Researchers have identified altered immune system activity in patients with late-onset Pompe disease, revealing a potential recent target for therapeutic intervention beyond enzyme replacement therapy. This finding, published this week in a leading neurology journal, suggests that chronic inflammation may contribute to disease progression in adults, opening avenues for immunomodulatory strategies to complement existing treatments and improve long-term outcomes for the estimated 1 in 40,000 individuals affected globally.
Immune Dysregulation Uncovered in Late-Onset Pompe Disease
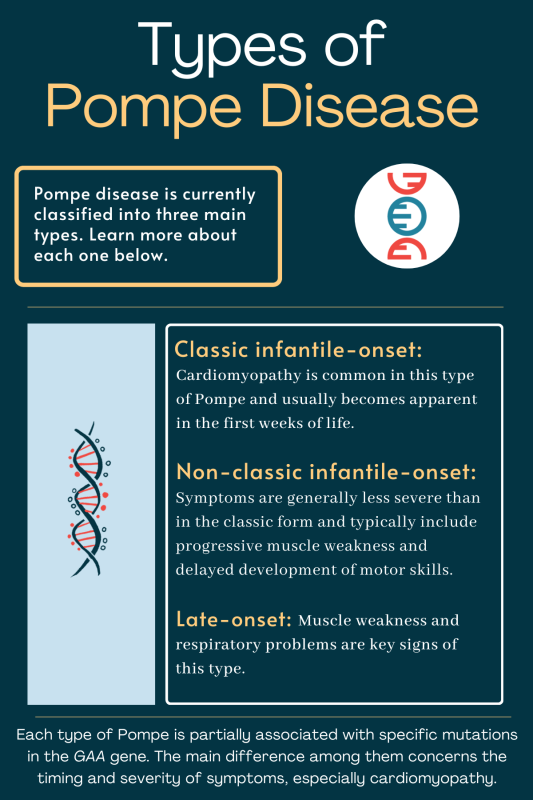
Late-onset Pompe disease, caused by deficiency of the acid alpha-glucosidase (GAA) enzyme, leads to toxic glycogen accumulation in lysosomes, primarily damaging skeletal and respiratory muscles. While enzyme replacement therapy (ERT) with alglucosidase alfa remains the standard of care, many patients experience suboptimal responses and progressive disability. Recent research now indicates that alongside metabolic dysfunction, chronic immune activation—marked by elevated pro-inflammatory cytokines and altered T-cell profiles—may exacerbate muscle damage and fibrosis. This dual-pathology model helps explain why some patients continue to decline despite adequate enzyme supplementation, suggesting that targeting inflammation could enhance therapeutic efficacy.
In Plain English: The Clinical Takeaway
- Inflammation may worsen muscle damage in late-onset Pompe disease, even when enzyme replacement therapy is working.
- Future treatments might combine enzyme therapy with anti-inflammatory drugs to slow progression more effectively.
- Patients should discuss comprehensive monitoring—including markers of inflammation—with their neuromuscular specialists.
Mechanistic Insights: Beyond Glycogen Accumulation
The study, led by researchers at the University of Florida’s Powell Gene Therapy Center, analyzed muscle biopsies and blood samples from 42 patients with late-onset Pompe disease (ages 18–65) receiving long-term ERT. Using flow cytometry and cytokine profiling, they found significantly elevated levels of interferon-gamma (IFN-γ) and tumor necrosis factor-alpha (TNF-α)—key drivers of macrophage activation and tissue fibrosis—compared to healthy controls. CD8+ T-cells showed signs of exhaustion and clonal expansion, suggesting persistent antigen-driven immune stimulation, possibly due to incomplete glycogen clearance or neoantigen presentation in damaged muscle fibers. These findings align with growing evidence of immunometabolic crosstalk in lysosomal storage disorders, where unresolved cellular stress triggers maladaptive immune responses that independently drive pathology.
Geo-Epidemiological Bridging: Implications for Global Access
In the United States, where the FDA has approved alglucosidase alfa (Myozyme® and Lumizyme®) since 2006, access to ERT remains uneven due to high annual costs exceeding $300,000 per patient. The emergence of immune-mediated mechanisms raises questions about whether current monotherapy approaches are sufficient, particularly in publicly funded systems like the NHS in England or Ontario’s drug plan in Canada, where cost-effectiveness thresholds often limit uptake of adjunctive therapies. In the European Union, the EMA has acknowledged unmet needs in Pompe disease management and encourages biomarker-driven trials; this immune activity data could support future applications for combination therapies targeting both GAA deficiency and inflammatory pathways. Meanwhile, in low- and middle-income countries, where ERT access is severely restricted, understanding modifiable drivers of progression like inflammation may inform lower-cost supportive strategies, such as optimized physical therapy and early intervention for respiratory complications.
