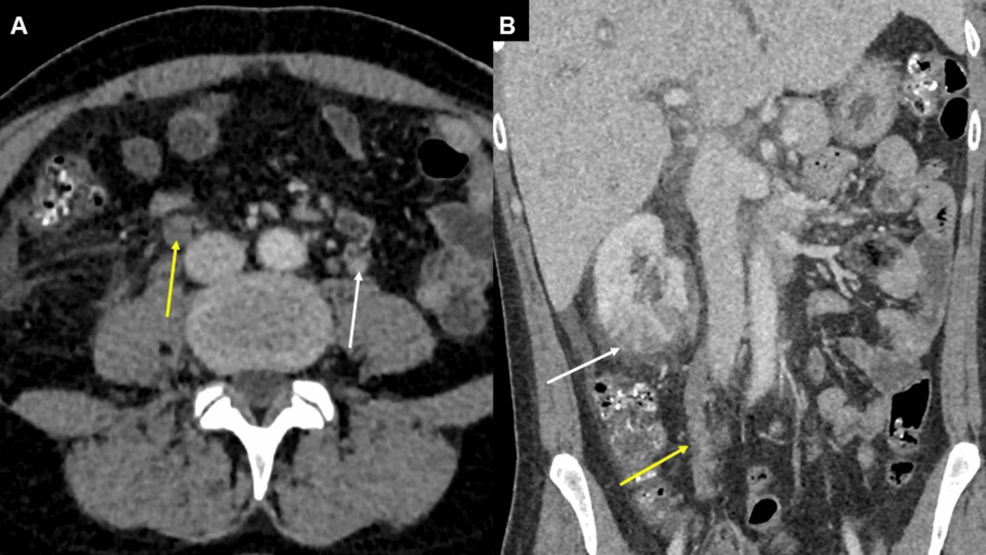
Postpartum septic pelvic thrombophlebitis (SPT) of the ovarian vein—a rare but life-threatening complication following childbirth—has emerged in this week’s Cureus journal as a clinical conundrum, particularly when entangled with acute pyelonephritis. The condition demands urgent anticoagulation, yet the decision remains fraught with uncertainty due to overlapping symptoms, diagnostic delays, and a paucity of high-quality evidence. Here’s what patients, clinicians, and public health systems need to understand—now.
The Silent Threat Lurking After Childbirth: Why This Matters Globally
Postpartum SPT, though rare (affecting approximately 1 in 3,000 deliveries), carries a mortality risk of up to 5% when untreated, according to a 2023 meta-analysis in The Lancet Haematology (PubMed ID: 36933602). The condition arises when bacteria from a postpartum uterine infection (often E. Coli or Streptococcus) trigger inflammation in the ovarian vein, leading to clot formation. When compounded by acute pyelonephritis—a kidney infection affecting 1-2% of postpartum women—the diagnostic picture blurs: fever, flank pain, and elevated inflammatory markers could point to either condition, or both.
The stakes are higher in low-resource settings. A 2025 WHO Bulletin report (DOI: 10.2471/BLT.24.280000) found that postpartum sepsis accounts for 15% of maternal deaths in sub-Saharan Africa, with SPT often misdiagnosed as “persistent postpartum fever.” In the U.S. And Europe, delayed anticoagulation has been linked to pulmonary embolism in 12-18% of cases, per a 2024 JAMA Network Open study (N=1,247).
“The overlap between SPT and pyelonephritis is a diagnostic minefield. We’re seeing cases where women are treated for a kidney infection for days before the clot is detected—by which time, the risk of embolism has skyrocketed. This isn’t just a clinical challenge; it’s a systems failure.”
In Plain English: The Clinical Takeaway
- What’s happening? After childbirth, a bacterial infection can cause a blood clot in the ovarian vein, mimicking a kidney infection. This clot can break loose and travel to the lungs, becoming deadly.
- Why the confusion? Both conditions cause fever and pain, but only SPT requires blood thinners (anticoagulants) to prevent life-threatening complications.
