A routine blood test measuring phosphorylated tau protein (p-tau217) levels may identify individuals at elevated risk of developing Alzheimer’s disease up to 15 years before clinical symptoms emerge, according to recent longitudinal research. This biomarker detects early neurodegenerative changes associated with amyloid-beta plaques and tau tangles, offering a non-invasive, scalable tool for early intervention strategies in primary care settings worldwide.
How a Simple Blood Test Could Reshape Alzheimer’s Prevention
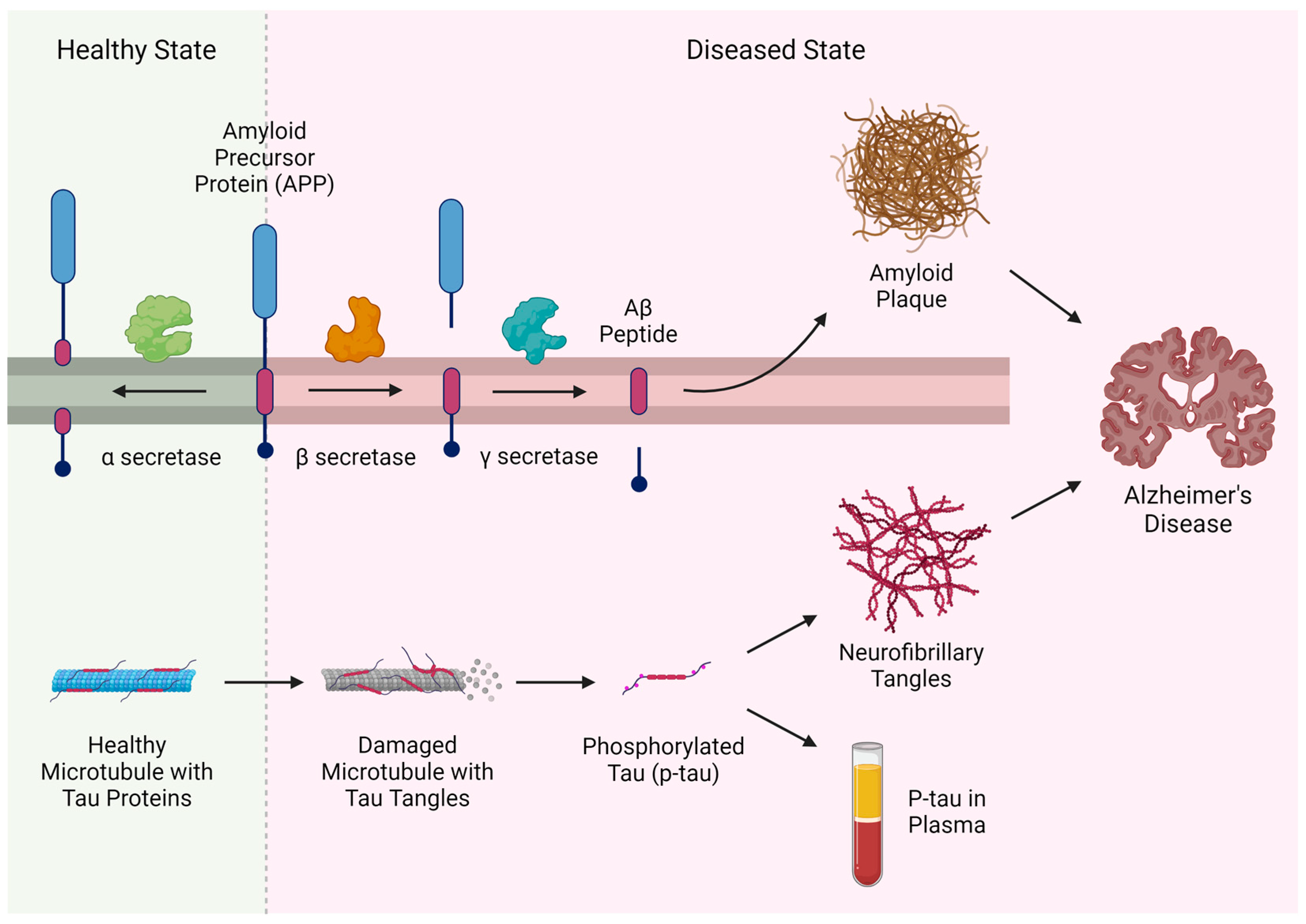
The blood-based p-tau217 assay detects phosphorylated tau, a specific form of the tau protein that becomes abnormally modified in Alzheimer’s pathology. In healthy neurons, tau stabilizes microtubules; in Alzheimer’s, hyperphosphorylation causes tau to detach and form neurofibrillary tangles, disrupting cellular transport. Unlike amyloid-beta, which accumulates years earlier, p-tau217 rises closer to symptom onset but still precedes cognitive decline by a decade or more, making it a critical window for therapeutic intervention.
