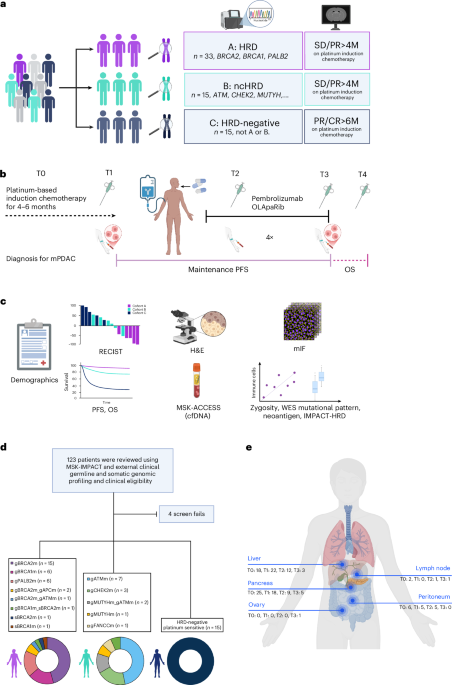
The phase 2 POLAR trial, published this week, evaluates the combination of pembrolizumab and olaparib as maintenance therapy for metastatic pancreatic cancer patients with homologous recombination deficiency (HRD). While the study did not meet its primary endpoint for objective response rate in the core HRD cohort, it demonstrated a median progression-free survival of 8.3 months and a 2-year overall survival rate of 56% in patients with BRCA1/2 or PALB2 mutations, offering a potential lifeline for a genetically defined subset of patients.
Pancreatic cancer remains one of the most lethal malignancies globally, with five-year survival rates historically stagnating below 12%. For patients harboring specific DNA repair defects, however, the therapeutic landscape is shifting. The POLAR trial represents a critical pivot toward precision maintenance therapy, moving beyond the standard “one-size-fits-all” chemotherapy approach. By stratifying patients based on genomic scars and mutation profiles, this research illuminates a path where immunotherapy and PARP inhibitors may synergize to extend life, even if a outright “cure” remains elusive for the majority. This data is vital for oncologists and patients navigating the complex decision matrix of second-line maintenance strategies in 2026.
In Plain English: The Clinical Takeaway
- Genetic Testing is Key: Patients with specific gene mutations (BRCA1, BRCA2, PALB2) responded better to the drug combination than those without, highlighting the need for comprehensive genomic profiling.
- Disease Control Over Cure: While the drugs did not shrink tumors in every patient, they successfully halted disease progression for a median of 8.3 months in the most responsive group.
- Safety Profile: The treatment was generally tolerable, with anemia being the most common serious side effect, and no unexpected safety risks were identified compared to using the drugs individually.
Decoding the Mechanism: Synthetic Lethality and Immune Priming
To understand the POLAR trial’s significance, one must understand the biological vulnerability it exploits. Olaparib is a PARP inhibitor, a class of drugs that blocks the repair of single-strand DNA breaks. In cells with Homologous Recombination Deficiency (HRD)—caused by mutations in genes like BRCA1 or BRCA2—the backup repair system is also broken. This concept, known as synthetic lethality, forces cancer cells to accumulate fatal DNA damage while sparing healthy cells.
Pembrolizumab, an immune checkpoint inhibitor, works differently. It releases the “brakes” on the immune system, allowing T-cells to attack the tumor. The hypothesis driving the POLAR trial was that olaparib-induced DNA damage might increase the tumor’s mutational burden, creating more neoantigens (foreign-looking proteins) that pembrolizumab-primed T-cells could recognize. The trial’s translational data supports this: Cohort A (core HRD) showed significantly higher tumor-infiltrating lymphocyte (TIL) density and frameshift indel burdens compared to HRD-negative cohorts. This suggests that while the primary endpoint was missed, the biological mechanism of immune priming in HRD-positive pancreatic cancer is valid and warrants further optimization.
Regulatory Landscape and Access in 2026
As of March 2026, the regulatory status of this combination remains a focal point for agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). While olaparib and pembrolizumab are individually approved for various indications, their combination in pancreatic cancer is largely confined to clinical trials or off-label use depending on the jurisdiction. In the United States, NCCN guidelines increasingly recommend germline and somatic testing for all pancreatic adenocarcinoma patients, which facilitates access to such targeted trials. However, in healthcare systems with stricter reimbursement protocols, such as the NHS in the UK, access may be limited until phase 3 data confirms a statistically significant overall survival benefit over standard of care.