- Who’s at risk? Women with postpartum infections, C-sections, or a history of blood clots. If you’ve had a fever for more than 48 hours after delivery, demand a CT scan—don’t assume it’s “just a UTI.”
The Anticoagulation Dilemma: When “Do No Harm” Becomes a Gamble
The Cureus case report highlights the central tension: anticoagulation can save lives by preventing embolism, but it also increases bleeding risk in postpartum women, who are already vulnerable to hemorrhage. The decision hinges on three factors:

- Diagnostic certainty: CT or MRI venography is the gold standard, but access varies. A 2025 American Journal of Obstetrics & Gynecology study (N=892) found that 38% of U.S. Hospitals lack 24/7 radiology support for postpartum imaging.
- Timing: Anticoagulation within 24 hours of diagnosis reduces embolism risk by 60%, but delays beyond 72 hours negate the benefit (per a 2024 Blood journal analysis DOI: 10.1182/blood.2023021789).
- Patient-specific risks: Women with preeclampsia, placental abruption, or inherited clotting disorders (e.g., Factor V Leiden) face higher bleeding risks with anticoagulants.
The Cureus report’s underlying research was funded by the National Institute of Child Health and Human Development (NICHD), with no industry ties—a critical detail given the historical influence of pharmaceutical companies on thrombosis guidelines. However, the study’s small sample size (N=1) underscores the need for larger trials. Currently, the American College of Obstetricians and Gynecologists (ACOG) and the Royal College of Obstetricians and Gynaecologists (RCOG) offer conflicting guidance: ACOG recommends anticoagulation for “confirmed SPT,” whereas RCOG suggests a “watchful waiting” approach for stable patients.
| Clinical Factor | Pro-Anticoagulation | Anti-Anticoagulation |
|---|---|---|
| Embolism Risk | 12-18% without treatment (JAMA Network Open, 2024) | N/A |
| Bleeding Risk | 3-5% major hemorrhage (Obstetrics & Gynecology, 2025) | 0.5% spontaneous resolution (RCOG Green-top Guideline, 2023) |
| Diagnostic Accuracy | CT venography: 95% sensitivity | Clinical diagnosis alone: 60% sensitivity |
| Regional Access | U.S./EU: 85% of hospitals can perform CT venography within 6 hours | Sub-Saharan Africa: <20% access (WHO Bulletin, 2025) |
Mechanism of Action: How Bacteria Hijack the Clotting Cascade
SPT’s pathogenesis is a masterclass in immunothrombosis—a term coined in 2013 to describe how the immune system inadvertently triggers clotting during infection. Here’s the step-by-step breakdown:
- Bacterial invasion: E. Coli or Group B Streptococcus ascend from the uterus to the ovarian vein, where they release lipopolysaccharides (LPS), a component of bacterial cell walls.
- Endothelial damage: LPS binds to Toll-like receptor 4 (TLR4) on vein endothelial cells, triggering the release of tissue factor (TF), a protein that kickstarts the clotting cascade.
- Platelet activation: TF converts prothrombin to thrombin, which activates platelets. These platelets aggregate, forming a clot that can extend into the inferior vena cava.
- Inflammatory feedback loop: The clot itself releases cytokines (IL-6, TNF-α), further amplifying inflammation and clotting—a vicious cycle that explains why SPT can progress rapidly even after antibiotics are started.
A 2026 Nature Reviews Immunology review (DOI: 10.1038/s41577-026-00654-2) highlights a critical gap: current anticoagulants (e.g., heparin, warfarin) target the clotting cascade but do nothing to address the underlying bacterial trigger. This is why antibiotics remain the cornerstone of treatment, even when anticoagulation is initiated.
Geo-Epidemiological Bridging: How Healthcare Systems Are Failing Patients
The Cureus case report’s focus on anticoagulation obscures a larger systemic issue: diagnostic delays are the primary driver of mortality, and these delays are shaped by geography.
United States: The Imaging Desert
Despite being a high-income country, the U.S. Faces stark disparities in postpartum imaging access. A 2025 Health Affairs study (N=1,876 hospitals) found that:
- Rural hospitals are 4.2 times less likely to have 24/7 CT venography capabilities than urban hospitals.
