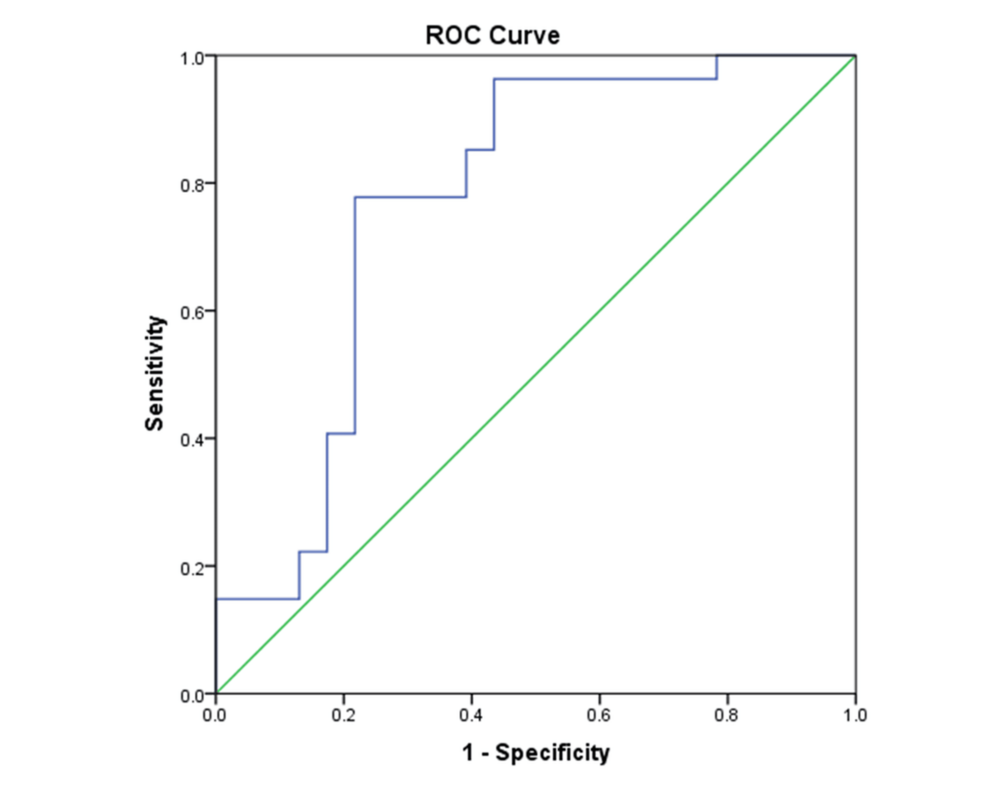
A recent cross-sectional study published in Cureus demonstrates that measuring pleural fluid lactate and the pleural fluid-to-serum lactate ratio effectively differentiates the causes of exudative pleural effusions. By analyzing these metabolic markers, clinicians can more accurately distinguish between parapneumonic effusions, malignancy, and tuberculosis, streamlining patient diagnosis.
For the millions of patients worldwide presenting with pleural effusions—the abnormal buildup of fluid between the layers of the pleura enclosing the lungs—the diagnostic journey is often fraught with ambiguity. Whereas clinicians traditionally rely on Light’s criteria to categorize fluid as an exudate (fluid rich in proteins and cells), the specific cause often remains elusive, requiring invasive biopsies or delayed culture results.
The utility of lactate as a biomarker represents a shift toward metabolic profiling. Lactate is a byproduct of anaerobic glycolysis—the process where cells produce energy without sufficient oxygen. In the pleural space, elevated lactate levels typically signal severe inflammation, tissue hypoxia, or the metabolic demands of malignant cells, providing a chemical “fingerprint” of the underlying pathology.
In Plain English: The Clinical Takeaway
- Faster Answers: Using lactate ratios can help doctors tell the difference between a lung infection and lung cancer more quickly than some traditional tests.
- Less Guesswork: It reduces the need for repeated, invasive needle procedures by providing a clearer chemical profile of the lung fluid.
- Targeted Treatment: By identifying the cause sooner, patients can start the correct medication (like antibiotics for pneumonia or chemotherapy for cancer) faster.
The Metabolic Mechanism: Why Lactate Signals Pathology
To understand the diagnostic utility of these markers, we must examine the mechanism of action regarding lactate production. In a healthy state, pyruvate is converted to acetyl-CoA for aerobic respiration. However, in the presence of severe infection or malignancy, the microenvironment becomes hypoxic (oxygen-deprived).
In parapneumonic effusions—fluid resulting from pneumonia—bacteria and infiltrating leukocytes (white blood cells) consume oxygen rapidly, triggering a surge in lactate production. In contrast, malignant effusions exhibit elevated lactate due to the “Warburg Effect,” where cancer cells prioritize glycolysis even in the presence of oxygen to fuel rapid growth.
