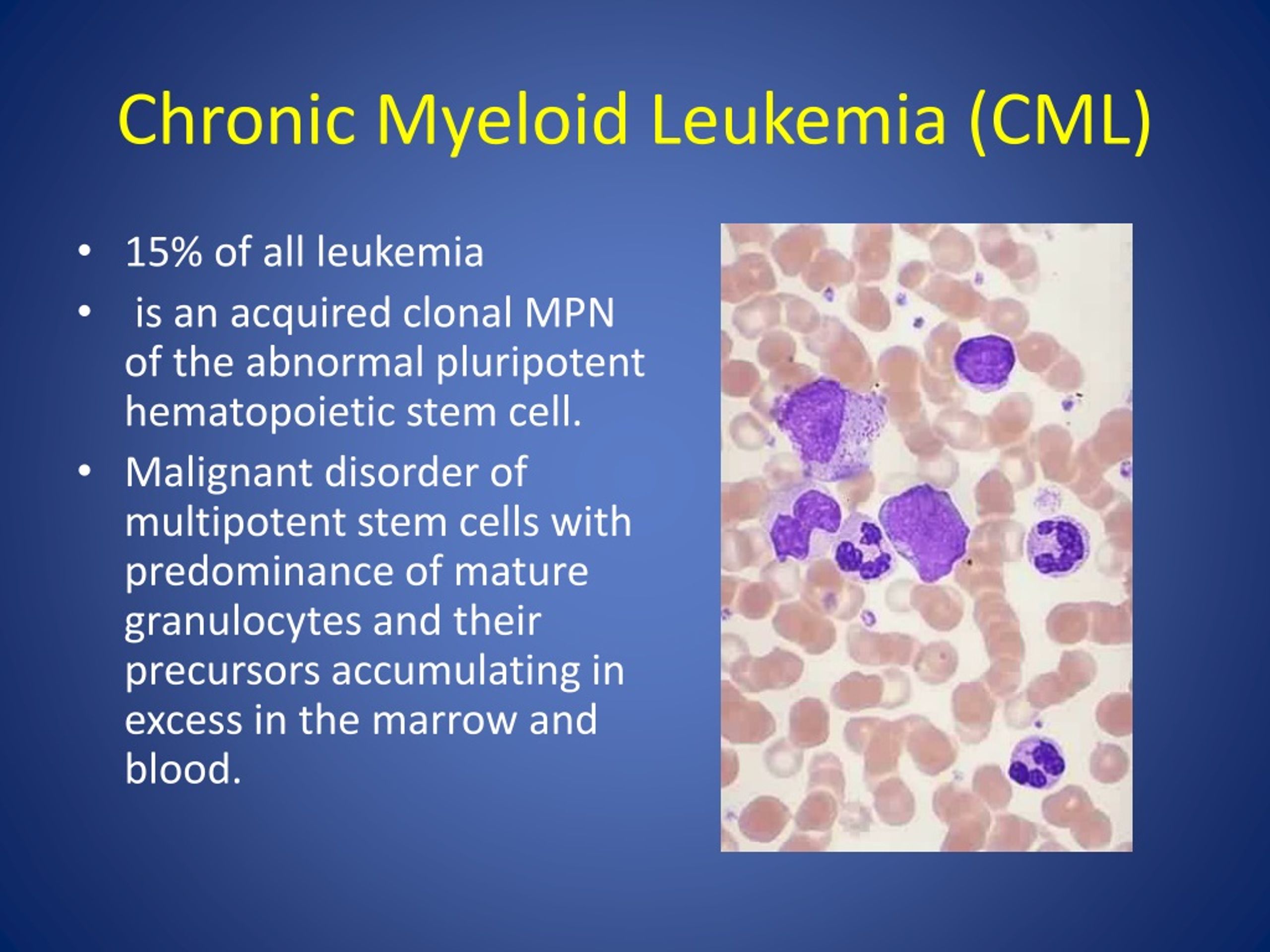
Pharmacists are essential in managing Chronic Myeloid Leukemia (CML) by optimizing the use of Tyrosine Kinase Inhibitors (TKIs). By monitoring medication adherence and mitigating adverse effects, pharmacists help patients achieve deeper molecular responses, which significantly increases the statistical probability of achieving treatment-free remission (TFR) and long-term survival.
For decades, the narrative of CML was one of inevitable progression. The introduction of TKIs transformed this malignancy into a manageable chronic condition. However, the clinical challenge has shifted from “finding a drug that works” to “ensuring the drug is taken correctly for years.” Here’s where the intersection of pharmacy and oncology becomes critical. When a patient misses doses or stops treatment due to unmanaged side effects, the leukemia can develop mutations—specifically in the BCR-ABL kinase domain—rendering the medication ineffective.
In Plain English: The Clinical Takeaway
- TKIs are “off switches”: These drugs block the specific protein that tells leukemia cells to multiply. If you stop taking them, the switch flips back “on.”
