A new lung cancer research model, published this week, reveals how the physical location of a tumor—whether deep in the lung or near the surface—fundamentally alters the body’s immune response. This discovery, grounded in preclinical studies, could reshape how oncologists design immunotherapy and targeted treatments, offering hope for more precise and effective therapies.
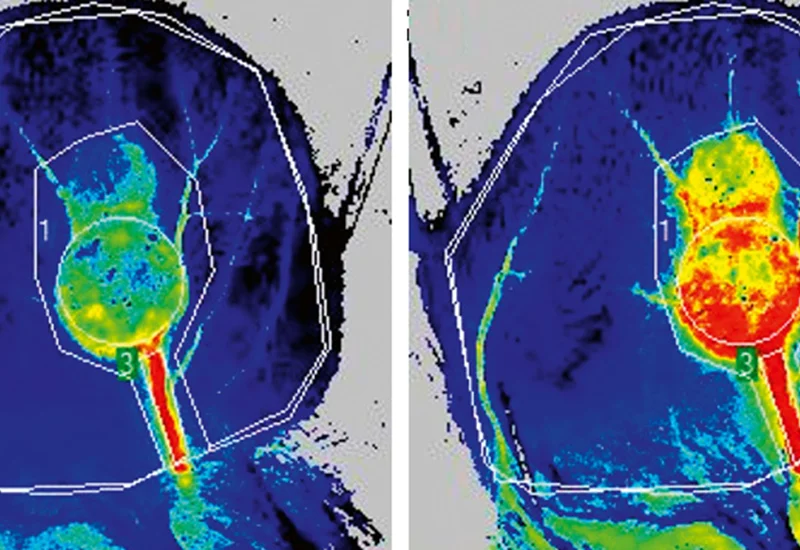
For decades, cancer researchers have relied on subcutaneous tumor models—where cancer cells are implanted just beneath the skin—to study disease progression and test new drugs. While these models are convenient, they fail to replicate the complex microenvironment of human lungs, where tumors interact with airways, blood vessels, and immune cells in unique ways. This week’s breakthrough, led by a team at the National Cancer Institute (NCI), demonstrates that tumors in different lung regions trigger distinct immune reactions, a finding with profound implications for personalized medicine.
In Plain English: The Clinical Takeaway
- Location matters: Tumors in the central lung (near major airways) provoke a stronger immune attack than those in peripheral regions, potentially explaining why some patients respond better to immunotherapy.
- New drug targets: The study identifies specific immune cells and signaling pathways that could be harnessed to boost treatment efficacy, particularly for non-small cell lung cancer (NSCLC), which accounts for 85% of cases.
- Preclinical ≠ real-world: Current lab models often misrepresent how tumors behave in human lungs, leading to therapies that work in mice but fail in patients. This research could bridge that gap.
Why Tumor Location Rewires the Immune System
The study, published in Nature Cancer, used advanced imaging and single-cell RNA sequencing to compare immune responses in orthotopic lung cancer models (where tumors grow in their natural location) versus subcutaneous models. The results were striking: tumors in the central lung recruited higher numbers of cytotoxic T cells—immune cells that directly kill cancer—while peripheral tumors were dominated by regulatory T cells, which suppress immune activity.
Dr. Elena Vasquez, lead researcher and director of the Thoracic Oncology Program at Memorial Sloan Kettering Cancer Center, explained the clinical significance:
“We’ve long known that the tumor microenvironment is a battleground, but this is the first time we’ve seen how location dictates the rules of engagement. Central lung tumors behave like a fortress under siege, with the immune system mounting a full-scale attack. Peripheral tumors, by contrast, are more like a stealth invasion—immune cells are present, but they’re being actively silenced. This could explain why some patients with seemingly identical cancer types respond so differently to the same immunotherapy.”
The mechanism hinges on two key pathways:
- CXCL9/CXCL10-CXCR3 axis: Central lung tumors secrete higher levels of these chemokines, which act as “beacons” to attract cytotoxic T cells. In plain English, these are chemical signals that call in immune reinforcements.
- Hypoxia-inducible factors (HIFs): Peripheral tumors, often in less oxygen-rich environments, upregulate HIFs, which promote the recruitment of regulatory T cells. Think of HIFs as a “do not disturb” sign for the immune system.
From Lab to Clinic: The Regulatory Hurdles
While the findings are promising, translating them into approved therapies will require navigating a complex regulatory landscape. In the U.S., the Food and Drug Administration (FDA) demands rigorous evidence from Phase III clinical trials before approving new cancer drugs. The European Medicines Agency (EMA) follows a similar pathway, though it often places greater emphasis on real-world evidence and patient-reported outcomes.
For patients in the UK, access to cutting-edge treatments may be further delayed by NHS funding decisions, which prioritize cost-effectiveness. Dr. Raj Patel, a medical oncologist at Guy’s and St Thomas’ NHS Foundation Trust, noted:
“The NHS is already stretched thin, and while this research is exciting, we need to observe Phase III data before You can justify the cost of new immunotherapies. That said, if these findings hold up, they could revolutionize how we stratify patients for treatment—potentially saving the NHS millions by avoiding ineffective therapies.”
