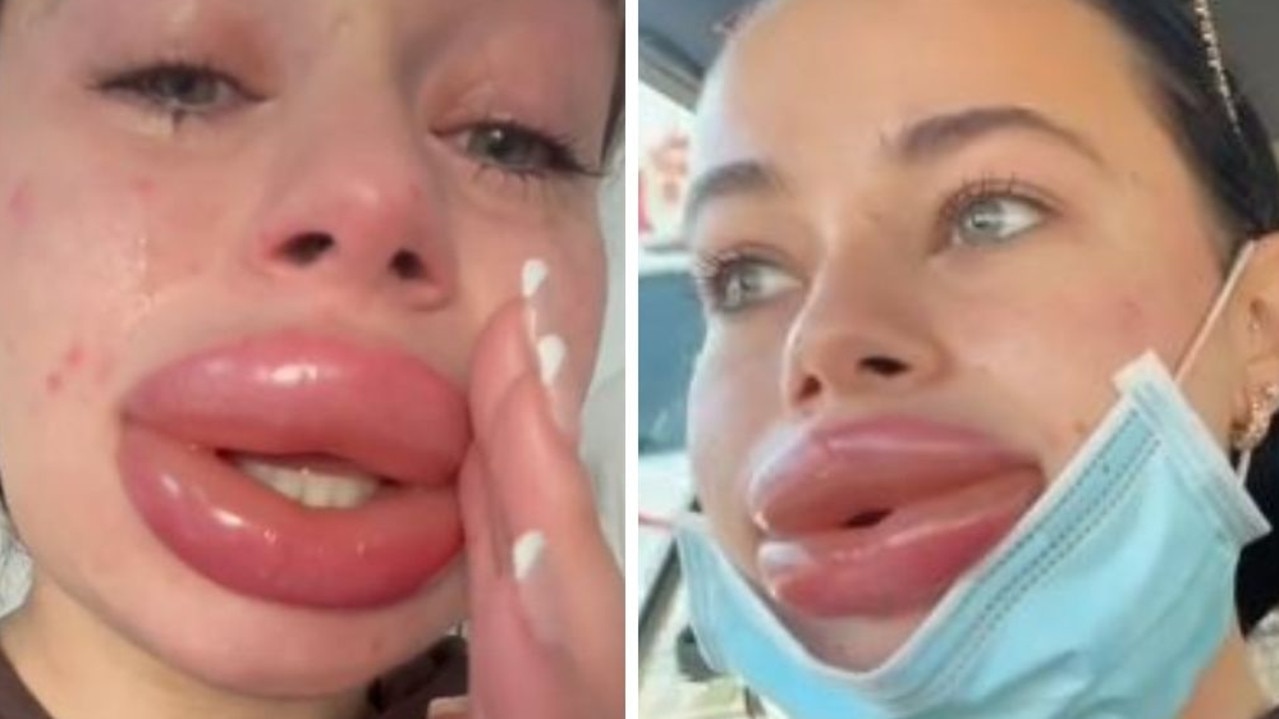
Mandy, a 34-year-old woman, experienced significant medical complications following low-cost lip filler injections. This case highlights the clinical risks associated with unregulated cosmetic procedures, where extreme price-cutting often bypasses essential medical oversight, leading to potential vascular occlusion, inflammatory granulomas, or severe tissue necrosis.
The decision to undergo aesthetic enhancement for a nominal fee of €300 is not merely a financial choice; it is a clinical gamble. In the rapidly expanding global market for non-surgical cosmetic procedures, a dangerous “shadow economy” has emerged. This economy operates on the periphery of established healthcare systems, offering high-risk dermal fillers at prices that fail to account for the cost of medical-grade materials, sterile environments, or the professional expertise required to navigate complex facial anatomy.
In Plain English: The Clinical Takeaway
- Price is a proxy for safety: Low-cost fillers often utilize sub-standard materials that lack the rigorous testing required by health authorities like the EMA or FDA.
- Anatomy is complex: The lips are densely packed with blood vessels; incorrect injection techniques can lead to permanent tissue death or blindness.
- Not all “swellings” are normal: While minor bruising is expected, lumps, persistent pain, or skin discoloration are signs of serious medical emergencies.
The Biology of Volume: Mechanism of Action and Cross-linking
Most modern lip fillers utilize Hyaluronic Acid (HA), a naturally occurring polysaccharide (a complex sugar) found in the human extracellular matrix. To ensure the filler remains in the tissue rather than being immediately absorbed by the body, manufacturers use a process called cross-linking. This involves adding a chemical agent, typically polybutylene glycol diglycidyl sebacate (BDDE), to bond the HA molecules into a long-lasting gel.