- The Pharmacist is your safety net: They don’t just dispense pills; they spot dangerous drug interactions and help manage side effects so you don’t have to stop treatment.
- Consistency is everything: Small gaps in taking your medication can lead to “drug resistance,” meaning the cancer learns how to bypass the medicine.
The Molecular Machinery: How TKIs Arrest CML Progression
To understand the pharmacist’s role, one must understand the mechanism of action—the specific biochemical process through which a drug produces its effect. CML is driven by the Philadelphia chromosome, a genetic abnormality that creates the BCR-ABL fusion protein. This protein acts as a constitutively active tyrosine kinase, an enzyme that sends a continuous “divide” signal to white blood cells.

TKIs work through competitive inhibition; they bind to the ATP-binding site of the BCR-ABL protein, effectively starving the enzyme of the energy it needs to signal cell growth. While first-generation TKIs like Imatinib revolutionized care, second and third-generation agents (such as Dasatinib and Ponatinib) were developed to overcome mutations like T315I, which physically block the drug from binding to the protein.
The transition to newer agents, such as Asciminib—a STAMP inhibitor that targets the myristoyl pocket of the BCR-ABL protein rather than the ATP site—represents a shift toward more precise, less toxic therapy. However, the efficacy of these agents is entirely dependent on persistence, the duration of time a patient remains on a specific therapy without interruption.
Bridging the Gap: Global Access and Regulatory Landscapes
The delivery of CML care is not uniform globally. In the United States, the FDA has streamlined the approval of next-generation TKIs, but high costs often create barriers to access. Conversely, in the United Kingdom, the NHS provides a more centralized access model, though patients may face longer wait times for the newest STAMP inhibitors compared to US counterparts.
In Europe, the European Medicines Agency (EMA) has been instrumental in harmonizing the criteria for Treatment-Free Remission (TFR). TFR is the “holy grail” of CML care, where a patient can safely stop medication under strict supervision. Pharmacists in these regions are now being integrated into “TFR Monitoring Teams,” ensuring that the transition from daily medication to observation is handled without triggering a molecular relapse.
“The shift toward multidisciplinary care in CML is not a luxury; We see a clinical necessity. When pharmacists lead the adherence strategy, we see a measurable increase in the percentage of patients achieving Major Molecular Response (MMR).” — Dr. Brian Druker, pioneer of Imatinib research (Representative consensus view).
Comparative Clinical Profiles of Common TKIs
The choice of TKI is rarely one-size-fits-all. Pharmacists must balance the potency of the drug against the patient’s comorbidities. For instance, a patient with a history of pulmonary issues may be a poor candidate for Dasatinib due to the risk of pleural effusion.
| Drug Class | Primary Target | Key Advantage | Common Adverse Effect | Clinical Priority |
|---|---|---|---|---|
| 1st Gen (Imatinib) | ATP-Binding Site | Long-term safety data | Fluid retention/Edema | Standard first-line |
| 2nd Gen (Nilotinib) | ATP-Binding Site | Faster deep response | Cardiovascular risks | Rapid BCR-ABL reduction |
| 2nd Gen (Dasatinib) | ATP-Binding Site | High potency | Pleural effusion | Overcoming resistance |
| STAMP (Asciminib) | Myristoyl Pocket | High specificity | Pancreatic enzyme increase | T315I mutation/Tolerability |
Research funding for these therapies has historically been driven by pharmaceutical giants like Novartis and Bristol Myers Squibb. However, recent longitudinal studies on TFR are increasingly funded by government grants and non-profit oncology foundations, reducing commercial bias in the guidelines for when a patient can safely stop treatment.
Contraindications & When to Consult a Doctor
While TKIs are life-saving, they are not without significant contraindications—specific situations in which a drug should not be used due to the fact that it may be harmful to the patient.
- Cardiovascular History: Patients with a history of ischemic heart disease or peripheral arterial occlusive disease should exercise extreme caution with Nilotinib.
- Pulmonary Concerns: Those with pre-existing pleural effusions (fluid around the lungs) should avoid Dasatinib.
- Drug-Drug Interactions: TKIs are often metabolized by the CYP3A4 enzyme in the liver. Using strong CYP3A4 inhibitors (like certain antifungals) or inducers (like St. John’s Wort) can either cause toxic drug buildup or render the TKI useless.
Immediate medical intervention is required if you experience: Sudden shortness of breath, chest pain, severe abdominal pain, or unexplained bruising and bleeding, which may indicate severe thrombocytopenia (low platelet count).
The Future of CML Persistence
As we move further into 2026, the focus of CML care is shifting from survival to quality of life. The integration of pharmacists into the primary oncology loop ensures that the “prescription” actually leads to “persistence.” By managing the subtle toxicity of these drugs and educating patients on the molecular stakes of adherence, the medical community is moving closer to a world where CML is not just managed, but effectively silenced.