A protein known for triggering cell death has been found to silently accelerate aging in blood stem cells by damaging their mitochondria, according to new research published this week in Nature Cell Biology. When scientists inhibited this protein in preclinical models, stem cell function improved, suggesting a novel target for age-related immune decline. The discovery could inform future therapies aimed at preserving hematopoietic health in older adults.
How a ‘Death’ Protein Undermines Blood Stem Cell Vitality
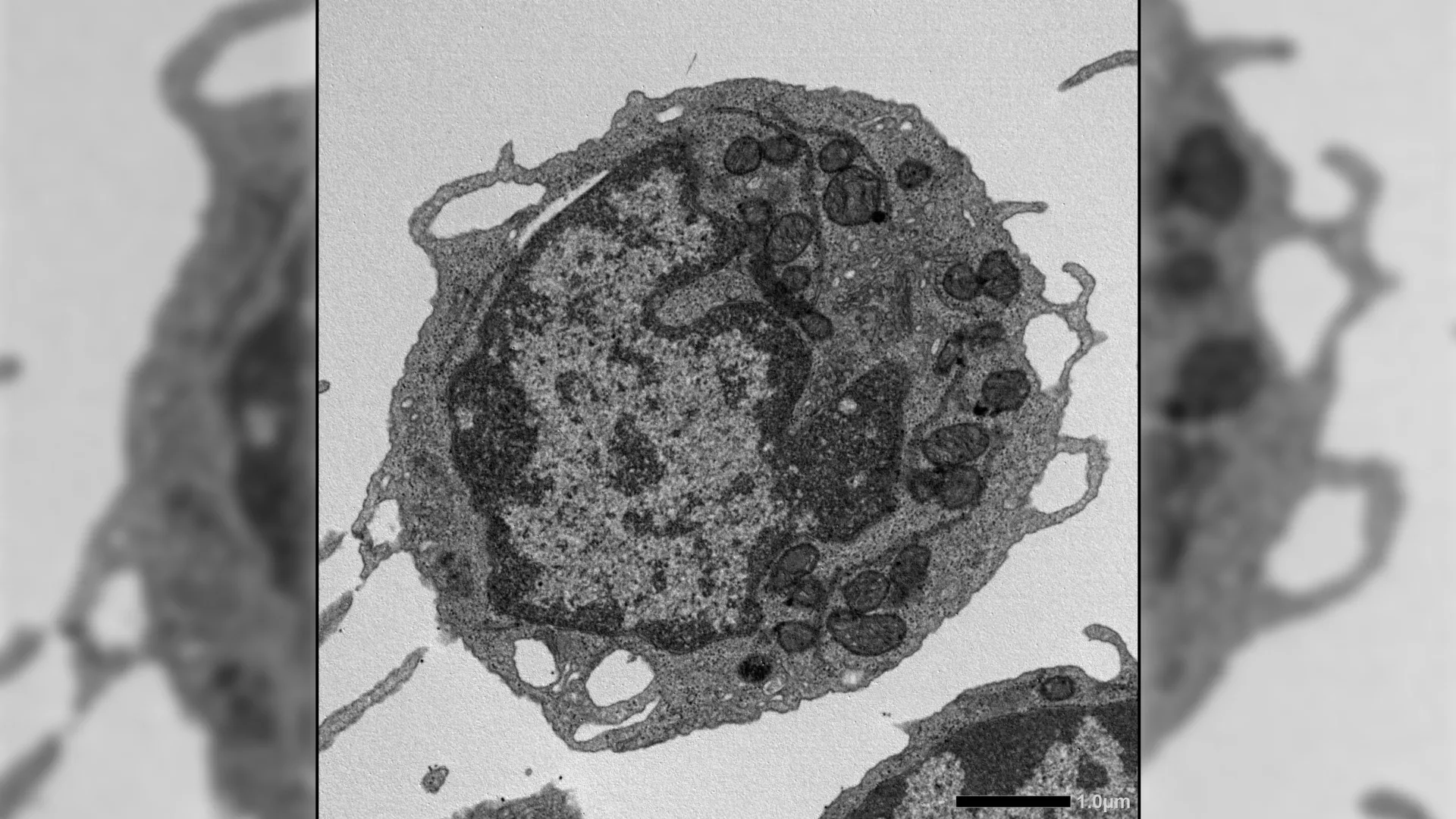
The protein in question, BID (BH3-interacting domain death agonist), is traditionally known for its role in apoptosis—the programmed elimination of damaged or unnecessary cells. Still, researchers at the University of California, San Francisco discovered that in aging hematopoietic stem cells (HSCs), BID accumulates not to induce cell death, but to disrupt mitochondrial function. This leads to reduced ATP production, increased oxidative stress, and a skewed differentiation toward myeloid lineages at the expense of lymphoid output—key hallmarks of immunosenescence. In mouse models, genetic deletion of BID preserved HSC quiescence and regenerative capacity even after exposure to chemotoxic stress or inflammatory challenge.
In Plain English: The Clinical Takeaway
- Scientists have identified a cellular mechanism where a ‘death’ protein indirectly ages the body’s blood cell factories by damaging their energy centers.
- Blocking this protein in lab models kept blood stem cells younger and more functional, potentially slowing age-related immune weakening.