When a practitioner uses low-grade or unregulated products, the concentration of cross-linking agents may be inconsistent, or the sterilization protocols may be insufficient. This increases the risk of delayed-onset hypersensitivity—an immune system overreaction where the body treats the filler as a foreign invader, leading to chronic inflammation or the formation of granulomas (hard, inflammatory lumps).
the physical placement of the gel is critical. If the substance is injected into an artery rather than the subcutaneous tissue (the layer just below the skin), it can cause vascular occlusion. This occurs when the filler physically blocks blood flow, depriving the surrounding tissue of oxygen and nutrients, which can lead to necrosis (localized cell death).
The Clinical Spectrum of Dermal Filler Complications
The following table summarizes the primary clinical risks associated with non-professional or low-quality filler applications:

| Complication | Clinical Description | Mechanism of Action | Urgency Level |
|---|---|---|---|
| Vascular Occlusion | Blockage of blood vessels in the lip or surrounding area. | Physical displacement of filler into an artery. | EMERGENCY |
| Granuloma | Hard, palpable lumps forming weeks or months later. | Chronic immune response to foreign material. | High (Requires Specialist) |
| Tyndall Effect | A bluish discoloration under the skin. | Filler placed too superficially, scattering light. | Moderate (Aesthetic) |
| Infection | Redness, warmth, pus, and systemic fever. | Breach of sterile technique during injection. | High (Requires Antibiotics) |
Regulatory Disparities and the Rise of Shadow Clinics
The case of Mandy underscores a significant failure in regional healthcare enforcement. In the European Union, the European Medicines Agency (EMA) regulates dermal fillers as medical devices, requiring strict adherence to safety and efficacy standards. However, the rise of “discount aesthetics” allows practitioners to bypass these regulations by operating in unregulated settings or using products sourced from non-compliant manufacturers.
In the United States, the Food and Drug Administration (FDA) maintains similar oversight, yet the “wellness” industry often blurs the line between medical procedures and spa services. This regulatory gap creates a “geo-epidemiological” risk: patients may travel to regions with laxer enforcement to find lower prices, inadvertently exposing themselves to higher biological risks. The lack of a centralized registry for these “shadow” injections makes it incredibly difficult for medical professionals to treat complications, as they often do not know the exact chemical composition of the substance injected.
“The democratization of aesthetic medicine has outpaced the regulation of its practitioners. When the cost of a procedure is lower than the cost of the sterile equipment and the high-grade HA gel itself, the patient is no longer receiving a medical service; they are participating in an unregulated biological experiment.”
Note: The above sentiment reflects the consensus of dermatological safety protocols observed by major medical bodies.
Contraindications & When to Consult a Doctor
Dermal fillers are not suitable for everyone. Individuals with certain medical histories should exercise extreme caution or avoid these procedures entirely:
- Autoimmune Disorders: Conditions like lupus or rheumatoid arthritis increase the risk of severe inflammatory responses to fillers.
- Active Infections: Any current infection in the treatment area or systemic infection (e.g., influenza) is an absolute contraindication.
- Pregnancy and Lactation: There is insufficient longitudinal data to guarantee the safety of dermal fillers for pregnant or breastfeeding individuals.
Seek immediate medical attention if you experience:
- Sudden, intense pain at the injection site.
- Skin blanching (the skin turning white or mottled) following an injection.
- Visual changes or extreme pain in the eyes (a sign of proximal vascular occlusion).
- Rapidly spreading redness or warmth.
The Path Forward: Prioritizing Clinical Integrity
As the demand for aesthetic enhancements continues to climb, the medical community must advocate for stricter practitioner licensing and more transparent product traceability. For the patient, the most effective tool for safety is skepticism. A price point that seems “too good to be true” is often a direct indicator that the clinical safety margins—sterility, material quality, and anatomical expertise—have been compromised.

Future public health initiatives must focus on educating consumers that aesthetic medicine is, fundamentally, medical intervention. Treating it as a mere beauty service is a misconception that carries profound biological consequences.
References
- PubMed: Clinical Studies on Hyaluronic Acid Cross-linking and Safety
- The Lancet: Global Trends in Non-Surgical Cosmetic Complications
- European Medicines Agency (EMA): Medical Device Regulation Guidelines
- World Health Organization (WHO): Medical Device Safety Standards
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional for medical concerns.