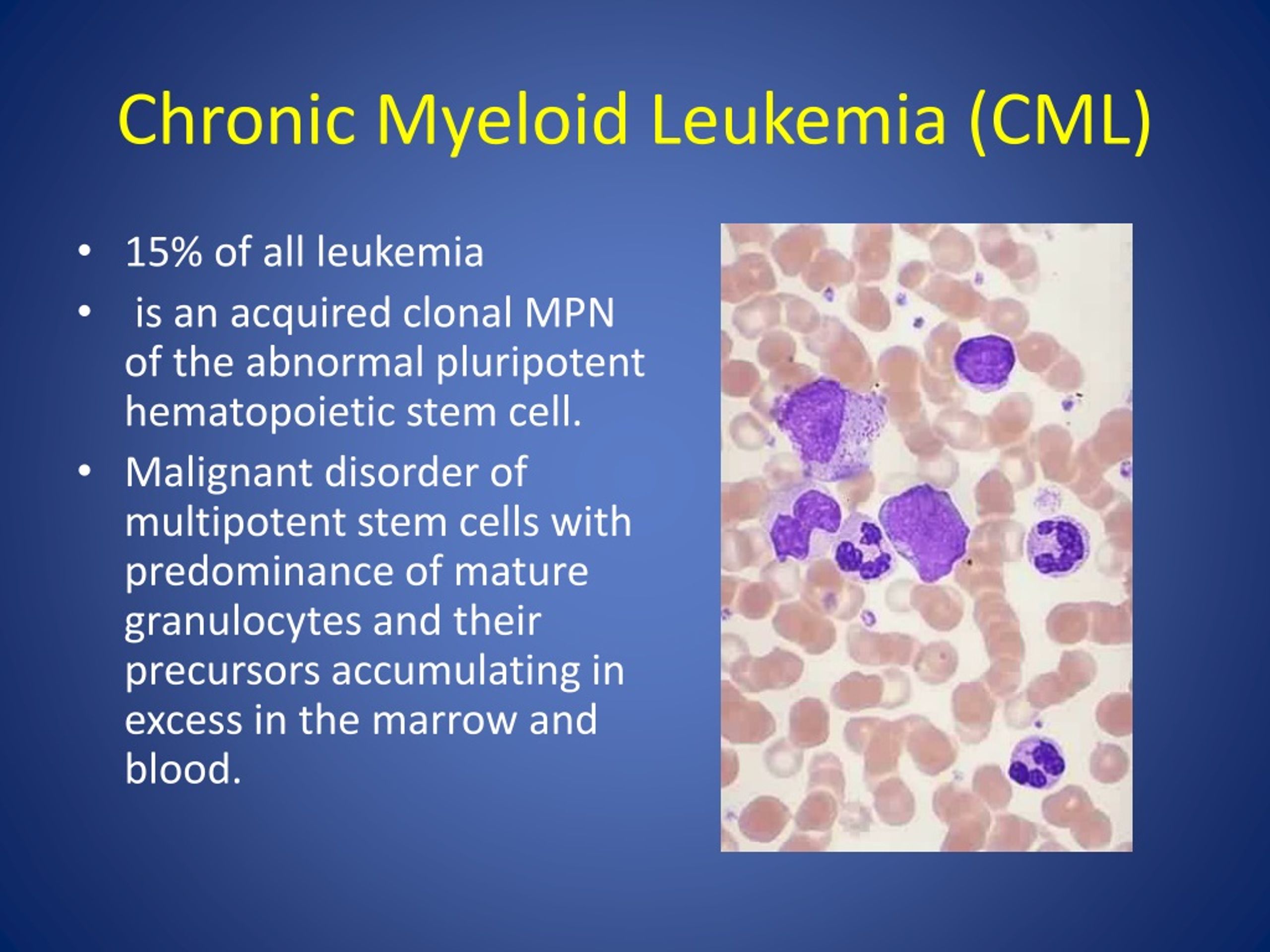
Recent clinical insights published this week highlight the targeting of PELI1 as a breakthrough strategy for Chronic Myeloid Leukemia (CML). By inhibiting this specific protein, researchers aim to eliminate resistant leukemic stem cells that evade standard Tyrosine Kinase Inhibitors (TKIs), offering a potential path toward a complete cure rather than lifelong management.
For the majority of CML patients, the introduction of Tyrosine Kinase Inhibitors (TKIs) transformed a once-fatal diagnosis into a manageable chronic condition. However, a persistent clinical challenge remains: the “TKI-resistant” population. While TKIs effectively clear the bulk of the cancer, they often fail to eradicate the leukemic stem cells (LSCs), which act as a reservoir for disease relapse. The identification of PELI1—an E3 ubiquitin ligase—as a driver of this resistance provides a precise molecular handle to dismantle the survival mechanisms of these stubborn cells.
In Plain English: The Clinical Takeaway
- Targeting the “Root”: While current drugs kill the visible cancer, PELI1 inhibitors target the “stem cells” that cause the cancer to return.
- Overcoming Resistance: This approach helps patients who have stopped responding to standard medications (TKIs).
