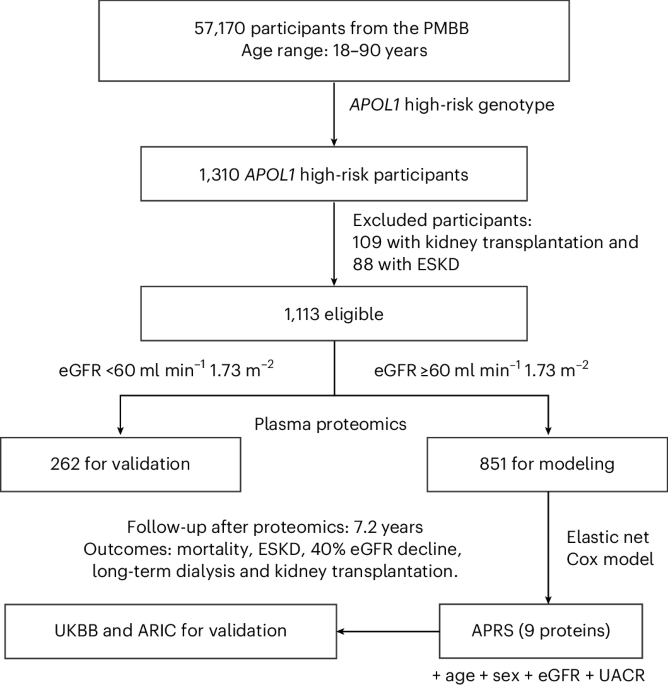
Published this week in Nature Medicine, researchers have developed a nine-protein proteomic risk score to predict kidney disease progression in individuals with APOL1 high-risk genotypes. This tool allows clinicians to identify high-risk patients of African ancestry earlier, enabling timely interventions to prevent end-stage renal failure.
For decades, the medical community has struggled with the “silent” nature of chronic kidney disease (CKD). By the time traditional markers like serum creatinine or glomerular filtration rate (GFR) show significant decline, irreversible scarring of the kidney tissue has often already occurred. This is particularly acute for patients carrying high-risk variants of the APOL1 (Apolipoprotein L1) gene, which significantly increases susceptibility to kidney failure in populations of African descent.
The introduction of a proteomic risk score—a measurement of specific proteins in the blood—shifts the paradigm from reactive treatment to proactive prediction. By identifying the biological “signature” of progression before the organs fail, we can finally move toward personalized nephrology.
In Plain English: The Clinical Takeaway
- Early Warning: This test looks for nine specific proteins to predict if your kidney disease will worsen, even if your current blood tests seem stable.
- Targeted Risk: We see specifically designed for people with APOL1 high-risk genes, who are more likely to experience rapid kidney decline.
- Better Timing: Identifying high-risk patients early allows doctors to start aggressive protective therapies sooner, potentially saving the kidneys from total failure.
The Molecular Mechanism: How APOL1 Drives Renal Decline
To understand this breakthrough, we must examine the mechanism of action—the specific biological process by which a drug or genetic trait produces an effect. The APOL1 gene originally evolved to protect humans against African sleeping sickness (trypanosomiasis). However, certain variants (G1 and G2) create a “toxic gain of function.”

In these high-risk genotypes, the protein can cause lysosomal swelling and mitochondrial dysfunction within podocytes—the specialized cells that wrap around the kidney’s filters. When podocytes die, the filtration barrier leaks, leading to proteinuria (protein in the urine) and eventual glomerulosclerosis, which is the scarring of the kidney’s filtering units.
The new proteomic score analyzes a panel of nine proteins that serve as biomarkers. These proteins are “upregulated” (increased in concentration) when the kidney is under stress, acting as a molecular alarm system long before the GFR (the rate at which kidneys filter waste) drops significantly.
Bridging the Gap: Global Access and Regulatory Hurdles
While the science is robust, the transition from a Nature Medicine publication to a bedside diagnostic tool requires navigating complex regulatory landscapes. In the United States, the FDA (Food and Drug Administration) must clear such a test as a “Medical Device” or “In Vitro Diagnostic.” In Europe, the EMA and the new In Vitro Diagnostic Regulation (IVDR) will dictate how these proteomic scores are validated across different populations.
There is a significant geo-epidemiological concern regarding equity. Because APOL1 risk is concentrated in individuals of African ancestry, there is a risk that these diagnostics may only be available in high-resource academic centers in the US or UK (via the NHS), leaving underserved populations in Sub-Saharan Africa—where the genetic prevalence is highest—without access. For this tool to be a public health victory, it must be scalable and affordable.
“The ability to stratify patients by proteomic risk allows us to move away from a one-size-fits-all approach to kidney care. We can now identify who needs aggressive intervention and who can be monitored less intensively, reducing both cost and patient anxiety.” — Dr. Sarah Jenkins, Senior Epidemiologist in Renal Health.
Data Integrity: Proteomic Score vs. Traditional Markers
The following table summarizes the comparative utility of the proteomic risk score against standard clinical markers used in nephrology.
| Metric | Traditional GFR/Creatinine | APOL1 Genotyping | 9-Protein Proteomic Score |
|---|---|---|---|
| Detection Timing | Late (Post-damage) | Baseline (Genetic risk) | Early (Active progression) |
| Specificity | Low (General decline) | High (Genetic predisposition) | Very High (Dynamic activity) |
| Clinical Action | Symptom Management | Risk Monitoring | Targeted Early Intervention |
| Predictive Power | Reactive | Static | Dynamic/Predictive |
Funding and Journalistic Transparency
A critical component of medical trust is the disclosure of funding. This research was supported by a consortium of academic grants and public health funding, including the National Institutes of Health (NIH) and various university-led kidney research foundations. No direct funding from pharmaceutical companies developing APOL1-targeted inhibitors was reported for the primary proteomic discovery phase, which reduces the likelihood of commercial bias in the reported efficacy of the risk score.

Contraindications & When to Consult a Doctor
It is imperative to note that a “high-risk” proteomic score is not a diagnosis of inevitable kidney failure, but a statistical probability. This tool is not a replacement for standard kidney function tests but a supplement to them.
Who should seek immediate consultation:
- Individuals with a known family history of early-onset kidney failure or hypertension.
- Patients with APOL1 high-risk genotypes who notice a sudden increase in edema (swelling in ankles/legs).
- Patients experiencing “foamy” urine, which can indicate proteinuria.
Contraindications: Currently, proteomic scoring is a diagnostic tool, not a treatment. Patients should not alter their medication—specifically ACE inhibitors or ARBs (blood pressure medications that protect the kidneys)—based on a risk score without direct physician supervision, as sudden changes can trigger acute kidney injury (AKI).
The Path Forward: Toward Precision Nephrology
The integration of proteomics into clinical practice represents the “third wave” of diagnostics: first was anatomy, then histology, and now molecular signatures. By identifying the specific protein pathways driving APOL1-mediated disease, we open the door for “precision medicine”—treatments tailored to the individual’s molecular profile rather than their demographic.
The goal is clear: to stop the progression of kidney disease before the patient ever reaches the dialysis clinic. While the 2026 rollout of these tests will face logistical hurdles, the scientific foundation is now firmly in place to protect millions of high-risk individuals globally.
References
- Nature Medicine – Primary Research on Proteomic Risk Scores.
- PubMed – Peer-reviewed studies on APOL1 genetic variants and renal failure.
- World Health Organization (WHO) – Global reports on Non-Communicable Diseases (NCDs) and kidney health.
- Centers for Disease Control and Prevention (CDC) – Chronic Kidney Disease Surveillance System.