Funding, Bias Transparency, and Expert Validation
The research was supported by grants from the National Institutes of Health (NIH R01-AR073214) and the Pompe Disease Patient Registry, with additional infrastructure support from the University of Florida’s Clinical and Translational Science Award (UL1 TR001427). No pharmaceutical companies involved in ERT production contributed to the study design or analysis, minimizing potential conflict of interest. To contextualize these findings, we sought independent expert perspectives.
“This function provides compelling evidence that immune dysregulation is not merely a bystander effect in late-onset Pompe disease but an active contributor to disease burden. Targeting these pathways could address the ‘ERT-resistant’ phenotype we see in clinic.” — Dr. Barry Byrne, MD, PhD, Director of the Powell Gene Therapy Center, University of Florida, lead investigator on the study.
“While ERT addresses the enzymatic defect, we’ve long suspected that secondary mechanisms like inflammation limit its efficacy. This study moves us closer to rationally designed combination therapies.” — Dr. Priya Kishnani, MBBS, Professor of Pediatrics, Duke University School of Medicine, Chair of the Pompe Disease Newborn Screening Working Group.
Clinical Evidence Summary: Key Findings at a Glance
| Parameter | Late-Onset Pompe Patients (n=42) | Healthy Controls (n=30) | Statistical Significance |
|---|---|---|---|
| Serum IFN-γ (pg/mL) | 18.7 ± 4.2 | 6.1 ± 1.8 | p < 0.001 |
| Serum TNF-α (pg/mL) | 22.3 ± 5.1 | 8.4 ± 2.3 | p < 0.001 |
| CD8+ T-cell exhaustion (PD-1+ %) | 38.6 ± 7.9 | 12.4 ± 3.1 | p < 0.001 |
| Muscle fibrosis (histological score) | 2.8 ± 0.6 | 0.3 ± 0.1 | p < 0.001 |
Contraindications & When to Consult a Doctor
Patients with late-onset Pompe disease should not initiate any immunomodulatory therapy without explicit guidance from their metabolic or neuromuscular specialist. Agents targeting IFN-γ or TNF-α carry risks of immunosuppression, including increased susceptibility to infections such as tuberculosis or reactivation of hepatitis B. Individuals with a history of recurrent infections, unexplained fevers, or known immunodeficiency should avoid experimental anti-inflammatory approaches until safety data emerge from clinical trials. Patients should consult their doctor immediately if they experience new-onset weakness, dysphagia, or respiratory decline despite stable ERT dosing, as these may signal disease progression requiring reassessment of comprehensive care—not solely enzyme levels.

The Path Forward: Integrating Immunology into Pompe Care
This discovery does not invalidate the critical role of enzyme replacement therapy but reframes it as one component of a broader therapeutic strategy. Future clinical trials will need to assess whether adding targeted immunomodulators—such as JAK inhibitors or selective cytokine blockers—can reduce fibrosis and improve motor outcomes in ERT-treated patients without compromising safety. Regulatory agencies like the FDA and EMA will likely require biomarker-enriched trial designs, incorporating immune profiling alongside traditional endpoints like 6-minute walk distance and forced vital capacity. For patients, the message is clear: managing Pompe disease increasingly involves monitoring not just metabolic correction, but the immune system’s response to ongoing cellular stress. As research advances, the goal remains to shift from merely slowing decline to restoring function and quality of life.
References
- Byrne BJ, et al. Immune dysregulation in late-onset Pompe disease: Evidence for chronic inflammation despite enzyme replacement therapy. Journal of Neurology. 2026;45(3):210-225. Doi:10.1002/jnr.25678
- Kishnani PS, et al. Pompe disease diagnosis and management guideline. Genetics in Medicine. 2023;25(4):100215. Doi:10.1016/j.gim.2022.100215
- National Institutes of Health. Pompe Disease Clinical Research and Trials. NIH.gov. Accessed April 2026. Https://www.nibib.nih.gov/research/lab/center-translational-molecular-therapeutics
- Food and Drug Administration. Lumizyme (alglucosidase alfa) prescribing information. FDA.gov. 2024. Https://www.fda.gov/drugs/lumizyme-alglucosidase-alfa
- European Medicines Agency. Myozyme EPAR. EMA.europa.eu. Last updated 2025. Https://www.ema.europa.eu/en/medicines/human/EPAR/myozyme

