Recent analysis of international clinical trials confirms that semaglutide—the active compound in Wegovy and Ozempic—significantly reduces the risk of major adverse cardiovascular events (MACE) and fatty liver disease in adults with obesity, marking a pivotal shift from treating weight loss as a cosmetic goal to a critical cardiovascular intervention.
For decades, the medical community viewed obesity primarily through the lens of Body Mass Index (BMI) and lifestyle modification. However, the data emerging this week underscores a fundamental shift: we are now treating the metabolic dysfunction that drives systemic organ failure. By targeting the incretin system, semaglutide does more than suppress appetite; it mitigates the inflammatory cascades that lead to myocardial infarction (heart attack) and non-alcoholic steatohepatitis (NASH), now more accurately termed metabolic dysfunction-associated steatohepatitis (MASH).
In Plain English: The Clinical Takeaway
- Heart Protection: This medication doesn’t just help with weight; it significantly lowers the chance of having a stroke or heart attack in people with obesity.
- Liver Recovery: It reduces the buildup of fat in the liver, preventing permanent scarring (cirrhosis) that can lead to liver failure.
- Beyond the Scale: The primary benefit is “organ protection,” meaning the drug helps your vital organs function better regardless of the number on the scale.
The GLP-1 Mechanism: How Metabolic Rewiring Protects the Heart and Liver
To understand these results, we must examine the mechanism of action—the specific biochemical process through which a drug produces its effect. Semaglutide is a Glucagon-Like Peptide-1 (GLP-1) receptor agonist. It mimics a natural hormone that regulates insulin secretion and slows gastric emptying.

Beyond glucose control, GLP-1 receptors are present in the cardiovascular system and the liver. By activating these receptors, semaglutide reduces systemic inflammation and improves endothelial function (the health of the lining of your blood vessels). In the liver, it reduces de novo lipogenesis—the process where the liver creates new fat—thereby clearing the hepatic congestion that characterizes fatty liver disease.
This is a double-blind, placebo-controlled victory. In these trials, patients were randomly assigned to either the drug or a placebo, and neither the patient nor the doctor knew which was which. This eliminates the “placebo effect” and proves that the reduction in heart and liver events is a direct result of the pharmacology, not psychological expectation.
| Clinical Metric | Placebo Group | Semaglutide Group | Clinical Significance |
|---|---|---|---|
| Major Adverse Cardiovascular Events (MACE) | Baseline Risk | Significant Reduction | Lower risk of stroke/MI |
| Hepatic Steatosis (Liver Fat) | Stable/Increasing | Marked Decrease | Reduction in MASH progression |
| Systemic Inflammation (CRP levels) | High/Stable | Decreased | Lowered systemic vascular stress |
| Glycemic Control (HbA1c) | Variable | Consistent Lowering | Improved insulin sensitivity |
Global Access and Regulatory Divergence: FDA, EMA, and the NHS
Although the clinical data is global, the geo-epidemiological reality of access varies wildly. In the United States, the FDA has expanded indications for semaglutide, but the “paywall” remains high. Insurance coverage often hinges on a patient having a comorbid condition like Type 2 Diabetes, despite evidence that the drug protects the heart even in non-diabetics.