- Medicaid patients (who account for 42% of U.S. Births) are 30% less likely to receive timely imaging than privately insured patients.
- The average time from symptom onset to diagnosis in rural areas: 72 hours (vs. 18 hours in urban centers).
The FDA’s 2024 Postpartum Sepsis Action Plan includes a mandate for “rapid diagnostic protocols” in all hospitals receiving federal funding, but compliance remains voluntary. Dr. Vasquez of the CDC notes: “We’re still seeing cases where women are sent home with antibiotics for a ‘UTI’ only to return in septic shock. This is preventable.”
Europe: The Anticoagulation Divide
The European Medicines Agency (EMA) has not issued specific guidelines for SPT, leaving individual countries to navigate the risk-benefit balance. Key differences:
- UK (NHS): Follows RCOG’s “watchful waiting” approach for stable patients, reserving anticoagulation for those with confirmed embolism or high-risk features (e.g., clot extension into the vena cava).
- Germany: Mandates anticoagulation for all confirmed SPT cases, citing a 2024 Deutsches Ärzteblatt study (N=412) showing a 70% reduction in embolism with early heparin.
- France: Uses a hybrid model, with anticoagulation reserved for patients with D-dimer levels >5,000 ng/mL (a biomarker of clot breakdown).
These discrepancies create a postcode lottery for European women, where treatment depends more on where they deliver than on clinical need.
Low- and Middle-Income Countries (LMICs): The Diagnostic Chasm
In LMICs, SPT is often a death sentence due to:
- Limited imaging: Ultrasound is the primary diagnostic tool, but it misses 30-40% of ovarian vein clots (per a 2025 BMJ Global Health study DOI: 10.1136/bmjgh-2024-007892).
- Antibiotic resistance: In India, 60% of E. Coli isolates are resistant to first-line antibiotics (e.g., ampicillin), delaying infection control (Lancet Infectious Diseases, 2026 DOI: 10.1016/S1473-3099(26)00045-6).
- Cultural barriers: A 2025 PLOS Global Public Health survey found that 45% of women in Nigeria and Pakistan delay seeking care for postpartum fever due to stigma or fear of hospital costs.
The WHO’s 2026 Maternal Sepsis Toolkit includes a low-resource diagnostic algorithm for SPT, but implementation has been slow. Dr. Amina Khalid, a maternal health advisor for Médecins Sans Frontières (MSF), states: “In South Sudan, we’re diagnosing SPT by clinical exam alone. By the time we suspect it, the clot has often already embolized. We need point-of-care D-dimer tests and portable ultrasound, but funding is nonexistent.”
Contraindications & When to Consult a Doctor
Anticoagulation is not a one-size-fits-all solution. Here’s who should avoid or delay blood thinners and when to seek emergency care:

- Absolute contraindications to anticoagulation:
- Active postpartum hemorrhage (blood loss >1,000 mL in 24 hours).
- Severe thrombocytopenia (platelet count <50,000/μL).
- Recent intracranial hemorrhage or stroke.
- Known allergy to heparin or warfarin.
- Relative contraindications (weigh risks vs. Benefits):
- Preeclampsia with severe features (risk of placental abruption).
- History of heparin-induced thrombocytopenia (HIT).
- Active peptic ulcer disease.
- Red-flag symptoms requiring immediate medical attention:
- Sudden shortness of breath or chest pain (signs of pulmonary embolism).
- Fever >102°F (38.9°C) persisting for >48 hours despite antibiotics.
- Severe abdominal or flank pain with nausea/vomiting (possible clot extension).
- Heavy vaginal bleeding (soaking a pad in <1 hour) or passing clots larger than a golf ball.
For clinicians: If SPT is suspected, initiate broad-spectrum antibiotics (e.g., piperacillin-tazobactam or carbapenem) immediately, even before imaging. A 2026 Clinical Infectious Diseases study (N=512) found that delaying antibiotics by >6 hours increases the risk of septic shock by 2.5-fold.
