“`html
International Research Collaboration Deepens Understanding of Cancer Development
Table of Contents
- 1. International Research Collaboration Deepens Understanding of Cancer Development
- 2. the Scope of the Investigation
- 3. key Findings and breakthroughs
- 4. How does multi‑omics profiling help decode pancreatic tumor heterogeneity?
- 5. Decoding Pancreatic Tumor Heterogeneity: A Multi‑Institutional Multi‑omics landscape
- 6. The Challenge of Intra-tumoral Heterogeneity
- 7. multi-Omics Approaches: A Deeper Dive
- 8. Multi-Institutional Collaboration: The Power of Scale
- 9. Identifying Novel Molecular Subtypes
- 10. Clinical Implications & Future Directions
- 11. Real-World Example: The COMPASS Study
- 12. Benefits of Multi-Omics Profiling
A sweeping, multinational study involving researchers from the United States and Germany has revealed crucial insights into the complex mechanisms driving cancer progression. The collaborative effort, focused on identifying key biological pathways and potential therapeutic targets, promises to accelerate the development of more effective cancer treatments. This groundbreaking research highlights the increasing importance of international cooperation in tackling global health challenges.
the Scope of the Investigation
The investigation, which commenced several years ago, brought together specialists from institutions including Stanford University, the Technical university of Munich, and the Massachusetts Institute of Technology. Researchers meticulously analyzed tumor samples and genetic data, utilizing cutting-edge technologies to explore the intricate interplay between genetic mutations, cellular signaling, and the tumor microenvironment. The project’s breadth involved a diverse range of expertise including pediatrics, genetics, internal medicine, surgery, and oncology.
key Findings and breakthroughs
Scientists pinpointed
How does multi‑omics profiling help decode pancreatic tumor heterogeneity?
Decoding Pancreatic Tumor Heterogeneity: A Multi‑Institutional Multi‑omics landscape
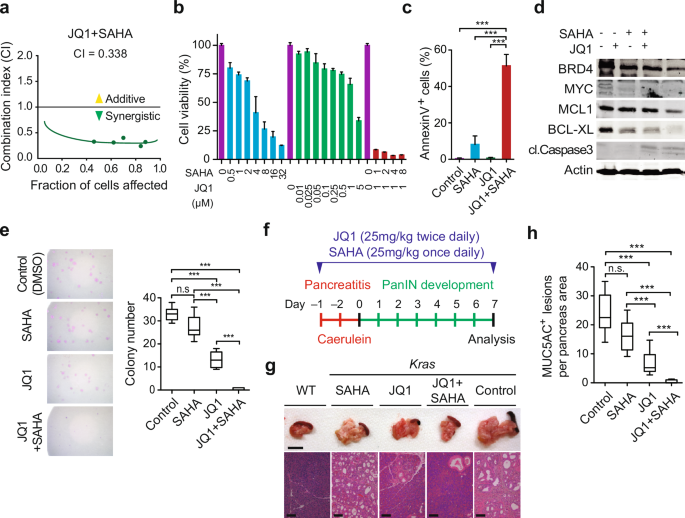
Pancreatic ductal adenocarcinoma (PDAC), one of the most aggressive cancers, presents a significant clinical challenge largely due to its remarkable heterogeneity. This isn’t simply variation between patients, but also within a single tumor. Understanding this complexity is crucial for personalized medicine and improved treatment outcomes. Recent advancements in multi-omics profiling are finally allowing us to dissect this intricate landscape.
The Challenge of Intra-tumoral Heterogeneity
Traditionally, a single biopsy was used to characterize a pancreatic tumor.However, this approach often provides an incomplete picture. Tumors aren’t uniform masses; they’re ecosystems of cells, each with unique genetic, epigenetic, and proteomic profiles. This intra-tumoral heterogeneity impacts:
* Treatment Response: Different regions of the tumor may respond differently to chemotherapy or radiation.
* Metastasis: Heterogeneity fuels the evolution of aggressive, metastatic subclones.
