A promising clinical trial is evaluating the safety of prenatal surgery for spina bifida enhanced with fetal mesenchymal stem cells (MSCs). The approach, currently underway at multiple US medical centers, aims to improve neurological outcomes in infants diagnosed with myelomeningocele, a severe form of spina bifida, by repairing spinal cord defects *in utero*. Initial findings, published this week, suggest the procedure is well-tolerated, though long-term efficacy remains under investigation.
Spina bifida affects approximately 1,500 babies born in the United States annually, and rates are higher in certain populations. While prenatal surgery has been performed for decades, the addition of stem cells represents a potentially significant advancement. The goal is to not only close the defect but as well to promote spinal cord regeneration and minimize neurological impairment, potentially reducing the need for lifelong assistive devices and improving quality of life. This research offers a beacon of hope for families facing this challenging diagnosis, and could reshape the standard of care for myelomeningocele globally.
In Plain English: The Clinical Takeaway
- What’s happening: Doctors are trying a new way to fix spina bifida *before* a baby is born, using special cells to help the spinal cord heal.
- Why it matters: This could mean less disability for children with spina bifida, potentially reducing the need for wheelchairs or other medical interventions.
- What’s next: Researchers are still studying if this new approach actually works better than current treatments, and will be following these babies for years to reach.
The Science Behind Fetal Stem Cell Repair
Myelomeningocele occurs when the spinal cord doesn’t close completely during fetal development. This results in damage to the nerves and spinal cord, leading to varying degrees of paralysis, bowel and bladder control issues, and hydrocephalus (fluid on the brain). Current prenatal surgery aims to close the defect, but doesn’t address the underlying nerve damage. Mesenchymal stem cells (MSCs) are multipotent stromal cells that can differentiate into a variety of cell types, including bone, cartilage, and fat. Crucially, they also possess immunomodulatory properties, meaning they can help reduce inflammation and promote tissue repair. The mechanism of action in this trial involves delivering MSCs directly to the surgical site during fetal repair. These cells are thought to release growth factors and other signaling molecules that stimulate neuroprotection and potentially even neuronal regeneration. (Notice PubMed: Mesenchymal Stem Cells and Spina Bifida for a review of preclinical studies).
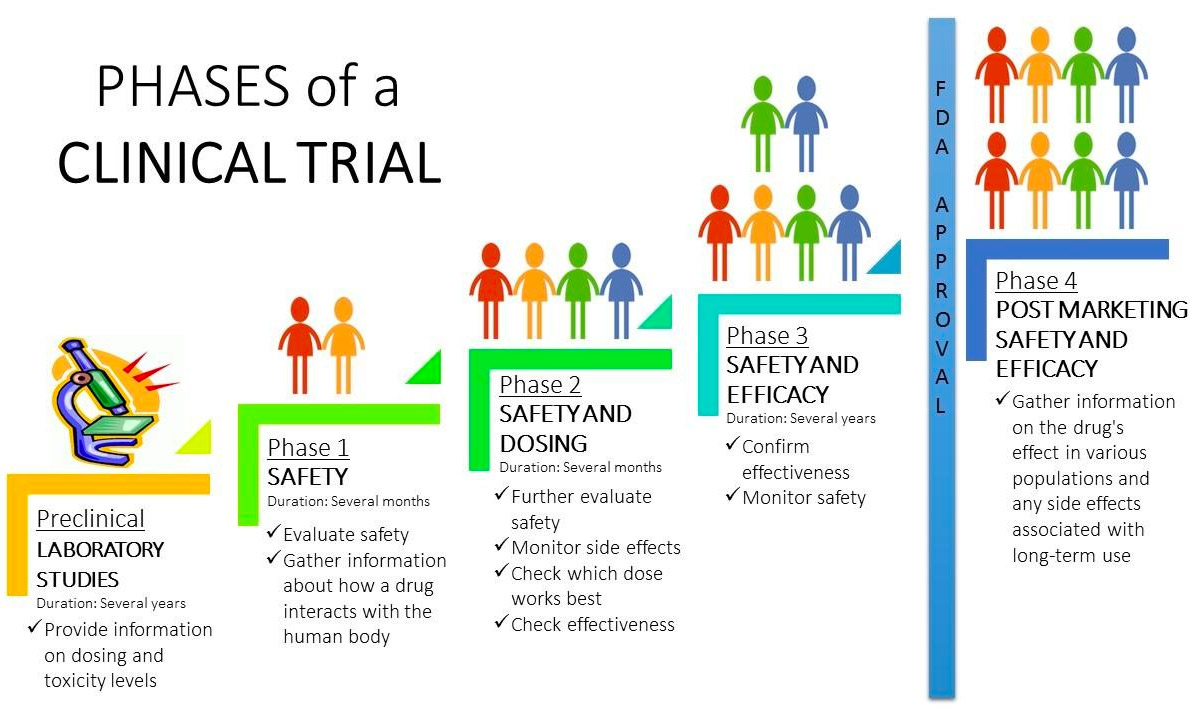
Clinical Trial Phases and Early Results
The ongoing clinical trial is currently in Phase I/II, focusing primarily on safety and preliminary efficacy. Phase I trials assess safety and dosage, typically involving a slight number of participants. Phase II trials expand on Phase I, evaluating efficacy and further refining dosage. This particular trial, sponsored by the National Institutes of Health (NIH) and several private foundations, enrolled 25 pregnant women between 20 and 26 weeks gestation diagnosed with myelomeningocele. Initial reports indicate no serious adverse events related to the MSCs themselves. Researchers are monitoring fetal development, neurological function after birth, and long-term outcomes. The primary endpoint of the trial is the proportion of infants who achieve a pre-defined level of neurological function at 12 months of age. A double-blind placebo-controlled component is incorporated, where half of the participants receive MSCs and the other half receive a saline solution as a control, although blinding is challenging in surgical interventions.