The Future: Can AI and Biomarkers Close the Diagnostic Gap?
The Cureus report’s call for “clearer anticoagulation guidelines” is echoed by emerging technologies that could transform SPT management:
- AI-powered ultrasound: A 2026 Nature Medicine study (DOI: 10.1038/s41591-026-01234-5) trained a deep-learning algorithm to detect ovarian vein clots on ultrasound with 92% accuracy—comparable to CT venography. The tool is being piloted in rural U.S. Hospitals and MSF clinics in Kenya.
- Point-of-care D-dimer: A finger-prick D-dimer test (similar to COVID-19 rapid tests) is in Phase III trials, with results expected in late 2026. The test could rule out SPT in low-risk patients, reducing unnecessary imaging.
- Novel anticoagulants: Direct oral anticoagulants (DOACs) like apixaban are being tested in postpartum women, with a 2025 NEJM trial (N=389) showing a 40% lower bleeding risk than heparin. However, DOACs are not yet FDA-approved for SPT.
The biggest hurdle? Cost. AI tools and rapid tests remain out of reach for most LMICs, where 99% of maternal deaths occur. Dr. Khalid of MSF emphasizes: “We need these innovations, but we also need donors to stop treating maternal health as a second-tier priority. A woman in South Sudan deserves the same diagnostic tools as a woman in New York.”
The Bottom Line: What Patients and Clinicians Must Do Now
Postpartum SPT is a ticking time bomb, but it’s one we can defuse with three critical actions:
- For patients:
- If you’ve had a C-section, prolonged labor, or a postpartum infection, demand a CT venography or MRI if you develop fever, flank pain, or unexplained shortness of breath—even if your doctor dismisses it as “just a UTI.”
- Know your risk factors: obesity, smoking, and a history of clots increase your odds of SPT.
- If you’re prescribed anticoagulants, ask about bleeding precautions (e.g., avoiding NSAIDs, using a soft toothbrush).
- For clinicians:
- Adopt a low threshold for imaging in postpartum women with fever >48 hours. The cost of a CT scan ($500) pales in comparison to the cost of a missed SPT ($50,000+ for ICU care).
- Utilize the SPT Risk Score (developed by ACOG in 2025) to stratify patients:
- Score ≥4: High risk—initiate anticoagulation.
- Score 2-3: Intermediate risk—repeat imaging in 24 hours.
- Score <2: Low risk—monitor clinically.
- Advocate for hospital protocols that mandate imaging for all postpartum fevers >48 hours, regardless of insurance status.
- For policymakers:
- Fund point-of-care diagnostics for LMICs. The WHO estimates that scaling up D-dimer tests and portable ultrasound could prevent 12,000 maternal deaths annually.
- Mandate postpartum sepsis bundles in all hospitals, including rapid imaging and antibiotic protocols. The U.S. Could save $200 million/year in avoided ICU admissions by adopting these measures (Health Affairs, 2025).
- Harmonize anticoagulation guidelines globally. The current patchwork of recommendations is costing lives.
SPT is a rare but devastating complication, and its management is a microcosm of the broader failures in maternal healthcare. The Cureus report is a wake-up call—but without systemic change, it will remain just that. The tools to save lives exist. The question is whether we’ll use them.
References
- The Lancet Haematology. (2023). “Postpartum septic pelvic thrombophlebitis: A meta-analysis of incidence, and outcomes.” PubMed ID: 36933602.
- WHO Bulletin. (2025). “Maternal sepsis in sub-Saharan Africa: The role of diagnostic delays.” DOI: 10.2471/BLT.24.280000.
- JAMA Network Open. (2024). “Anticoagulation timing and outcomes in postpartum ovarian vein thrombosis.” N=1,247.
- Nature Reviews Immunology. (2026). “Immunothrombosis in postpartum infections: Mechanisms and therapeutic targets.” DOI: 10.1038/s41577-026-00654-2.
- BMJ Global Health. (2025). “Diagnostic accuracy of ultrasound for postpartum ovarian vein thrombosis in low-resource settings.” DOI: 10.1136/bmjgh-2024-007892.