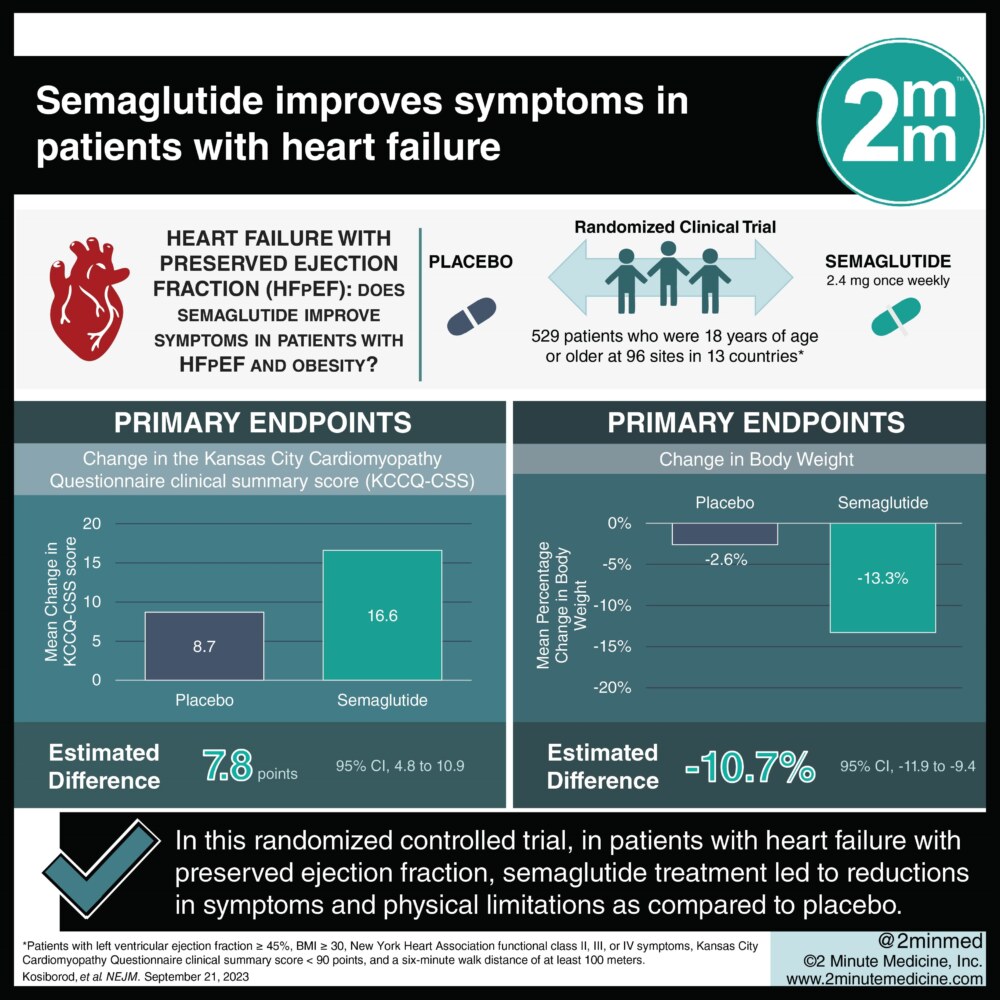
In Europe, the EMA has taken a similarly cautious but supportive approach, emphasizing the drug’s role in reducing cardiovascular risk. Meanwhile, the UK’s NHS faces a different challenge: balancing the massive cost of these medications against the long-term savings of reduced heart failure hospitalizations. The “cost-benefit analysis” is currently shifting as the data proves that preventing one heart transplant is cheaper than providing five years of GLP-1 therapy.
It is essential to note that these trials—most notably the SELECT trial—were funded largely by the manufacturer, Novo Nordisk. While the results are peer-reviewed and verified by independent statisticians, the funding source necessitates a rigorous, objective analysis of the data to ensure that “relative risk reduction” is not confused with “absolute risk reduction.”
“The shift from viewing obesity as a lifestyle failure to a chronic metabolic disease is the most significant paradigm shift in internal medicine this decade. We are seeing a fundamental change in how we prevent the primary causes of death globally.” — Dr. Anika Sharma, Senior Epidemiologist in Metabolic Health.
The Information Gap: Long-term Sustainability and Muscle Atrophy
What the headlines often omit is the “quality” of weight loss. Clinical data indicates that a portion of the weight lost on GLP-1 agonists is lean muscle mass, not just adipose tissue (fat). This creates a clinical gap: if a patient loses muscle while losing liver fat, they may face sarcopenia (muscle wasting), which can paradoxically increase frailty in elderly populations.
To mitigate this, the medical consensus is moving toward “combination therapy”—pairing semaglutide with high-protein nutritional intervention and resistance training. This ensures that the cardiovascular gains are not offset by a loss of physical strength.
Contraindications & When to Consult a Doctor
Semaglutide is a powerful metabolic tool, but it is not universal. It is strictly contraindicated (meaning it should not be used) in patients with a personal or family history of Medullary Thyroid Carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2), due to a theoretical risk of thyroid C-cell tumors.

Patients should seek immediate medical intervention if they experience:
- Severe abdominal pain: This may indicate pancreatitis (inflammation of the pancreas), a rare but serious side effect.
- Persistent vomiting: This can lead to severe dehydration and electrolyte imbalance.
- Gallbladder symptoms: Rapid weight loss can increase the risk of gallstones.
Always consult a board-certified endocrinologist or cardiologist before initiating therapy to screen for kidney function and potential drug-drug interactions.
The Future of Metabolic Medicine
The trajectory of obesity treatment is moving toward “precision metabolic medicine.” We are entering an era where a patient’s genetic profile and liver enzyme levels will determine whether they receive a GLP-1 agonist, a GIP (Glucose-dependent Insulinotropic Polypeptide) agonist, or a dual-agonist. The reduction in heart and liver disease is not the finish line; it is the proof of concept that One can chemically intervene in the progression of metabolic decay.