* Drug Resistance: The presence of pre-existing resistant cells can lead to treatment failure.
multi-Omics Approaches: A Deeper Dive
Multi-omics integrates data from various “omics” layers – genomics, transcriptomics, proteomics, metabolomics, and epigenomics – to provide a holistic view of the tumor. This approach is proving invaluable in unraveling pancreatic cancer’s complexities.
1. Genomics: Whole-genome sequencing identifies mutations in key genes like KRAS, TP53, SMAD4, and CDKN2A. However, mutations alone don’t tell the whole story.
2. Transcriptomics (RNA-Seq): Analyzing gene expression reveals which genes are actively being used by cancer cells.This can identify altered signaling pathways and potential drug targets. Spatial transcriptomics, a newer technique, maps gene expression within the tumor tissue, revealing regional differences.
3. Proteomics: Measuring protein levels provides a functional readout of the genome and transcriptome. Proteomic analysis can identify post-translational modifications and protein isoforms that influence cancer behavior.
4. Metabolomics: This examines the small molecule metabolites present in the tumor microenvironment. Metabolites reflect the tumor’s metabolic state and can reveal vulnerabilities.
5. Epigenomics: Epigenetic changes (DNA methylation, histone modifications) alter gene expression without changing the DNA sequence itself.These changes are frequently enough reversible and can contribute to tumor heterogeneity and drug resistance.
Multi-Institutional Collaboration: The Power of Scale
Analyzing pancreatic tumor heterogeneity effectively requires large datasets. This is where multi-institutional collaborations become essential. Pooling data from multiple centers allows researchers to:
* Increase Statistical Power: Identify subtle but significant patterns that might be missed in smaller studies.
* Improve Generalizability: Ensure findings are applicable to a broader patient population.
* Validate Discoveries: Replicate results across self-reliant cohorts.
Recent work, as highlighted in publications like those found on PubMed (https://pubmed.ncbi.nlm.nih.gov/39253579/), emphasizes the recapitulation of molecular spectra across different omics levels through these collaborative efforts. This is a crucial step towards translating research findings into clinical practice.
Identifying Novel Molecular Subtypes
Multi-omics profiling is leading to the identification of distinct molecular subtypes of pancreatic cancer, beyond the conventional classifications. these subtypes are characterized by unique genomic, transcriptomic, and proteomic signatures. Examples include:
* Classical Subtype: Typically characterized by KRAS mutations and activation of the KRAS signaling pathway.
* Basal-like Subtype: Often associated with TP53 mutations and a more aggressive clinical course.
* Squamous Subtype: Displays features of squamous cell differentiation.
* immune-Inflamed Subtype: exhibits a strong immune cell infiltrate.
Identifying a patient’s subtype can help predict their response to specific therapies and guide treatment decisions.
Clinical Implications & Future Directions
The ultimate goal of decoding pancreatic tumor heterogeneity is to improve patient outcomes. Here’s how multi-omics is paving the way:
* Personalized Treatment Strategies: Tailoring therapy based on a patient’s tumor subtype and specific molecular alterations.
* Biomarker Finding: Identifying biomarkers that predict treatment response or disease progression.
* Drug Growth: Developing targeted therapies that specifically address the vulnerabilities of different subtypes.
* early Detection: Identifying biomarkers for early detection of pancreatic cancer, when treatment is most effective.
Looking ahead, advancements in single-cell multi-omics technologies will provide even greater resolution, allowing us to characterize the heterogeneity within individual cells. Integrating artificial intelligence and machine learning will be crucial for analyzing the vast amounts of data generated by these technologies.
Real-World Example: The COMPASS Study
The Consortium of Pancreatic cancer Stem Cell (COMPASS) study exemplifies the power of multi-institutional collaboration. This initiative, involving multiple research centers, aims to comprehensively characterize pancreatic cancer stem cells and their role in tumor heterogeneity and metastasis. By combining genomic, transcriptomic, and proteomic data from a large cohort of patients, COMPASS is identifying novel therapeutic targets and biomarkers.
Benefits of Multi-Omics Profiling
* Improved diagnostic Accuracy: more precise characterization of tumor subtypes.
* Enhanced Prognostic Prediction: Better assessment of disease risk and potential outcomes.
* Optimized Treatment Selection: Personalized therapy based on individual tumor