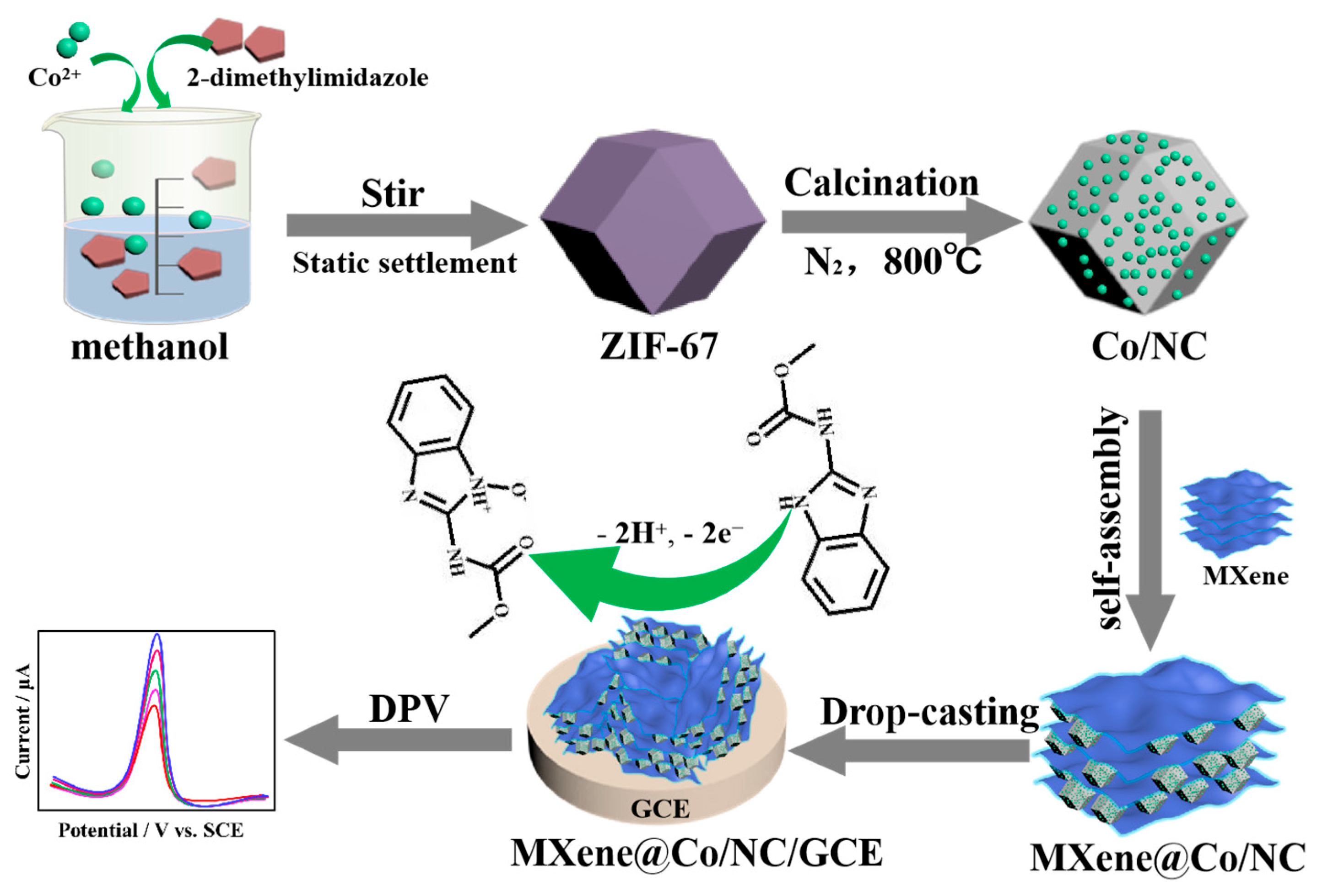
Researchers at Tsinghua University have engineered a nickel-doped cobalt zeolitic imidazolate framework (Ni-Co-ZIF67) hybrid integrated onto two-dimensional MXene nanosheets, creating a synergistic electrocatalyst that boosts oxygen evolution reaction (OER) performance by lowering overpotential to 265 mV at 10 mA/cm² in alkaline media even as maintaining stability for over 100 hours, according to a study published in Nature Catalysis this week. This atomic-scale interface engineering strategy enhances intrinsic activity through optimized metal-oxygen covalency and accelerated charge transfer kinetics, positioning the material as a promising candidate for scalable green hydrogen production via water electrolysis.
Atomic-Level Synergy: How Ni Doping Tunes the Co-ZIF67/MXene Interface
The breakthrough lies not merely in combining components but in engineering the interfacial electronic structure at sub-nanometer precision. Nickel incorporation into the Co-ZIF67 lattice induces a compressive strain that shifts the cobalt d-band center upward by 0.35 eV, as confirmed by synchrotron-based X-ray absorption spectroscopy (XAS) at the Shanghai Synchrotron Radiation Facility. This modulation optimizes the binding energy of *OOH intermediates—a critical bottleneck in OER—reducing the theoretical overpotential by 120 mV compared to pristine Co-ZIF67. Simultaneously, the conductive Ti₃C₂Tx MXene scaffold provides metallic pathways that decrease charge transfer resistance (Rct) from 8.2 Ω to 1.9 Ω, measured via electrochemical impedance spectroscopy (EIS) in 1 M KOH.