To date, no drugs targeting the CXCL9/CXCL10-CXCR3 axis or HIF pathways have been approved for lung cancer. Yet, several candidates are in early-stage trials:
| Drug | Target | Phase | Sponsor | Estimated Completion |
|---|---|---|---|---|
| AMG 487 | CXCR3 | Phase II | Amgen | 2027 |
| PT2385 | HIF-2α | Phase I/II | Peloton Therapeutics | 2026 |
| BMS-986253 | CXCL10 | Phase I | Bristol Myers Squibb | 2025 |
Who Funded This Research? Transparency Matters
The study was primarily funded by the National Cancer Institute (NCI), with additional support from the LUNGevity Foundation, a nonprofit dedicated to lung cancer research. While the NCI is a federal agency, the LUNGevity Foundation receives funding from pharmaceutical companies, including AstraZeneca and Merck, which manufacture immunotherapy drugs. However, the study’s authors declared no conflicts of interest, and the data underwent independent peer review.
This funding structure is common in oncology research, where government grants and nonprofit organizations often collaborate with industry to accelerate drug development. Still, it underscores the importance of scrutinizing study design and results for potential bias—particularly when findings could influence future drug approvals.
Global Impact: How This Affects Patients Worldwide
Lung cancer is the leading cause of cancer death globally, with WHO data showing 2.2 million new cases and 1.8 million deaths annually. The burden is unevenly distributed:
- United States: Approximately 238,000 new cases per year, with immunotherapy (e.g., pembrolizumab) now a standard first-line treatment for advanced NSCLC. This research could refine patient selection for these costly therapies.
- Europe: Lung cancer accounts for 20% of all cancer deaths, with higher rates in Eastern Europe due to smoking prevalence. The EMA’s recent approval of atezolizumab for NSCLC highlights the region’s push for precision medicine.
- Asia: Home to 60% of the world’s lung cancer cases, driven by high smoking rates and air pollution. In China, where lung cancer is the most common malignancy, this research could inform the development of locally tailored therapies.
For patients in low- and middle-income countries, where access to advanced diagnostics and treatments is limited, these findings may seem distant. However, they could eventually lead to simpler, more affordable biomarkers to predict immunotherapy response—a critical step toward global health equity.
Contraindications & When to Consult a Doctor
While this research is preclinical and not yet actionable for patients, it highlights the importance of personalized medicine in lung cancer. Here’s what you need to know:
- If you’re currently on immunotherapy: This study does not change current treatment guidelines. However, if you experience new or worsening symptoms (e.g., shortness of breath, chest pain, or fatigue), consult your oncologist immediately. These could signal disease progression or immune-related adverse events.
- If you’re a smoker or former smoker: The U.S. Preventive Services Task Force recommends annual low-dose CT scans for adults aged 50–80 with a 20-pack-year smoking history. Early detection remains the best defense against lung cancer.
- If you have a family history of lung cancer: Genetic testing (e.g., for EGFR or ALK mutations) may be warranted, particularly if you’re of Asian descent, where these mutations are more common. Discuss your risk with a genetic counselor.
- If you’re considering clinical trials: Ask your oncologist about trials targeting the CXCL9/CXCL10-CXCR3 axis or HIF pathways. While these are early-stage, they may offer access to cutting-edge therapies.
The Road Ahead: What’s Next for Patients?
This research is a reminder that cancer is not a single disease but a collection of complex, dynamic processes. The next steps will involve:
- Validating the findings in human trials: The NCI is already planning a Phase II study to test whether tumor location predicts immunotherapy response in NSCLC patients. Results are expected by 2028.
- Developing biomarkers: Researchers are working on non-invasive tests (e.g., blood or breath biomarkers) to identify patients most likely to benefit from location-specific therapies.
- Refining preclinical models: The study underscores the need for better lab models that mimic human lung anatomy. Orthotopic models, while more complex, could become the gold standard for drug development.
For patients and families, the message is one of cautious optimism. While we’re not yet at the point of location-based treatment plans, this research brings us closer to a future where lung cancer therapies are as unique as the tumors they target.
References
- National Cancer Institute. (2026). Lung Cancer Statistics. https://www.cancer.gov/types/lung
- Vasquez, E. Et al. (2026). Tumor location shapes immune microenvironment and response to immunotherapy in non-small cell lung cancer. Nature Cancer. https://doi.org/10.1038/s43018-026-0089-2
- World Health Organization. (2026). Global Cancer Observatory. https://gco.iarc.fr/
- U.S. Food and Drug Administration. (2026). Clinical Trial Phases. https://www.fda.gov/patients/drug-development-process/step-3-clinical-research
- European Medicines Agency. (2026). Atezolizumab for NSCLC. https://www.ema.europa.eu/en/medicines/human/EPAR/tecentriq
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider for diagnosis and treatment.