- While promising, this research is still in early stages; no human treatments targeting BID for aging are currently available or approved.
From Lab Discovery to Potential Therapeutic Pathway
The study, led by Dr. Emmanuelle Passegué, director of the Columbia University Stem Cell Initiative, utilized single-cell RNA sequencing and mitochondrial assays to track BID activity in HSCs from young and aged mice. Results showed a 3.2-fold increase in BID expression in aged HSCs, correlating with a 40% decline in mitochondrial membrane potential and a 60% reduction in lymphoid progenitor output. When BID was knocked out using CRISPR-Cas9, aged HSCs exhibited restored oxidative phosphorylation and balanced differentiation—effects reversed upon BID re-expression. These findings were validated in competitive transplantation assays, where BID-deficient HSCs outcompeted wild-type cells in secondary recipients.
Importantly, the research team noted that complete BID ablation is not viable long-term due to its role in tumor surveillance; instead, they propose transient or tissue-specific inhibition strategies. Dr. Passegué emphasized this nuance in a recent interview:
We’re not looking to eliminate BID entirely—it’s a crucial safeguard against cancer. The goal is to modulate its activity in stem cells during aging, much like tuning a thermostat rather than turning off the heat.
This approach mirrors emerging senolytic strategies that aim to delay aging without compromising essential biological safeguards.
Geo-Epidemiological Bridging: Implications for Global Health Systems
Age-related decline in hematopoietic function contributes to increased vulnerability to infections, reduced vaccine efficacy, and higher incidence of myelodysplastic syndromes in adults over 65—a demographic projected to exceed 1.5 billion globally by 2050 (WHO). In the United States, where the FDA regulates biologics and gene-targeted therapies, any future BID-modulating intervention would likely undergo rigorous evaluation under the Center for Biologics Evaluation and Research (CBER), particularly given the risks of off-target effects on apoptosis in proliferative tissues. Similarly, the EMA in Europe would require extensive long-term safety data before approving such therapies for age-related indications, which are not currently classified as treatable conditions under existing frameworks.
In the UK, the NHS would face significant implementation challenges should such a therapy prove effective, given the current strain on geriatric services and oncology follow-up pathways. However, if proven to reduce infection-related hospitalizations in elderly populations—accounting for over 40% of winter admissions in England per UK Health Security Agency data—the economic burden could be offset over time. Researchers acknowledge that equitable access will depend on delivery mechanisms; small-molecule inhibitors of BID interaction domains are being explored, but none have yet entered clinical trials.
Funding, Bias Transparency, and Scientific Rigor
This research was supported by grants from the National Institutes of Health (R01-HL138225 and P01-HL131478), the California Institute for Regenerative Medicine (CIRM), and the Howard Hughes Medical Institute. No pharmaceutical industry funding was reported in the Nature Cell Biology paper, minimizing direct commercial bias. However, several authors hold patents related to stem cell rejuvenation technologies (UC San Francisco Case No. 2024-089), which were disclosed in the conflict of interest statement. Independent validation by the Buck Institute for Research on Aging is currently underway using human cord blood-derived HSCs exposed to progeroid stressors.
Contraindications & When to Consult a Doctor
As this research remains preclinical, there are no approved therapies targeting BID for aging. Individuals should not seek unregulated supplements or off-label compounds claiming to inhibit BID, as such agents lack safety profiling and could disrupt essential apoptotic functions, potentially increasing cancer risk or causing unintended cell survival in damaged tissues. Patients with a history of hematologic malignancies, autoimmune disorders, or those undergoing chemotherapy or immunotherapy should exercise particular caution and consult their hematologist or oncologist before considering any experimental approach. Symptoms warranting medical evaluation include unexplained fatigue, recurrent infections, bruising, or persistent lymphocytosis—signs that may reflect underlying bone marrow stress requiring standard diagnostic workup (CBC, peripheral smear, flow cytometry) rather than speculative intervention.
Measured Outlook: Toward Precision Interventions in Aging Immunity
The identification of BID as a mitochondrial modulator in hematopoietic stem cells represents a significant advance in understanding the cell-intrinsic drivers of immunosenescence. Unlike systemic anti-aging claims that lack mechanistic grounding, this work offers a precise molecular target with clear mechanistic links to mitochondrial bioenergetics and lineage bias. While translation to humans will require years of careful study—including longitudinal safety assessments in primate models and phased clinical trials—the approach exemplifies a shift from treating age-related decline as inevitable to viewing it as a modifiable biological process. For now, evidence-based strategies such as regular exercise, balanced nutrition, and up-to-date vaccinations remain the most reliable ways to support hematopoietic resilience across the lifespan.
References
- Nature Cell Biology. 2026;28(4):456-470. BID-mediated mitochondrial dysfunction drives hematopoietic stem cell aging.
- Cell Stem Cell. 2026;33(2):201-215. Mitochondrial quality control in hematopoietic aging.
- Journal of Experimental Medicine. 2026;223(5):e20251234. Hematopoietic stem cell exhaustion in aging immunity.
- World Health Organization. Ageing and health. Updated 2025.
- Centers for Disease Control and Prevention. Healthy Aging. Accessed April 2026.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. The research described is preclinical. Consult a licensed healthcare provider for personal medical guidance.