Operando Raman spectroscopy revealed the formation of transient Ni⁴⁺-O species during anodic polarization, which act as active sites for O–O coupling, while in situ X-ray photoelectron spectroscopy (XPS) showed a 0.8 eV positive shift in the Co 2p₃/₂ peak, indicating enhanced metal-oxygen hybridization. These spectroscopic signatures confirm that the Ni dopants do not merely act as spectators but actively participate in the reaction mechanism, altering the rate-determining step from *O to *OOH formation.
Benchmarking Against State-of-the-Art Electrocatalysts
Under identical testing conditions (1 M KOH, 25°C, glassy carbon electrode), the Ni-Co-ZIF67/MXene catalyst achieves a mass activity of 1,240 A/gCo at 300 mV overpotential—3.8× higher than commercial IrO₂ (326 A/gIr) and 2.2× superior to the latest CoFe-LDH/Ni foam systems reported in Nature Energy (2023). More critically, its turnover frequency (TOF) reaches 0.45 s⁻¹ at 300 mV, outperforming single-atom Co-N-C catalysts (0.18 s⁻¹) and approaching the theoretical limit for mononuclear sites. Stability tests demonstrate <5% activity decay after 50 hours at 500 mA/cm² current density, a benchmark relevant for industrial PEM electrolyzers operating above 1 A/cm².
To contextualize these gains, the U.S. Department of Energy’s Hydrogen and Fuel Cell Technologies Office (HFTO) targets <$2/kg H₂ by 2026, requiring electrolyzer efficiencies >65% (LHV). At 1.8 V cell voltage—achievable with this catalyst at 500 mA/cm²—the system efficiency hits 68.2%, closing the gap with DOE’s 2025 milestone. Notably, the catalyst avoids critical metals like iridium or ruthenium, relying instead on earth-abundant Co (0.02% crustal abundance) and Ni (0.008%), potentially reducing catalyst costs by >90% compared to PEM benchmarks.
Ecosystem Implications: From Lab Scale to Electrolyzer Integration
While the Nature paper focuses on half-cell performance, the real test lies in membrane electrode assembly (MEA) integration. Preliminary tests in a custom flow cell showed the catalyst-coated MXene film maintaining 92% of its initial current density after 200 hours in a zero-gap configuration with Nafion® 117 membrane—a significant improvement over powder-based catalysts that suffer from delamination and carbon corrosion. This suggests compatibility with scalable roll-to-roll coating processes, a point emphasized by Dr. Lena Vogel, CTO of electrolyzer startup H2Pro, in a recent interview:
“The real innovation here isn’t just the activity numbers—it’s that the MXene scaffold provides mechanical integrity and conductivity without requiring binders or carbon supports. That’s rare for MOF-derived catalysts and could eliminate a major failure mode in commercial stacks.”
the synthesis route—hydrothermal growth of Ni-Co-ZIF67 on pre-delaminated MXene flakes followed by low-temperature pyrolysis (400°C in Ar)—is compatible with existing wet-chemical manufacturing lines. Unlike high-temperature carbide-derived MXene production, this method avoids HF etching, reducing safety hazards and enabling potential adoption in academic and industrial labs alike. GitHub repositories hosting the synthesis protocols and characterization scripts (e.g., Tsinghua-ElectroCat/Ni-Co-ZIF67-MXene) have already seen 200+ stars, indicating strong open-source traction in the electrocatalysis community.
Broader Impact: Catalyst Design Beyond Oxygen Evolution
The electronic modulation strategy demonstrated here—using transition metal doping to tune the d-band center of MOF-derived frameworks on conductive 2D substrates—has direct implications for other electrochemical reactions. Preliminary DFT calculations suggest the same Ni-Co-ZIF67/MXene platform could achieve overpotentials <300 mV for urea oxidation reaction (UOR) and <200 mV for hydrazine oxidation, opening pathways for wastewater treatment-coupled hydrogen production. As noted by Professor Yang Shao-Horn of MIT in a commentary on the study:
“This work exemplifies how atomic-scale interface design can transcend traditional scaling relations. By decoupling activity from stability through scaffold engineering, we’re seeing a new paradigm where MOFs aren’t just precursors but active, tunable components.”
Critically, the approach avoids the pitfalls of many “high-activity” nanomaterials that degrade under realistic conditions. The MXene’s surface termination groups (–OH, –F) anchor the ZIF67 nanoparticles via coordination bonds, preventing agglomeration even at high anodic potentials where conventional carbon supports corrode. This interfacial stability mechanism could inform the design of catalysts for other sluggish anodic reactions, such as ammonia oxidation or organic electrosynthesis, where catalyst longevity remains a key barrier to industrial deployment.
The Takeaway: A Blueprint for Sustainable Electrocatalysis
This research advances the field not by chasing record-breaking activity numbers in isolation, but by establishing a design principle: synergistic interface engineering between tunable MOF derivatives and conductive 2D substrates can simultaneously optimize intrinsic activity, charge kinetics, and long-term stability. For the green hydrogen economy, where catalyst cost and durability dictate scalability, such innovations are essential to meet the International Energy Agency’s (IEA) net-zero scenario requiring 500 GW of electrolyzer capacity by 2030. While challenges remain in scaling MXene production and validating performance under dynamic renewable energy inputs, the Ni-Co-ZIF67/MXene system offers a compelling, evidence-based pathway toward affordable, efficient water splitting—one atom at a time.