- Not a Standalone Yet: This is likely to be used as a “combination therapy” alongside existing drugs, not as a total replacement.
The Molecular Mechanism: How PELI1 Fuels Leukemia Survival
To understand the significance of PELI1, we must look at the cellular machinery of the cancer cell. PELI1 (Pellino-1) functions as an E3 ubiquitin ligase. In simple terms, this is a protein that acts as a “molecular tagger,” marking other proteins for degradation or modification. In CML, PELI1 is frequently overexpressed, meaning the body produces too much of it.

The danger of PELI1 lies in its ability to activate the NF-κB pathway—a primary signaling route that tells a cell to survive and proliferate. When PELI1 is hyperactive, it prevents apoptosis (programmed cell death), essentially granting the leukemic stem cell “immortality” even when exposed to high doses of TKI medication. By utilizing a small-molecule inhibitor to block PELI1, clinicians can effectively “strip” the cell of its protective shield, making it susceptible to death once again.
This mechanism of action is particularly critical because it addresses the BCR-ABL1 mutation—the hallmark of CML. While TKIs target the BCR-ABL1 protein directly, PELI1 acts as a secondary survival circuit. Blocking both simultaneously creates a “synthetic lethality” where the cancer cell has no remaining escape routes.
Bridging the Gap: Global Access and Regulatory Hurdles
The transition from laboratory discovery to bedside application varies significantly across global healthcare systems. In the United States, the FDA often utilizes “Fast Track” or “Orphan Drug” designations for therapies targeting resistant CML, which could accelerate the approval of PELI1 inhibitors. In contrast, the European Medicines Agency (EMA) typically requires more extensive comparative efficacy data against existing second- and third-generation TKIs before granting market authorization.
For patients under the NHS in the UK, access to such innovative therapies usually depends on the NICE (National Institute for Health and Care Excellence) cost-effectiveness analysis. Because PELI1 inhibitors target a specific subset of resistant patients, the pricing model will be critical in determining whether these drugs are available on the national formulary or restricted to private clinical trials.
“The ability to target the leukemic stem cell is the ‘Holy Grail’ of CML research. If One can successfully inhibit PELI1 without compromising the patient’s healthy immune response, we move from treating a chronic disease to achieving a treatment-free remission.”
Comparative Analysis: Standard TKI vs. PELI1 Inhibition
The following table summarizes the clinical distinction between current standard-of-care treatments and the emerging PELI1-targeted approach.
| Feature | Standard TKI Therapy | PELI1 Targeted Inhibition |
|---|---|---|
| Primary Target | BCR-ABL1 Fusion Protein | E3 Ubiquitin Ligase (PELI1) |
| Cellular Focus | Bulk Leukemic Cells | Leukemic Stem Cells (LSCs) |
| Clinical Goal | Disease Stabilization / Control | Eradication of Resistance / Cure |
| Common Limitation | Acquired Drug Resistance | Potential for Systemic Inflammation |
| Current Status | FDA/EMA Approved (Standard) | Preclinical / Early Phase Trials |
Funding, Bias, and Scientific Integrity
Transparency in funding is paramount to avoid “industry bias” in medical reporting. Much of the foundational research into PELI1 has been driven by academic grants from the National Institutes of Health (NIH) and various university-affiliated cancer centers. However, as these targets move toward Phase II and III clinical trials, private biotechnology firms typically acquire the licensing. We see essential for patients and providers to monitor whether trial results are published in peer-reviewed journals like The Lancet or if they are released primarily via corporate press releases, which may omit negative data.
Contraindications & When to Consult a Doctor
While PELI1 inhibition is promising, it is not without risk. Because PELI1 plays a role in the innate immune system’s response to bacteria, systemic inhibition could theoretically lead to immunocompromise (a weakened immune system). Patients with existing severe autoimmune disorders or those currently undergoing aggressive chemotherapy may face higher risks of opportunistic infections.

Consult your hematologist immediately if you experience:
- Unexplained persistent fever or chills (signs of neutropenia).
- Rapidly increasing white blood cell counts despite TKI adherence.
- Severe fatigue or bruising that suggests bone marrow suppression.
The Path Forward: Toward Treatment-Free Remission
The targeting of PELI1 represents a shift in the oncology paradigm: moving from “blunt force” chemotherapy to “surgical” molecular intervention. We are entering an era where the goal is no longer just to keep the patient alive, but to achieve Treatment-Free Remission (TFR)—allowing patients to stop medication entirely without the disease returning.
While we must remain cautiously optimistic and wait for robust, double-blind placebo-controlled trial data, the molecular evidence is compelling. By closing the “survival gap” left by TKIs, PELI1 inhibitors could redefine the survival curve for CML patients worldwide.
References
- PubMed Central (PMC) – Hematology and Oncology Research
- World Health Organization (WHO) – International Classification of Haematological Disorders
- Centers for Disease Control and Prevention (CDC) – Cancer Statistics and Surveillance
- The European Journal of Medicine (EMJ) – Clinical Research Archives