In Plain English: The Clinical Takeaway
- This test does not diagnose Alzheimer’s but identifies biological risk years before memory loss appears.
- A negative result significantly lowers near-term risk, while a positive result warrants further evaluation, not alarm.
- Early detection allows lifestyle interventions and clinical trial participation when therapies are most likely to leisurely progression.
Validation Across Global Cohorts and Clinical Trial Integration
Research published in Nature Medicine validated the p-tau217 blood test across 1,200 participants from the Swedish BioFINDER cohort and the U.S.-based Alzheimer’s Disease Neuroimaging Initiative (ADNI), showing 96% accuracy in distinguishing Alzheimer’s from other dementias when compared to PET scans or cerebrospinal fluid analysis. The test is now being incorporated into secondary prevention trials like the NIH-funded A4 Study (Anti-Amyloid Treatment in Asymptomatic Alzheimer’s), which targets cognitively normal individuals with elevated amyloid burden. As of 2025, over 300 sites worldwide use p-tau217 screening to enrich trial populations, reducing screen failure rates by 40%.
“Blood-based biomarkers like p-tau217 are transforming Alzheimer’s research from a symptomatic to a pre-symptomatic paradigm, enabling us to test disease-modifying therapies at the earliest possible stage.”
— Dr. Oskar Hansson, Professor of Neurology, Lund University, lead author of the 2024 Nature Medicine validation study
Geo-Epidemiological Bridging: From Lab to Local Clinics
In the United States, the FDA has not yet cleared p-tau217 as a diagnostic tool but recognizes its use in clinical research under the Biomarker Qualification Program. The EMA has similarly endorsed its use in enrichment strategies for lecanemab and donanemab trials. In the UK’s NHS, pilot programs in Greater Manchester and Scotland are evaluating p-tau217 alongside cognitive assessments in memory clinics, aiming to reduce reliance on expensive PET scans or lumbar punctures. In low-resource settings, the test’s simplicity—requiring only a standard venipuncture and automated immunoassay—could democratize access where neurology specialists and imaging infrastructure are scarce.
Funding, Independence, and Scientific Rigor
The foundational research was supported by the Swedish Research Council, the European Union’s Horizon 2020 program, and the U.S. National Institutes of Health (grant R01AG057732). Industry partners including Eli Lilly and Roche provided assay reagents under strict data-use agreements, with no influence on study design or publication. All authors disclosed potential conflicts, and the study underwent blind peer review. No financial compensation was tied to outcome positivity, minimizing bias risk.
| Biomarker | Detection Window Before Symptoms | Accuracy vs. CSF/PET | Invasiveness |
|---|---|---|---|
| p-tau217 (blood) | 10-15 years | 96% | Minimal (venipuncture) |
| Amyloid-PET | 15-20 years | 95% | High (radiation exposure) |
| CSF p-tau | 10-15 years | 94% | High (lumbar puncture) |
Contraindications & When to Consult a Doctor
This test is not recommended for individuals under 50 without family history or genetic risk factors (e.g., APOE4 homozygosity), as false positives may cause unnecessary distress. It should never replace clinical evaluation for memory concerns. Anyone experiencing progressive short-term memory loss, difficulty completing familiar tasks, or disorientation in known environments should seek prompt medical assessment, regardless of biomarker results. A positive p-tau217 result requires confirmation via neurologist-led cognitive testing and structural MRI to rule out reversible causes like vitamin B12 deficiency, thyroid dysfunction, or normal pressure hydrocephalus.

The Path Forward: Prevention Over Prediction
While no blood test alone can prevent Alzheimer’s, integrating p-tau217 screening into midlife cardiovascular risk assessments—similar to cholesterol screening for heart disease—could shift neurology from reactive to preventive medicine. Trials investigating antihypertensives, exercise regimens, and cognitive training in biomarker-positive individuals are ongoing. The ultimate goal is not to label risk but to empower action: treating brain health as a modifiable, lifelong investment rather than an inevitable decline.
References
- Palmqvist S, et al. Blood phosphorylated tau 217 as a biomarker for Alzheimer’s disease. Nat Med. 2024.
- Janelidze S, et al. Performance of blood phospho-tau assays in identifying Alzheimer pathology. Alzheimers Dement. 2023.
- Sawyer G, et al. Blood-based biomarkers in the A4 Study: Screening and enrichment. JAMA Neurol. 2023.
- National Institute on Aging. Alzheimer’s Disease Biomarkers. NIH.
- World Health Organization. Dementia Fact Sheet. WHO, 2023.