The funding for the POLAR trial typically involves a consortium of academic medical centers, such as Memorial Sloan Kettering Cancer Center (indicated by the use of MSK-IMPACT sequencing), often in collaboration with pharmaceutical sponsors. Transparency regarding funding is essential; industry-sponsored trials can sometimes introduce bias in endpoint selection, though the POLAR trial’s use of independent radiology review for RECIST evaluation helps mitigate this risk.
| Cohort | Genetic Profile | Participants (N) | Median PFS | 2-Year OS Rate |
|---|---|---|---|---|
| Cohort A | Core HRD (BRCA1/2, PALB2) | 33 | 8.3 Months | 56% |
| Cohort B | Non-core HRD (ATM, CHEK2, etc.) | 15 | 4.8 Months | N/A (Median 18 Mo) |
| Cohort C | HRD Negative (Platinum Sensitive) | 15 | 3.3 Months | N/A (Median 10 Mo) |
Interpreting the Data: Beyond the Primary Endpoint
Critically, the POLAR trial did not meet its prespecified co-primary endpoint for Objective Response Rate (ORR) in Cohort A, which stood at 35%. However, in oncology, Progression-Free Survival (PFS) and Overall Survival (OS) are often more clinically meaningful metrics than tumor shrinkage alone. The 8.3-month median PFS in Cohort A is a notable improvement over historical controls for maintenance therapy in this setting. The exploratory analysis revealed that patients with PALB2 mutations had a median PFS of 12.0 months, outperforming even the BRCA2 subgroup. This granularity is essential for clinicians; it suggests that not all HRD mutations confer the same level of sensitivity to PARP inhibition, and PALB2 status should be weighed heavily in treatment planning.
The trial also highlighted the prognostic value of baseline CA 19-9 levels. A stratified multivariable Cox analysis confirmed that higher baseline CA 19-9 was significantly associated with worse PFS and OS, independent of other risk factors. This reinforces the utility of this biomarker not just for diagnosis, but for risk stratification during maintenance therapy.
Contraindications & When to Consult a Doctor
While the POLAR regimen offers hope, it is not suitable for every patient. The combination is contraindicated in patients with a history of severe hypersensitivity to pembrolizumab or olaparib, and those with severe renal or hepatic impairment. Given the mechanism of action, patients with active autoimmune diseases requiring systemic immunosuppression should generally avoid checkpoint inhibitors due to the risk of exacerbating their condition.
Patients on this regimen must be vigilant for Immune-Related Adverse Events (irAEs). In the POLAR trial, grade 3 irAEs included pneumonitis, and colitis. Symptoms such as persistent cough, shortness of breath, severe diarrhea, or abdominal pain warrant immediate medical consultation. Olaparib is associated with anemia; patients should report unusual fatigue, pallor, or shortness of breath, as dose modifications or transfusions may be required. Regular monitoring of blood counts and liver function is mandatory throughout the treatment course.
The trajectory of pancreatic cancer treatment is moving toward hyper-personalization. The POLAR trial confirms that while a universal “magic bullet” remains out of reach, matching the right molecular key to the right genetic lock can yield meaningful extensions in survival. For the 5-7% of pancreatic cancer patients with germline BRCA mutations, and the broader subset with somatic HRD, this combination represents a validated, biologically rational standard of care to discuss with their oncology team.
References
- Nature Medicine. “Pembrolizumab and olaparib in homologous-recombination-deficient metastatic pancreatic cancer: the phase 2 POLAR trial.” Published online March 2026.
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology: Pancreatic Adenocarcinoma. Version 2026.
- Lord, C. J., & Ashworth, A. “PARP inhibitors: Synthetic lethality in the clinic.” Science, 2017.
- U.S. Food and Drug Administration (FDA). “Olaparib (Lynparza) and Pembrolizumab (Keytruda) Prescribing Information.” Updated 2025.
- Memorial Sloan Kettering Cancer Center. “MSK-IMPACT High-Throughput Sequencing Assay.” Clinical Laboratory Improvement Amendments (CLIA) Documentation.