The “Pleural Fluid-to-Serum Lactate Ratio” is a critical refinement. By comparing the lactate in the chest fluid to the lactate in the bloodstream, clinicians can determine if the lactate is being produced locally within the pleural space or if We see simply reflecting a systemic issue, such as sepsis or liver failure. A high ratio strongly suggests a localized pleural pathology.
Global Clinical Application and Healthcare Integration
The implementation of lactate profiling varies significantly across global healthcare systems. In the United States, where the FDA emphasizes rapid diagnostics to reduce hospital stays, the integration of bedside lactate analyzers (Point-of-Care Testing) could significantly reduce the “time-to-diagnosis” for exudative effusions.
In the United Kingdom, the NHS focuses heavily on cost-effective pathways. Utilizing a lactate ratio as a first-line screening tool before proceeding to more expensive pleural biopsies could reduce the burden on thoracic surgery departments. Similarly, in regions where tuberculosis (TB) remains endemic, such as Southeast Asia and Africa, differentiating TB from malignancy is a critical public health priority.
However, the lack of a standardized “cutoff” value for lactate across different laboratories remains a hurdle. For this to become a gold standard, international consensus from bodies like the World Health Organization (WHO) or the American Thoracic Society is required to ensure a ratio of 2.0 in one hospital means the same thing in another.
| Etiology | Typical Lactate Level | Fluid-to-Serum Ratio | Primary Driver |
|---|---|---|---|
| Parapneumonic Effusion | High | Significantly Elevated | Bacterial Hypoxia |
| Malignant Effusion | Moderate to High | Elevated | Warburg Effect |
| Tuberculous Effusion | Low to Moderate | Mildly Elevated | Chronic Inflammation |
| Transudative (Heart Failure) | Low | Near 1.0 | Systemic Pressure |
Funding, Bias, and the Evidence Gap
The study in Cureus is an observational cross-sectional analysis. While these provide vital “proof of concept,” they are susceptible to selection bias. The research was conducted as an academic endeavor; however, it is imperative to note that no pharmaceutical funding was involved, which increases the objectivity of the findings.
The “Information Gap” in current research lies in the longitudinal outcome. We grasp lactate helps diagnose, but we do not yet know if lactate levels can predict how a patient will respond to treatment. For instance, does a plummeting lactate level after 48 hours of antibiotics signal a successful recovery, or is it an irrelevant fluctuation?
“The transition from descriptive pleural fluid analysis to metabolic profiling allows us to move beyond simply naming the fluid and start understanding the cellular distress of the pleura.”
This perspective aligns with broader trends in precision medicine, as seen in PubMed indexed trials focusing on biomarkers for pleural diseases, where the goal is to move away from “blind” drainage toward targeted molecular therapy.
Contraindications & When to Consult a Doctor
It is critical to understand that lactate profiling is a diagnostic tool, not a treatment. There are no “contraindications” to the test itself, as it involves analyzing fluid already being removed via thoracentesis (a procedure to drain fluid from the chest).
Patients should seek immediate medical intervention if they experience:
- Acute Dyspnea: Sudden, severe shortness of breath that does not improve with rest.
- Pleuritic Chest Pain: Sharp, stabbing pain that worsens during inhalation.
- Hemoptysis: Coughing up blood, which may indicate a malignant process or pulmonary embolism.
- High Fever with Productive Cough: Indicative of a potential parapneumonic effusion requiring urgent drainage to prevent empyema (pus in the pleural space).
Consult a pulmonologist or thoracic surgeon if you have a chronic effusion. Do not attempt to interpret laboratory lactate values without a clinical correlation, as systemic lactic acidosis (due to shock or organ failure) can skew results.
The Path Toward Precision Pleural Diagnostics
The diagnostic utility of pleural fluid lactate marks a significant step forward in respiratory medicine. By bridging the gap between simple protein counts and complex histological biopsies, the lactate ratio offers a window into the metabolic state of the lung’s lining.
As we move toward 2027, the integration of this marker into standard clinical protocols—supported by larger, multi-center trials—could redefine the triage process for pleural effusions. For the patient, In other words fewer needles, faster diagnoses, and a more rapid return to respiratory health.
References
- National Center for Biotechnology Information (NCBI) / PubMed – Pleural Effusion Biomarkers.
- The Lancet – Global Respiratory Health and Infectious Disease Trends.
- JAMA (Journal of the American Medical Association) – Clinical Guidelines for Thoracentesis and Fluid Analysis.
- Centers for Disease Control and Prevention (CDC) – Pneumonia and Tuberculosis Diagnostic Protocols.