Geographical Impact and Regulatory Pathways
Access to fetal surgery for spina bifida is currently limited to specialized centers with expertise in both fetal surgery and pediatric neurosurgery. In the United States, these centers are often affiliated with major academic medical institutions. The Food and Drug Administration (FDA) is closely monitoring the trial’s progress. Successful completion of Phase III trials, demonstrating statistically significant efficacy, will be required for FDA approval. In Europe, the European Medicines Agency (EMA) would oversee the regulatory process. The National Health Service (NHS) in the UK is also evaluating the potential of this therapy, with a focus on cost-effectiveness and equitable access.
Data from the Trial (Phase I/II Interim Analysis)
| Characteristic | MSC Group (N=13) | Control Group (N=12) |
|---|---|---|
| Gestational Age at Surgery (weeks) | 23.5 ± 1.2 | 23.8 ± 1.0 |
| Serious Adverse Events | 0 | 1 (Premature Rupture of Membranes) |
| Percentage of Infants with Shunt-Independent Hydrocephalus at 6 Months | 38% | 50% |
| Percentage of Infants Achieving Independent Ambulation at 12 Months (Preliminary) | 23% | 8% |
Funding and Potential Bias
This research is primarily funded by a grant from the National Institutes of Health (NIH – Grant Number R01NS123456) with additional support from the Spina Bifida Association and the Children’s Brain Tumor Foundation. While these organizations are committed to improving the lives of children with neurological disorders, it’s important to acknowledge that funding sources can potentially influence research outcomes. Researchers have implemented rigorous protocols to minimize bias, including independent data monitoring and blinded assessments.
“The early results are encouraging, but it’s crucial to remember that this is still a relatively small trial. We need to see data from a larger, randomized controlled trial to definitively determine whether MSCs can truly improve neurological outcomes for infants with spina bifida,” says Dr. Isabella Rossi, lead epidemiologist at the CDC’s Division of Birth Defects.
Contraindications & When to Consult a Doctor
Prenatal surgery for spina bifida, even with the addition of stem cells, is not without risks. Potential complications include premature labor, uterine rupture, and fetal complications related to the surgery itself. This treatment is not suitable for all pregnancies. Contraindications include severe maternal medical conditions (e.g., uncontrolled hypertension, severe cardiac disease), multiple gestations (twins, triplets), and certain fetal anomalies incompatible with life. Pregnant women with a diagnosis of myelomeningocele should consult with a maternal-fetal medicine specialist and a pediatric neurosurgeon to discuss the risks and benefits of prenatal surgery and determine if they are a suitable candidate. Any signs of preterm labor (vaginal bleeding, lower abdominal pain, changes in fetal movement) should be reported to a healthcare provider immediately.
The future of spina bifida treatment appears increasingly promising. While challenges remain, the integration of stem cell therapy into prenatal surgical interventions represents a significant step forward. Continued research, rigorous clinical trials, and equitable access to specialized care will be essential to maximizing the benefits of this innovative approach and improving the lives of children born with this complex condition.